Clear Sky Science · en
Weaning drives microbiome-mediated epigenetic regulation to shape immune memory in mice
Why the First Solid Meals Matter
When a baby—mouse or human—moves from milk to solid food, their gut becomes home to a far richer cast of microbes. This new community doesn’t just help digest dinner; it sends signals that can sculpt the body’s defences for life. This study in mice shows that the brief period around weaning acts like an immune “training camp,” where gut bacteria permanently tune intestinal cells through subtle chemical marks on DNA. Disrupting this window, for instance with early antibiotics, can weaken gut defences and raise the risk of bowel inflammation and cancer later on.
The Changing Life of Gut Microbes
Before weaning, a young mouse’s intestine is relatively simple, nourished by mother’s milk and a limited set of microbes. After weaning, new foods bring in many more bacterial species and microbial products. The researchers compared mice raised in normal, microbe-rich conditions with mice kept germ-free. They focused on long-lived intestinal stem cells, which continually regenerate the gut lining. Although the overall DNA chemistry of these cells looked similar with or without microbes, specific stretches of DNA—control regions that act like volume knobs for nearby genes—were noticeably different when microbes were present. Many of these regions sat near genes involved in recognising and responding to microbes.

How Microbes Write Lasting Notes on DNA
The team found that after weaning, certain DNA control regions in stem cells lost tiny chemical tags called methyl groups. This “demethylation” occurred especially at sites that regulate genes used to present microbial fragments to immune cells. Once these regions were stripped of methyl tags, the change was passed down as stem cells divided and matured into the everyday cells lining the gut. As a result, adult intestinal cells in microbe-exposed mice were primed to switch on defence genes quickly, even long after the initial weaning period had passed. In contrast, germ-free mice, or mice whose guts were colonised only later in life, failed to develop this durable priming to the same extent.
A Brief Burst of Signals with Long-Term Impact
What drives these DNA changes? During weaning, the expanding microbiome sparks a short-lived wave of inflammatory messengers, including one called interferon-gamma. The authors blocked this signal in young mice just after weaning and found that the critical demethylation and gene priming largely disappeared; blocking it in adults had little effect. In miniature gut “organoids” grown in dishes, exposure to low levels of interferon-gamma was enough to trigger rapid demethylation and long-lasting readiness of key genes. After this treatment, the organoids responded faster and more strongly to later challenges, even to different types of signals—a behaviour akin to a trained memory in the gut lining.
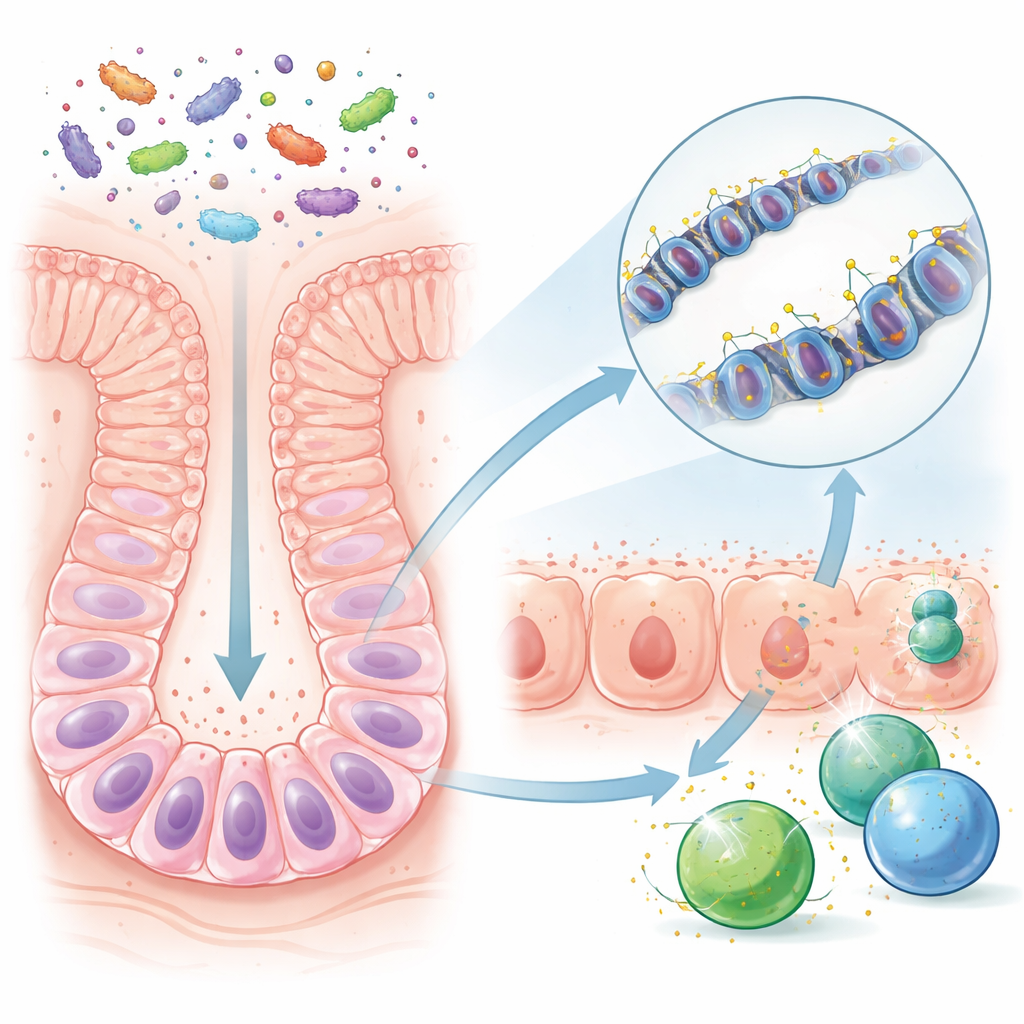
Antibiotics, Missing Microbes, and a Vulnerable Gut
The study also tested what happens when this early-life choreography is disturbed by antibiotics. Mice given low-dose penicillin from before birth through weaning retained many microbes overall but lost specific groups of Gram-positive bacteria that are strong stimulators of interferon-gamma and producers of metabolites that aid DNA demethylation. These mice showed higher DNA methylation at the crucial control regions, weaker expression of antigen-presenting genes in their gut lining, and fewer interferon-gamma–producing immune cells. When later exposed to a chemical that induces colitis, they developed more severe and longer-lasting disease, and they were more prone to colon tumours.
What This Means for Lifelong Gut Health
Taken together, the work paints the post-weaning period as a narrow but powerful window during which diet-driven changes in gut microbes leave lasting marks on intestinal stem cells. These epigenetic marks do not constantly push immune genes into overdrive; instead, they keep the system on alert, ready to mount a swift, balanced response to future threats. Interfering with this early microbial training—through delayed colonisation or broad antibiotic use—can compromise this built-in memory and leave the gut more susceptible to chronic inflammation and cancer. Though the study was done in mice, it raises important questions about how infant feeding choices, microbiome-targeted therapies, and early antibiotic exposure might shape human intestinal health for decades.
Citation: Yang, L., Peery, R.C., Zhou, S. et al. Weaning drives microbiome-mediated epigenetic regulation to shape immune memory in mice. Nat Microbiol 11, 1064–1079 (2026). https://doi.org/10.1038/s41564-026-02295-6
Keywords: gut microbiome, weaning, epigenetic memory, intestinal immunity, early-life antibiotics