Clear Sky Science · en
IL-1 delineates squalene-based adjuvant efficacy and reactogenicity in a cell-type-specific manner
Why vaccine add-ons matter
Modern vaccines often need a boost to work their best, and that boost comes from ingredients called adjuvants. While these helpers can make vaccines far more protective, they can also cause sore arms or brief fevers. This study looks closely at a widely used class of oil-based adjuvants and asks a key question: can we keep their strong protection while dialing down the side effects?
Oil droplets that wake up the immune system
The researchers focused on squalene-based adjuvants—tiny oil-in-water droplets already used in flu and COVID-19 vaccines. Two of these, including the COVID-19 adjuvant A-910823, contain a form of vitamin E called α-tocopherol, which had been linked to stronger antibody responses but also to more inflammation. Using a COVID-19 spike protein vaccine in mice, the team compared adjuvants with and without α-tocopherol and examined the nearby muscle and draining lymph nodes, where immune responses are first organized. They found that α-tocopherol made a big difference: it switched on many more genes tied to inflammation and cell movement, especially in lymph nodes.
The signal called IL-1 at the center of the story
Among all the gene changes, one pathway stood out—signaling driven by a family of molecules known as IL-1. In mice given A-910823, both IL-1α and IL-1β were strongly increased, but in different cell types. IL-1α came mainly from eosinophils, a type of white blood cell best known from allergy and asthma. IL-1β, in contrast, rose in several myeloid cells, including neutrophils, monocytes, and dendritic cells. When the researchers removed α-tocopherol from the adjuvant, IL-1β in particular dropped sharply. Similar patterns appeared when they tested human-approved mimics of other squalene adjuvants, showing that this behavior is a shared feature of α-tocopherol–containing oil emulsions. 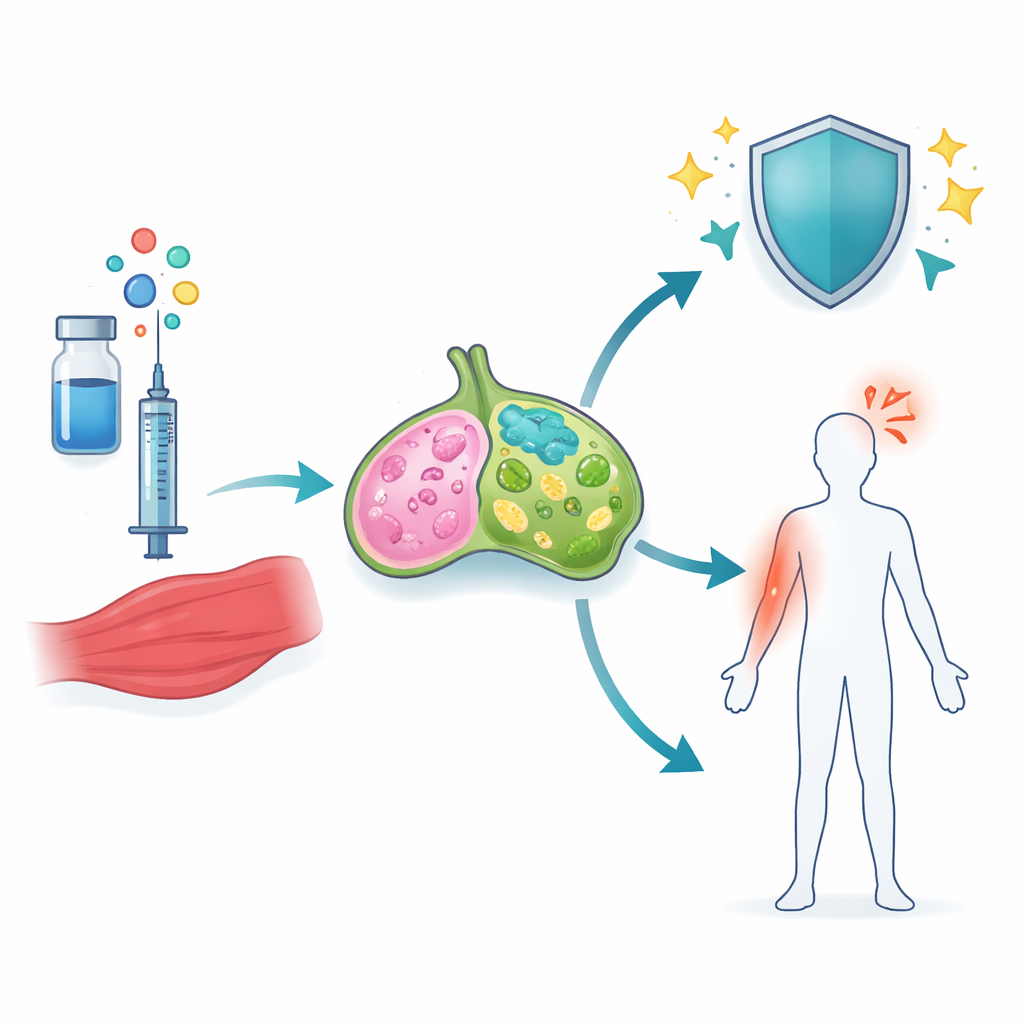
Separating protection from side effects
The team then asked how these IL-1 signals translate into real-world outcomes: protective antibodies and short-term reactions like swelling and fever. By using mice lacking specific receptors or signaling molecules, and by blocking IL-1β, they mapped out distinct circuits. Strong antibody responses and the formation of specialized helper T cells and germinal center B cells were driven by an IL-1β–dependent pathway acting through a key adaptor protein, MyD88, inside CD11c-positive cells such as dendritic cells. Local arm swelling, however, depended mainly on IL-1α coming from eosinophils near the injection site. Systemic fever followed yet another route: IL-1β triggered the release of IL-6, which in turn activated the enzyme COX2, a well-known driver of fever that can be blocked by common anti-inflammatory drugs. When IL-6 or COX2 was inhibited, fever was reduced but antibody responses remained strong. 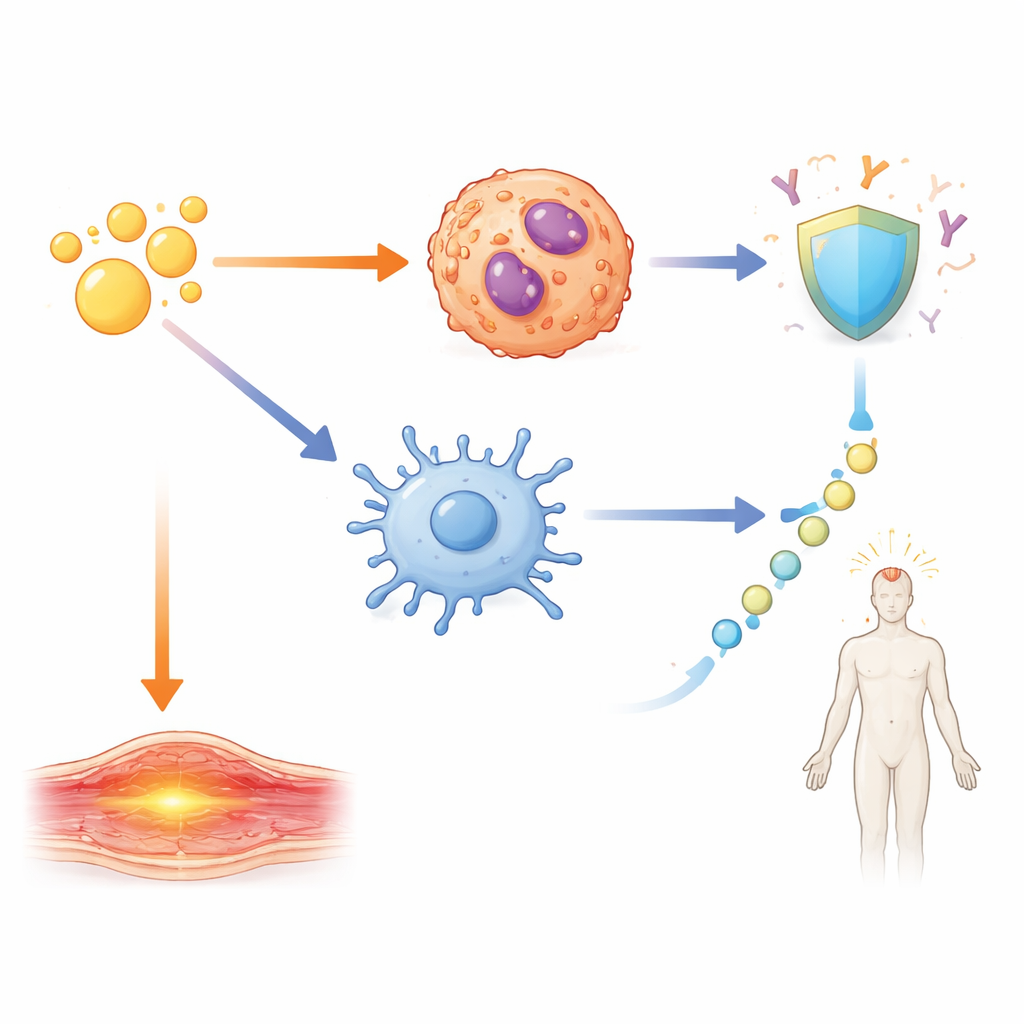
From mice to humans
To check whether these findings might matter in people, the researchers exposed human blood cells from healthy volunteers to different adjuvants in the lab. Just as in mice, α-tocopherol–containing squalene emulsions provoked eosinophils to produce IL-1α, while similar emulsions lacking α-tocopherol did not. Other types of adjuvants, including aluminum salts and saponin-based formulations, produced little or no IL-1α in eosinophils. This suggests that α-tocopherol–driven IL-1 signaling, especially in eosinophils, is a distinctive fingerprint of this adjuvant family and could serve as a marker of local reactogenicity in future human studies.
Designing safer, stronger vaccines
Overall, the study shows that the same adjuvant can use separate cell types and signaling routes to generate protection and to cause short-lived reactions. IL-1β acting through dendritic cells is crucial for strong, long-lasting antibody responses, while IL-1α from eosinophils and an IL-1β–IL-6–COX2 chain govern swelling and fever. For vaccine design, this means that efficacy and reactogenicity are not locked together; they can, in principle, be tuned independently. By targeting or modulating the specific branches that drive side effects—without disturbing the branch that powers immunity—future adjuvants could deliver vaccines that are both highly effective and easier to tolerate.
Citation: Yoshioka, Y., Nishinaka-Yoshioka, A., Kobiyama, K. et al. IL-1 delineates squalene-based adjuvant efficacy and reactogenicity in a cell-type-specific manner. npj Vaccines 11, 67 (2026). https://doi.org/10.1038/s41541-026-01420-0
Keywords: vaccine adjuvants, squalene emulsion, interleukin-1, reactogenicity, alpha-tocopherol