Clear Sky Science · en
Integrated multi-omics and single-cell analysis reveals CDKN2A-mediated cuproptosis mechanisms driving thyroid carcinoma progression
Why Copper and Thyroid Cancer Matter
Thyroid cancer is usually treatable, but a troubling minority of patients develop tumors that recur, spread, or resist standard therapies. At the same time, researchers have discovered a new way cells can die, triggered by the metal copper. This study brings those two stories together: it explores how copper-linked cell death pathways, and the genes that control them, shape the behavior of thyroid tumors and their surrounding immune cells. By combining big-data analysis with single-cell and animal experiments, the authors uncover a surprising gene network that seems to help thyroid cancer grow and spread—and may offer new targets for future treatments. 
Looking at Copper-Linked Genes Across Many Patients
The researchers started with “multi-omics” datasets—large collections of genetic and molecular measurements—from hundreds of thyroid cancer patients. They focused on 19 genes known to be involved in copper-triggered cell death and asked how these genes behaved in tumors compared with normal thyroid tissue. Using statistical clustering, they grouped patients into two molecular subtypes based on the activity of these copper-related genes. One subtype showed stronger signals related to immune responses, while the other was more tied to changes in metabolism, the cell’s energy and building-block machinery. These distinct patterns suggested that copper biology might influence not just tumor cells themselves, but also how tumors interact with the body’s defenses.
Immune Surroundings and Patient Outlook
Next, the team examined how these copper-linked subtypes related to the tumor microenvironment—the mix of immune cells and supporting tissue around the cancer. They found that one subtype had higher expression of immune “checkpoint” molecules, which are key targets of modern immunotherapy drugs, and greater infiltration by many types of immune cells. They also built a risk score based on copper-associated genes that could separate patients with better versus worse survival. Patients with high scores tended to have shorter overall survival and a different balance of immune cells in their tumors, including fewer cancer-fighting T cells. This suggests that copper-related pathways could influence how well the immune system can recognize and attack thyroid tumors.
Zeroing In on a Key Trouble-Making Gene
Among the 19 copper-linked genes, one stood out: CDKN2A. Unlike most of the others, which were reduced in tumors, CDKN2A was consistently elevated in thyroid cancer and strongly associated with poorer outcomes. Single-cell sequencing—an approach that reads out gene activity in thousands of individual cells—showed that CDKN2A was especially active in thyroid tumor cells and certain immune and support cells. In laboratory experiments, boosting CDKN2A in thyroid cancer cells made them grow, invade, and migrate more aggressively, both in dishes and in mice. Silencing CDKN2A had the opposite effect, shrinking tumors and reducing lung metastases. These findings are striking because CDKN2A is classically viewed as a “brake” on cell division in many cancers; here it appears to act more like an accelerator, at least in the thyroid setting.
A Hidden RNA Circuit That Feeds Tumor Growth
To understand why CDKN2A is so active, the authors searched for an upstream regulatory circuit. They uncovered a three-part RNA network involving a long noncoding RNA called GAS5, a small regulatory RNA called miR-128-3p, and the CDKN2A gene. In essence, GAS5 acts as a sponge that binds miR-128-3p, preventing this small RNA from attaching to and dampening CDKN2A. When GAS5 levels are high, miR-128-3p is sequestered, CDKN2A escapes control, and cancer cells become more aggressive. When GAS5 is knocked down, miR-128-3p rebounds, CDKN2A levels fall, and tumor cells lose much of their ability to grow and spread. 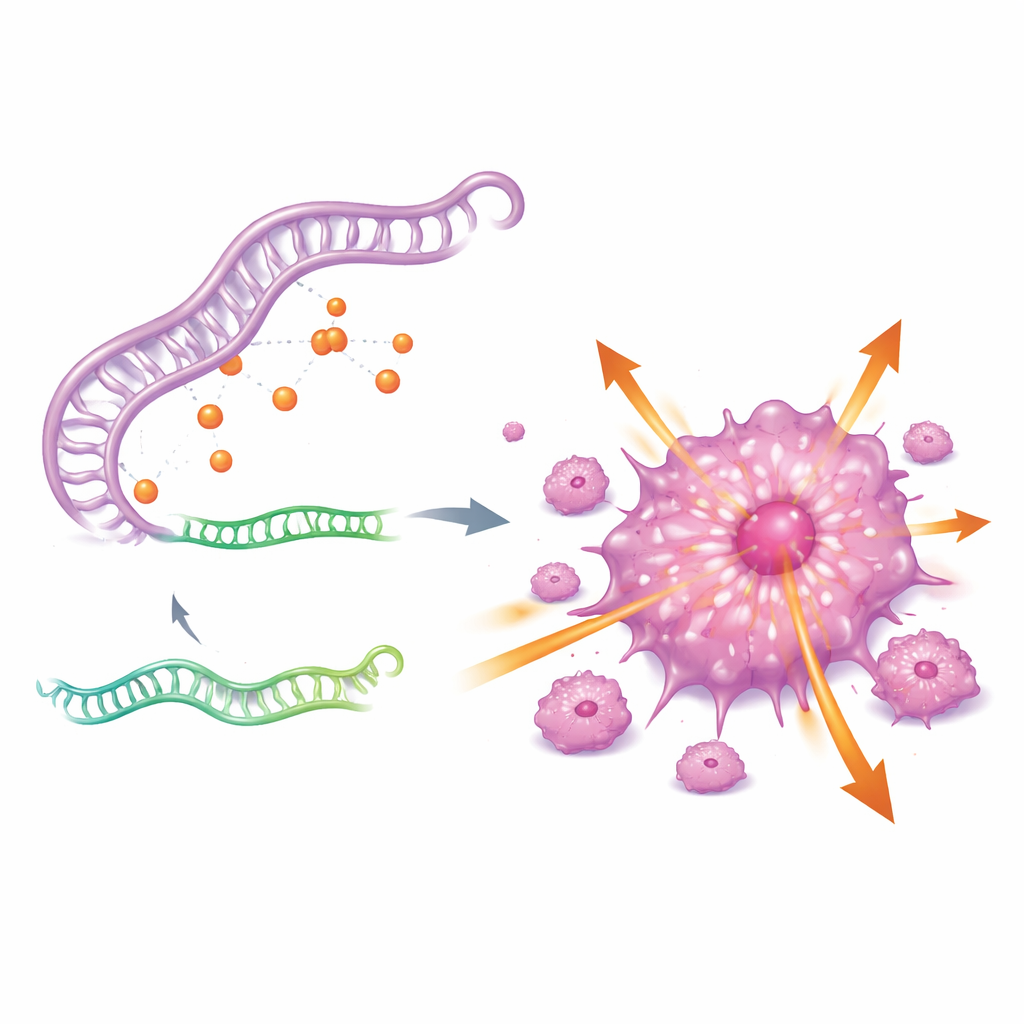
What This Means for Future Care
This work paints a systems-level picture of how a copper-linked cell death pathway, thyroid tumor cells, and surrounding immune cells are intertwined. While the study does not yet prove that copper-triggered cell death is directly driving thyroid cancer, it clearly shows that copper-related genes—and especially the GAS5/miR-128-3p/CDKN2A network—are tied to more dangerous disease. For patients, that could eventually translate into new blood or tissue tests to better gauge risk, and new drugs that disrupt this RNA circuit or modulate copper-related processes to tilt the balance away from tumor growth and toward tumor control.
Citation: Huang, J., Wang, L. Integrated multi-omics and single-cell analysis reveals CDKN2A-mediated cuproptosis mechanisms driving thyroid carcinoma progression. npj Syst Biol Appl 12, 61 (2026). https://doi.org/10.1038/s41540-026-00663-w
Keywords: thyroid cancer, cuproptosis, tumor microenvironment, CDKN2A, noncoding RNA