Clear Sky Science · en
TGF-α/EGFR-mediated lymphatic metastasis reveals a repositionable therapeutic target in breast cancer
Why this study matters for patients
When breast cancer spreads beyond the breast, it often first appears in the lymph nodes. Doctors know that tumors with high levels of a molecule called EGFR tend to behave aggressively, but drugs that shut down EGFR have not worked well against breast cancer. This study uncovers a missing piece of the puzzle: EGFR may not mainly drive tumor growth, but instead help tumor cells “navigate” into lymphatic vessels and lymph nodes. That shift in perspective could open the door to new ways of stopping metastasis early, before it becomes life‑threatening.
A hidden highway for tumor cells
The researchers focused on triple‑negative breast cancer, a hard‑to‑treat subtype that often has high EGFR levels and early lymph node involvement. In a mouse model, they engineered breast cancer cells to overproduce EGFR and implanted them into the mammary gland. Surprisingly, these EGFR‑rich tumors did not grow faster and did not send more metastases to the lungs than control tumors. Instead, far more cancer cells appeared in nearby lymph nodes, even at early time points. Microscopy showed that lymphatic vessels sat mostly around the outer rim of the tumor, suggesting that tumor cells must actively move outwards to find and enter these vessels. 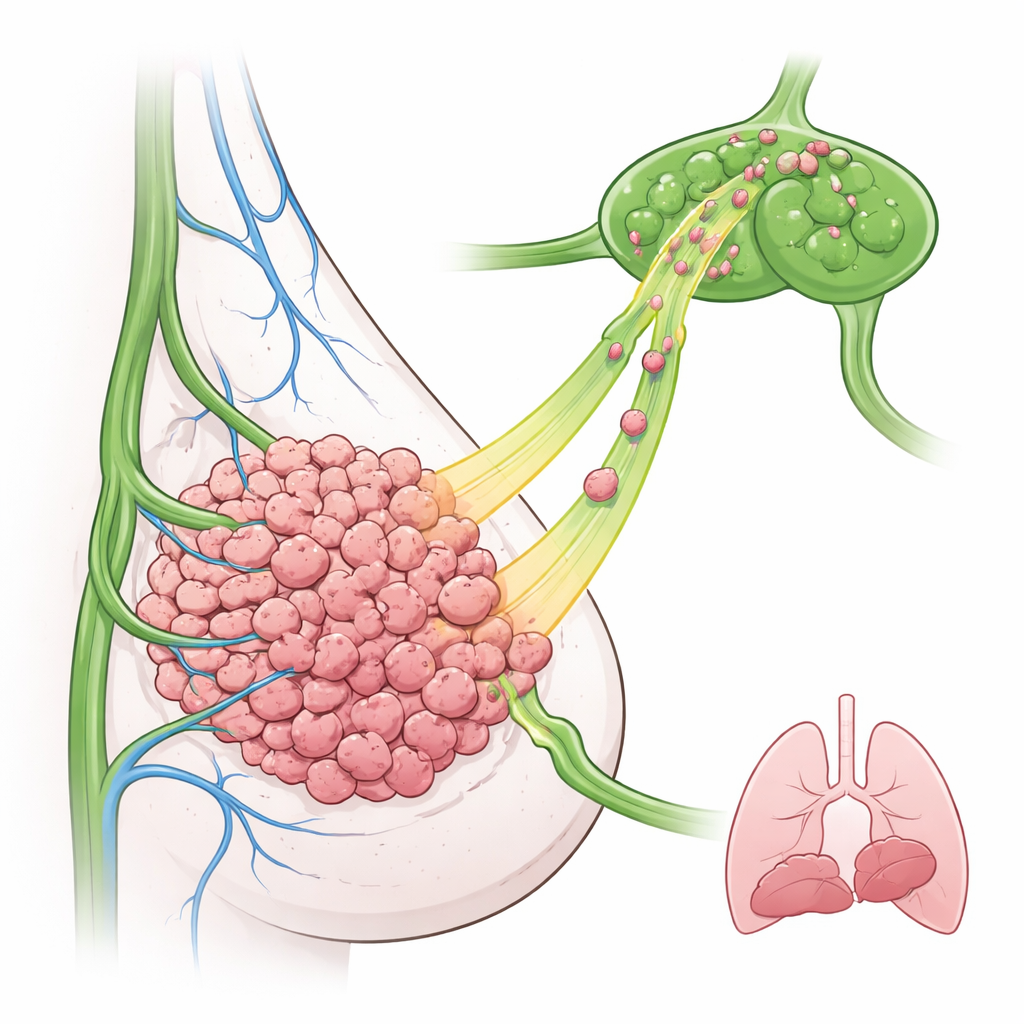
Signals from the lymph vessels themselves
The team then asked whether the lymphatic vessels are just passive pipes or active guides. They studied lymphatic endothelial cells, the cells that line lymphatic vessels, in both mice and human breast tumors. When these cells were exposed to another tumor‑related molecule, TGF‑β, they began producing growth factors that can activate EGFR. One factor, TGF‑α, stood out: it was strongly increased, released into the surrounding fluid, and in tumor tissue it was concentrated on lymphatic, not blood, vessels. Human data showed that breast cancers with higher TGFA levels, especially the basal‑like subtype related to triple‑negative disease, were linked to worse patient outcomes. This pointed to TGF‑α as a likely “scent trail” leading EGFR‑positive tumor cells into lymphatics.
How tumor cells follow the trail
To test this idea, the researchers used invasion and live‑cell migration assays. They placed EGFR‑overexpressing tumor cells above a gel and supplied either fluid from lymphatic cells or purified growth factors below. The EGFR‑rich cells invaded strongly toward lymphatic cell fluid and toward TGF‑α alone, moving in a clear, directed fashion rather than wandering randomly. Another factor, CTGF, which is more associated with blood vessels, did not promote directional movement and could even dampen TGF‑α’s effect. Inside the tumor cells, TGF‑α switched on a signaling molecule called STAT3, along with other pathways. When STAT3 was chemically blocked, the tumor cells largely lost their ability to move toward lymphatic signals. The same TGF‑α‑driven, EGFR‑dependent chemotaxis was observed in a human triple‑negative breast cancer cell line, confirming that this behavior is not limited to mice. 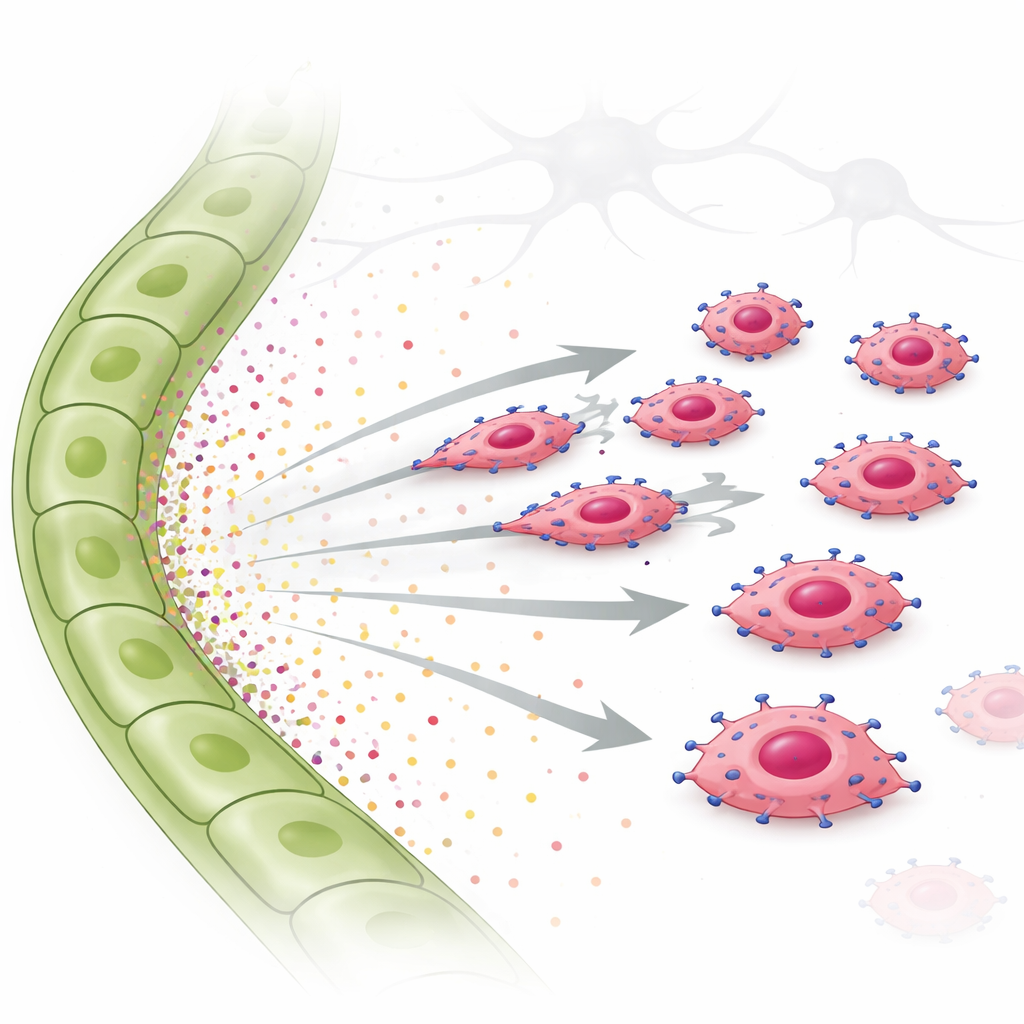
Repurposing an existing antibody
The study also explored whether this guidance system can be interrupted with a drug. The authors used Fepixnebart, a laboratory antibody originally developed to neutralize TGF‑α and a related factor in non‑cancer conditions. In invasion assays, this antibody sharply reduced the ability of EGFR‑positive cells to move toward lymphatic‑derived fluid. In mice, a single dose given when tumor cells were implanted significantly lowered the number of cancer cells found later in nearby lymph nodes, without affecting the primary tumor’s size. At the same time, tumors with high EGFR were associated with tumor‑draining lymph nodes that contained more total cells but a relative shortage of CD8 “killer” T cells and an increase in regulatory T cells, suggesting a shift toward a more suppressive immune environment where cancer can more easily take hold.
What this means for future treatment
Taken together, the work reframes EGFR in triple‑negative breast cancer: rather than simply being a growth accelerator, EGFR helps tumor cells sense and follow a TGF‑α signal emitted by lymphatic vessels, steering them into lymph nodes and accompanying an immune shift that may favor metastasis. Because Fepixnebart and other EGFR‑targeted agents already exist and have been tested for safety in humans, this pathway represents a “repositionable” therapeutic target. Instead of expecting these drugs to shrink established tumors on their own, clinicians might one day use them to block early lymphatic spread and improve the effectiveness of immune‑based therapies, potentially changing how high‑risk breast cancers are managed.
Citation: Shi, W., Pan, Y., Rathod, B. et al. TGF-α/EGFR-mediated lymphatic metastasis reveals a repositionable therapeutic target in breast cancer. npj Breast Cancer 12, 52 (2026). https://doi.org/10.1038/s41523-026-00941-0
Keywords: triple-negative breast cancer, lymphatic metastasis, EGFR signaling, TGF-alpha, tumor immune microenvironment