Clear Sky Science · en
De-repression of protein phosphatase 5 by the chaperone organizer HOP1 activates plant NLR immunity
How Plants Decide Between Growth and Self-Defense
Plants constantly face would‑be invaders—bacteria, fungi and viruses—yet they cannot simply abandon growth to fight every threat. This paper uncovers how a tiny molecular “brake and trigger” system in the model plant Arabidopsis helps decide when to unleash an extreme immune reaction that can save the plant but also stunt its growth. Understanding this switch offers insight into how crops might be bred or engineered to be disease‑resistant without paying too high a price in yield.
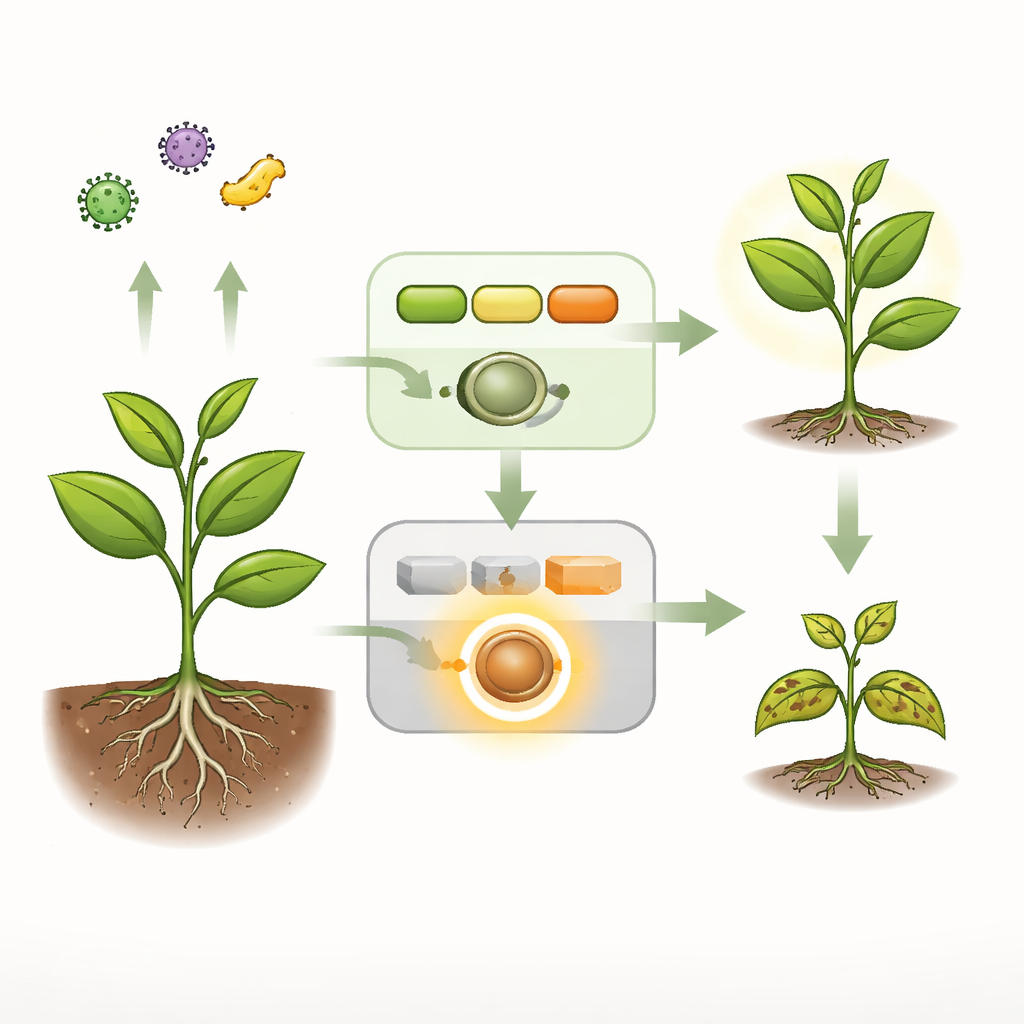
Everyday Alarms Versus Emergency Sirens
Plants run two main security systems. The first is a fast, broad alarm that detects general microbial fingerprints at the cell surface and turns on protective responses. A second, more drastic layer relies on internal sentries called NLR receptors that can trigger powerful, often lethal, defense in cells where pathogens are detected. In healthy Arabidopsis, a signaling chain of three enzymes—collectively called the MEKK1–MKK1/2–MPK4 cascade—both relays the first alarm and quietly keeps one particular NLR receptor, named SUMM2, from firing without cause. When this chain is broken by mutation or by a bacterial effector protein, plants show spontaneous cell death, high defense gene activity and severe dwarfism, all signs of runaway immunity.
A Hidden Enzyme Awakens
The authors discovered that a phosphatase enzyme called LET7, a plant version of the conserved protein PP5, sits at the heart of this runaway response. On its own, LET7 is built like a folded pocketknife: one region (a TPR domain) bends back onto the catalytic region and keeps it largely shut off. Genetic screens showed that plants lacking LET7 no longer suffered from the lethal symptoms caused when the upstream signaling chain is silenced, placing LET7 downstream of that cascade. Biochemical tests revealed that when the catalytic region of LET7 is freed or made hyperactive by specific mutations, plants develop strong defense responses and cell death, whereas a catalytically dead version cannot trigger these effects. Thus, the phosphatase activity of LET7 is both necessary and sufficient to drive autoimmunity.
A Molecular Organizer Releases the Brake
How does the molecular pocketknife open? The team identified a co‑chaperone protein called HOP1 as a key organizer. HOP1 normally helps heat‑shock proteins fold and stabilize other proteins. Here, it latches onto the TPR region of LET7 and outcompetes the internal contact that keeps LET7 closed. Structural modeling and precise amino‑acid swaps on both partners showed that this binding surface is finely tuned: weakening the contact between HOP1 and LET7 reduces LET7 activation. When the MEKK1–MKK1/2–MPK4 chain is intact, the association between HOP1 and LET7 is low and LET7 stays repressed. When the chain is disrupted—by mutations, by overabundance of another scaffold protein, or by a bacterial effector—the HOP1–LET7 interaction intensifies, unleashing LET7’s catalytic activity. Remarkably, the human counterparts of HOP1 and PP5 can substitute for the plant pair, pointing to a conserved activation mechanism across kingdoms.
Cutting a Key Switch and Guarding a Sensor
Once active, LET7 targets a kinase called CRCK3, an enzyme previously linked to the same immune pathway. CRCK3 exists in phosphorylated and dephosphorylated forms, which can be distinguished as two bands on protein gels. In plant cells and test‑tube assays, active LET7 selectively removes phosphate marks from CRCK3, while inactive LET7 does not. Enhancing LET7 activity or co‑expressing it with CRCK3 boosts dephosphorylation and leads to cell death and classic immune markers, but these effects are blunted when HOP1 or the NLR receptor SUMM2 is missing. In parallel, the authors found that both LET7 and HOP1, through their TPR regions, physically bind to SUMM2 and increase its protein abundance without broadly stabilizing other NLR receptors. Thus, the same module that removes phosphate from CRCK3 also helps maintain the sensor that reads that signal.
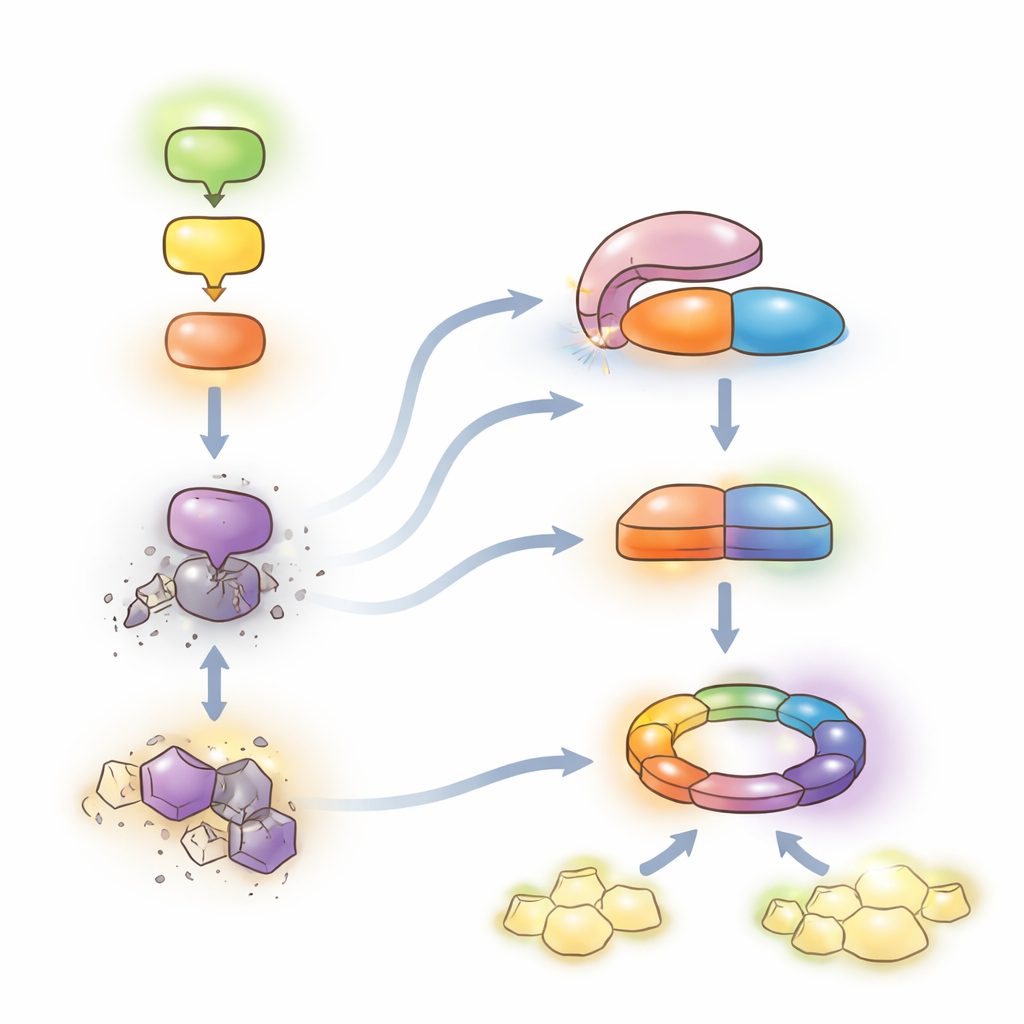
A Delicate Balance Between Protection and Growth
Together, the findings support a model in which the MEKK1–MKK1/2–MPK4 chain restrains a powerful second‑line defense. When this chain is intact, LET7 stays autoinhibited and SUMM2 levels are kept in check, allowing plants to rely mainly on their everyday alarm system while continuing to grow. When pathogens disrupt the chain, HOP1 engages LET7, unlocking its phosphatase activity. LET7 then dephosphorylates CRCK3 and, together with HOP1, stabilizes SUMM2, tipping the system into full NLR‑mediated immunity and local cell death—highly protective but costly to growth. By revealing this conserved “de‑repression” mechanism for PP5 enzymes, the work outlines a molecular lever that could, in principle, be adjusted in crops to strengthen disease resistance while minimizing harmful autoimmunity.
Citation: Yan, Y., Zhao, Z., Yeo, IC. et al. De-repression of protein phosphatase 5 by the chaperone organizer HOP1 activates plant NLR immunity. Nat. Plants 12, 802–817 (2026). https://doi.org/10.1038/s41477-026-02253-4
Keywords: plant immunity, protein phosphatase 5, NLR receptors, signal transduction, Arabidopsis