Clear Sky Science · en
MeCP2 Governs maternal hyperandrogenism-induced cortical defects and behavioral alterations via noncanonical AR-dependent regulation of Mef2c
Why this research matters to families
Many conditions that affect learning, social skills, and behavior are more common in boys than in girls, but the reasons for this sex difference are still unclear. This study uses mice to explore how high levels of male hormones during pregnancy can subtly reshape the developing brain of male offspring, and how a well-known brain protein, MeCP2, helps translate those hormonal signals into lasting changes in brain structure and behavior.
Hormones before birth and later brain health
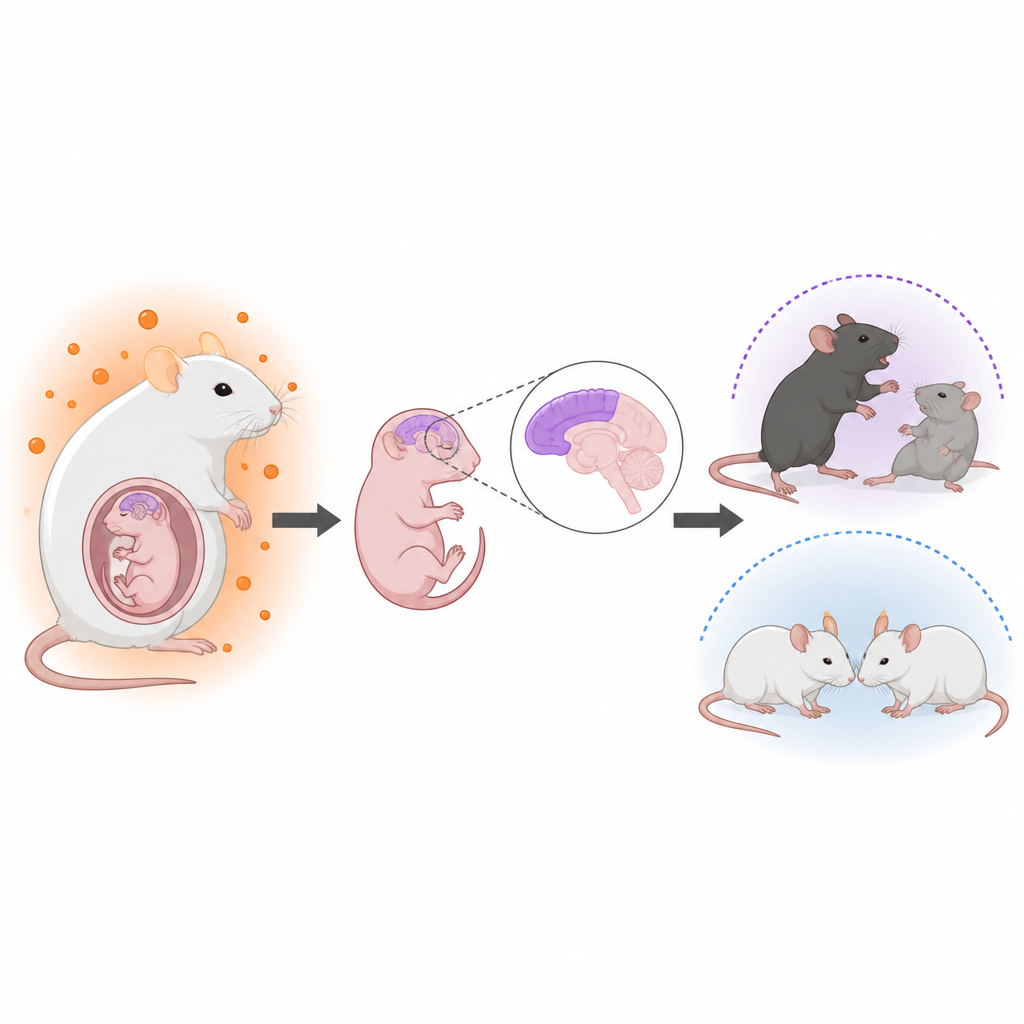
Doctors have long noticed links between elevated male hormones in the womb and a higher chance of neurodevelopmental conditions such as autism. For example, women with polycystic ovary syndrome often have higher androgen levels and their children show a slightly increased risk of autism. However, it has not been clear how these hormones act on the fetal brain or why sons seem more affected than daughters. The authors set out to build a realistic mouse model of maternal hyperandrogenism and then track both brain development and behavior in the offspring.
How the team modeled hormone exposure
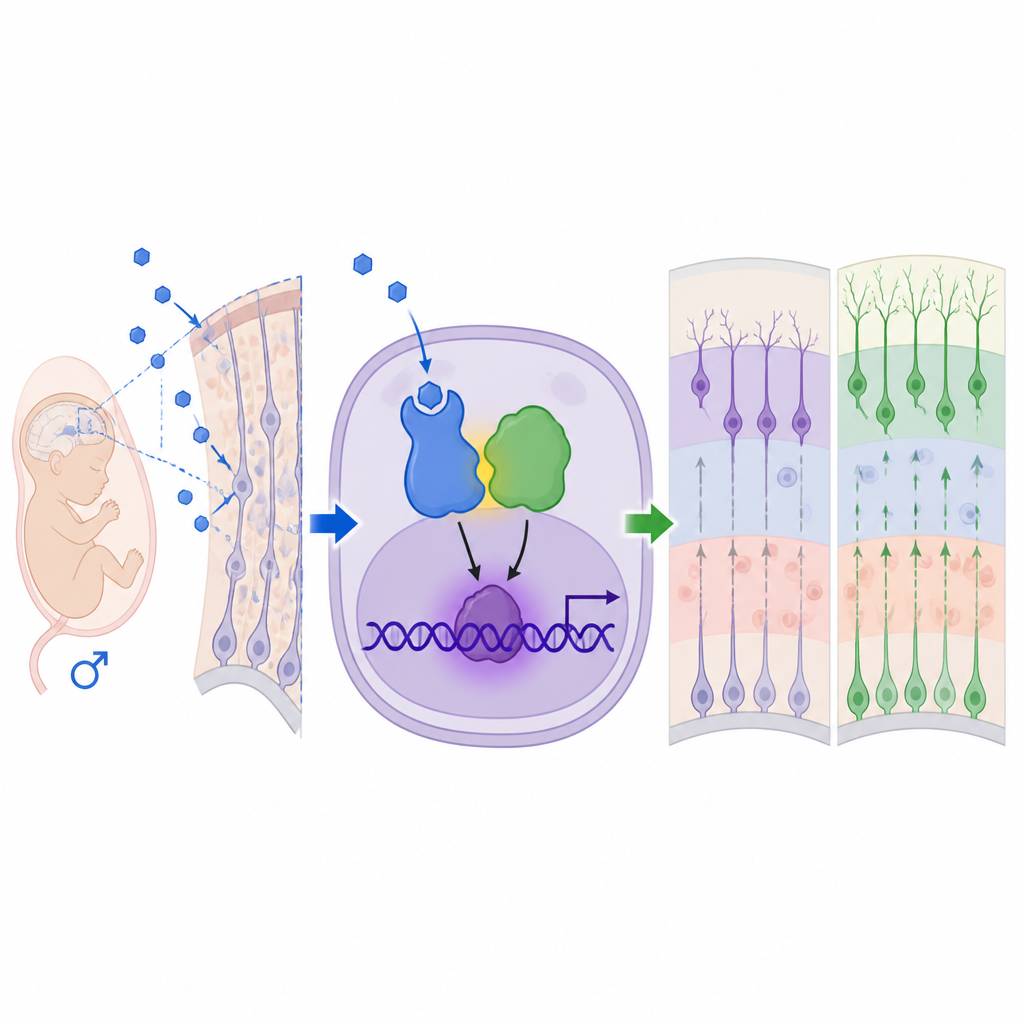
The researchers treated pregnant mice with a drug that blocks aromatase, the enzyme that normally converts testosterone into estrogen. This raised testosterone levels in the mothers, in the amniotic fluid around the fetuses, and in the fetal brains, without changing overall pregnancy outcomes or birth weights. Using genetic labeling and microscopy, they focused on the cerebral cortex, the outer layer of the brain that supports thinking and social behavior. In male offspring, but only mildly in females, they found that neural stem and progenitor cells were pushed to create neurons earlier and in greater numbers, while the pool of stem cells and some later cell types such as astrocytes was reduced. The normal layered arrangement of cortical neurons became noticeably less orderly, even though total brain size did not change.
From changed cortex to altered behavior
As adults, male offspring from hyperandrogenic mothers showed differences in behavior that mirror several features often seen in autism. In social tests, these males spent less time interacting with other mice and showed less interest in new social partners, while their movement levels remained normal. They also displayed more repetitive behaviors, such as burying more marbles, and stronger signs of anxiety in open field and elevated plus maze tests. Female offspring, raised under the same conditions and cross-fostered to control mothers after birth, showed far milder or no such behavioral changes, highlighting a male bias in vulnerability.
A nonstandard pathway inside developing brain cells
To understand how androgens exert these effects, the team zoomed in on androgen receptors, the proteins that sense these hormones. They found higher levels and nuclear localization of androgen receptor in the male fetal cortex, and blocking this receptor with the drug flutamide reversed the excess neurogenesis and most behavioral changes. Gene expression analyses pointed to MEF2C, a transcription factor already linked to neurodevelopmental disorders, as a key downstream target. Surprisingly, androgen receptor did not act alone or through its usual DNA docking sites. Instead, it physically teamed up with MeCP2, a protein encoded by a gene on the X chromosome best known from Rett syndrome and MeCP2 duplication syndrome. Together, androgen receptor and MeCP2 formed an activator complex that bound at a MeCP2-recognized site in the Mef2c regulatory region, boosting MEF2C levels and driving neural stem cells toward neurons.
What happens when MeCP2 itself is increased
The authors then turned to a mouse model carrying an extra copy of MeCP2, which had previously been shown to display increased cortical neurogenesis and autism-like behaviors. They discovered that these MeCP2-duplicated males also had elevated MEF2C and similar cortical changes, even without extra prenatal androgens. Dialing down Mef2c or blocking androgen receptors in these mice normalized cortical layering and eased many of the social, repetitive, and anxiety-like behaviors. This suggests that too much MeCP2 can engage the same hormone-sensitive pathway, amplifying male-specific risk when androgen signals are present.
How this work changes our view of sex differences
Overall, the study reveals a previously unrecognized route by which maternal androgen exposure can shape the male fetal cortex and later behavior. Instead of acting only through its classic targets, the androgen receptor can hijack MeCP2 to switch on MEF2C, thereby tipping the balance of neural stem cell fates and subtly rewiring cortical circuits. Because MeCP2 sits on the X chromosome and is already implicated in several human neurodevelopmental syndromes, this hormone-sensitive partnership offers a concrete molecular explanation for why boys may be more prone to certain brain-based conditions, while also suggesting that both genes and prenatal hormone environments need to be considered together.
Citation: Wang, YM., Jia, Y., Wu, Y. et al. MeCP2 Governs maternal hyperandrogenism-induced cortical defects and behavioral alterations via noncanonical AR-dependent regulation of Mef2c. Nat Commun 17, 4225 (2026). https://doi.org/10.1038/s41467-026-72942-3
Keywords: prenatal androgens, MeCP2, cortical development, MEF2C, autism risk