Clear Sky Science · en
The history and function of a circular RNA
How tiny RNA loops help stressed brains cope
When we are sleep-deprived, injured, or emotionally overwhelmed, brain cells must rapidly adjust their activity to survive and keep circuits functioning. This article describes an unusual molecule in the brain, a circular piece of RNA called Cdr1as, and shows how it works together with other small RNAs to fine‑tune nerve cell communication under stress. Understanding this hidden control system could open new paths for treating stroke, depression, and other brain disorders—and may even yield blood tests that report on brain health from afar.
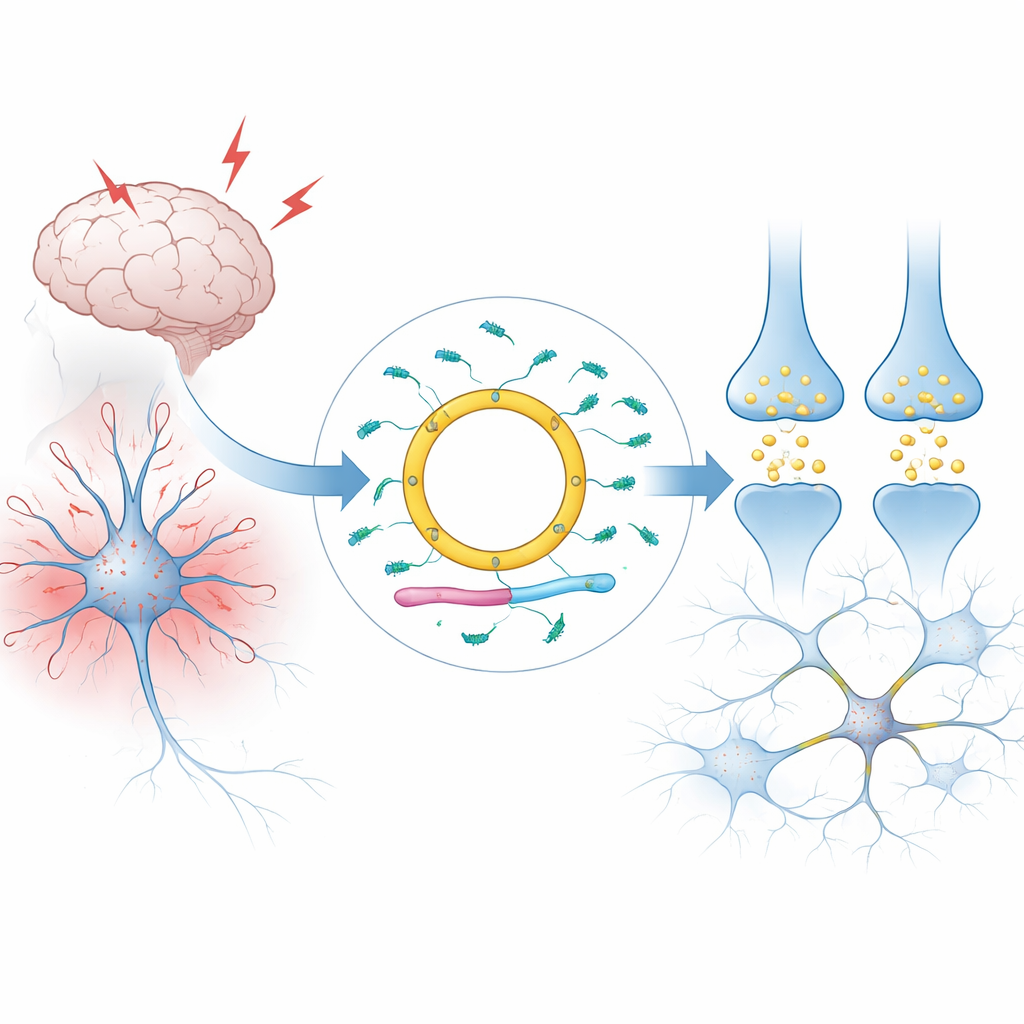
A circular message in brain cells
Most RNAs in our cells are linear: they have two ends and are quickly broken down. Cdr1as is different. It forms a closed loop, making it unusually stable and allowing it to accumulate to very high levels in mammalian brains, especially in specific types of neurons. The authors explain how Cdr1as is produced from a larger RNA unit on the X chromosome by a process that splices its ends together. In humans and mice, nearly all RNA from this locus is converted into the circular form, with barely any leftover linear version. Large surveys of tissues and single cells show that Cdr1as is, for practical purposes, a neuron‑specific molecule that is scarce in other organs and in supporting brain cells such as glia.
A dense web of tiny regulators
Cdr1as gains its importance from the company it keeps. Its circular sequence carries an exceptional number of docking sites for a small regulatory RNA called miR‑7—around seventy in humans and about 130 in mice. miR‑7 belongs to the celebrated family of microRNAs, newly Nobel‑recognized molecules that silence many protein‑coding messages at once. In neurons, miR‑7 targets messages involved in stress responses and the release of the chemical transmitter glutamate. Cdr1as binds miR‑7 in living brain tissue, affecting how long miR‑7 survives, where it is located inside the neuron, and how strongly it can act on its many targets. A second small RNA, miR‑671, can slice and destroy Cdr1as, while a long RNA called Cyrano causes the active breakdown of miR‑7 itself. Together, these three non‑coding RNAs and two microRNAs form a tightly connected circuit.
Keeping nerve activity in balance under pressure
Using mouse models lacking individual pieces of this circuit, the authors summarize a decade of work showing how it shapes brain function. Removing Cdr1as reduces mature miR‑7 levels and disrupts the expression of “immediate early” genes that normally respond to activity and stress. Neurons without Cdr1as fire in a more erratic, overly excitable way, and mice show altered reactions to sensory cues. In contrast, removing Cyrano leads to a strong build‑up of miR‑7, which in turn prevents Cdr1as from accumulating, revealing a feedback loop. Experiments that mimic stroke‑like conditions in dishes of neurons and in living mice demonstrate that levels of Cdr1as, miR‑7, and Cyrano shift in a coordinated, time‑dependent manner. These shifts change glutamate release at synapses, influencing how well neurons survive and how networks recover after injury.
An evolutionary add‑on for fine control
The review also places this circuit in evolutionary context. miR‑7 is ancient, appearing with the first animals that developed centralized nervous systems, and is deeply conserved because it helps control early developmental decisions, such as those guided by the master regulator PAX6. By contrast, Cyrano arose later in vertebrates, miR‑671 even later in mammals with primitive placentas, and Cdr1as only in placental mammals. The authors argue that these newer RNAs were gradually layered onto the older miR‑7 system to refine how secretion and stress responses are tuned in specialized brain and hormone‑secreting cells. They propose that Cdr1as may act as a transporter, carrying miR‑7 into nerve endings where local protein synthesis and transmitter release are controlled.
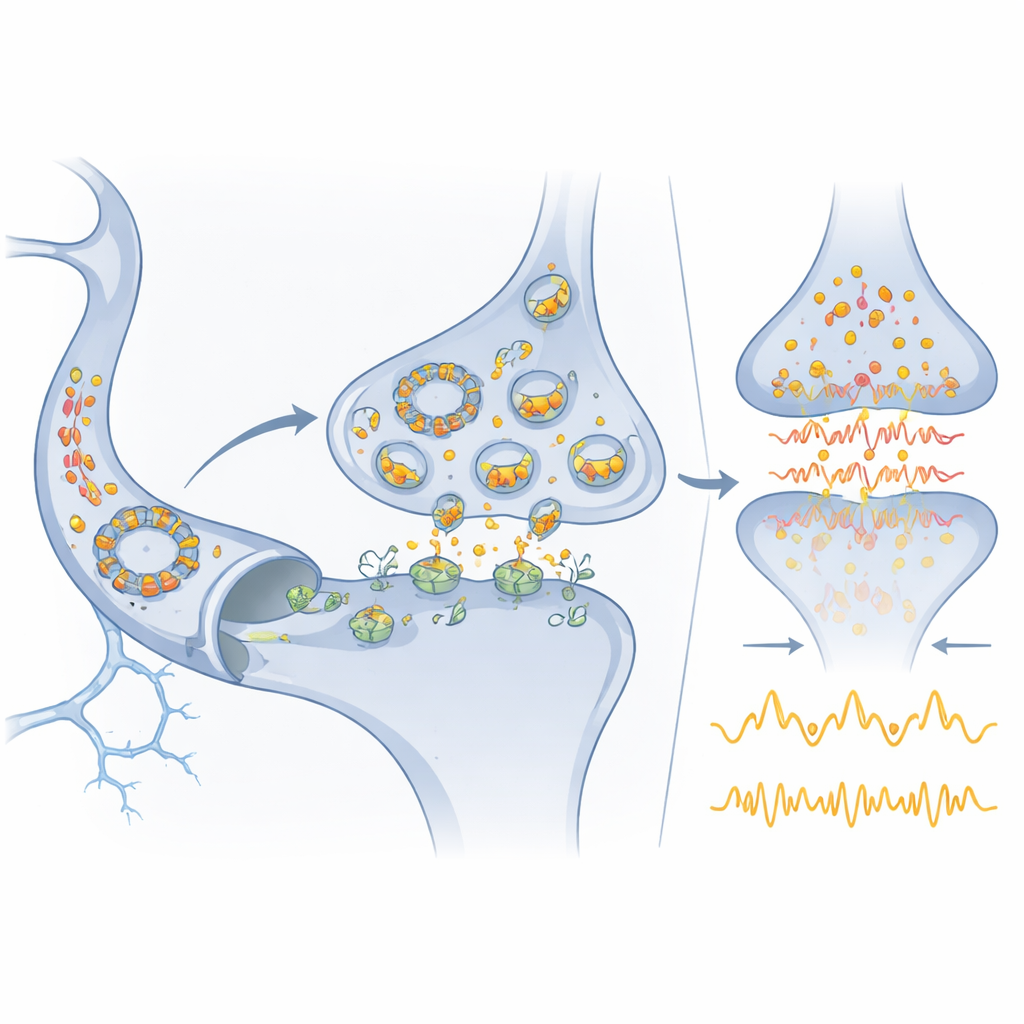
From molecular circuit to medicine
Because Cdr1as is so brain‑enriched yet stable enough to be detected in blood, it could serve as a convenient marker of brain state in neurological and psychiatric diseases. miR‑7, already found in various bodily fluids, may similarly help track or modify disease processes. The authors outline how drugs or RNA‑based therapies targeted to miR‑7, Cdr1as, or miR‑671 might one day dampen damaging glutamate surges in stroke or rebalance misfiring networks in mood and developmental disorders. At the same time, they caution that most proposed circular RNA regulators likely lack the exceptional abundance and binding capacity of Cdr1as. As a result, this system becomes a benchmark: it shows that RNA‑only circuits can rival protein networks in precision and robustness, but also that true regulatory heavy‑hitters among circular RNAs are probably rare and must be identified with rigorous, quantitative tests.
Citation: Cerda-Jara, C.A., Scoyni, F., Zolotarov, G. et al. The history and function of a circular RNA. Nat Commun 17, 3862 (2026). https://doi.org/10.1038/s41467-026-71822-0
Keywords: circular RNA, microRNA-7, neuronal stress, glutamatergic signaling, RNA biomarkers