Clear Sky Science · en
Disruption of the SAGA CORE triggers collateral degradation of KAT2A
When Cell Machinery Falls Apart
Inside every cell, essential jobs like turning genes on and off are carried out by huge molecular machines built from many different protein parts. This study looks at one such machine, called the SAGA complex, which helps switch genes on and is often hijacked in cancer. The researchers discovered that if you damage key structural pieces of SAGA, you don’t just weaken the machine—you trigger the selective destruction of one of its main working parts, a protein called KAT2A, revealing a hidden weakness that drugs could potentially exploit.
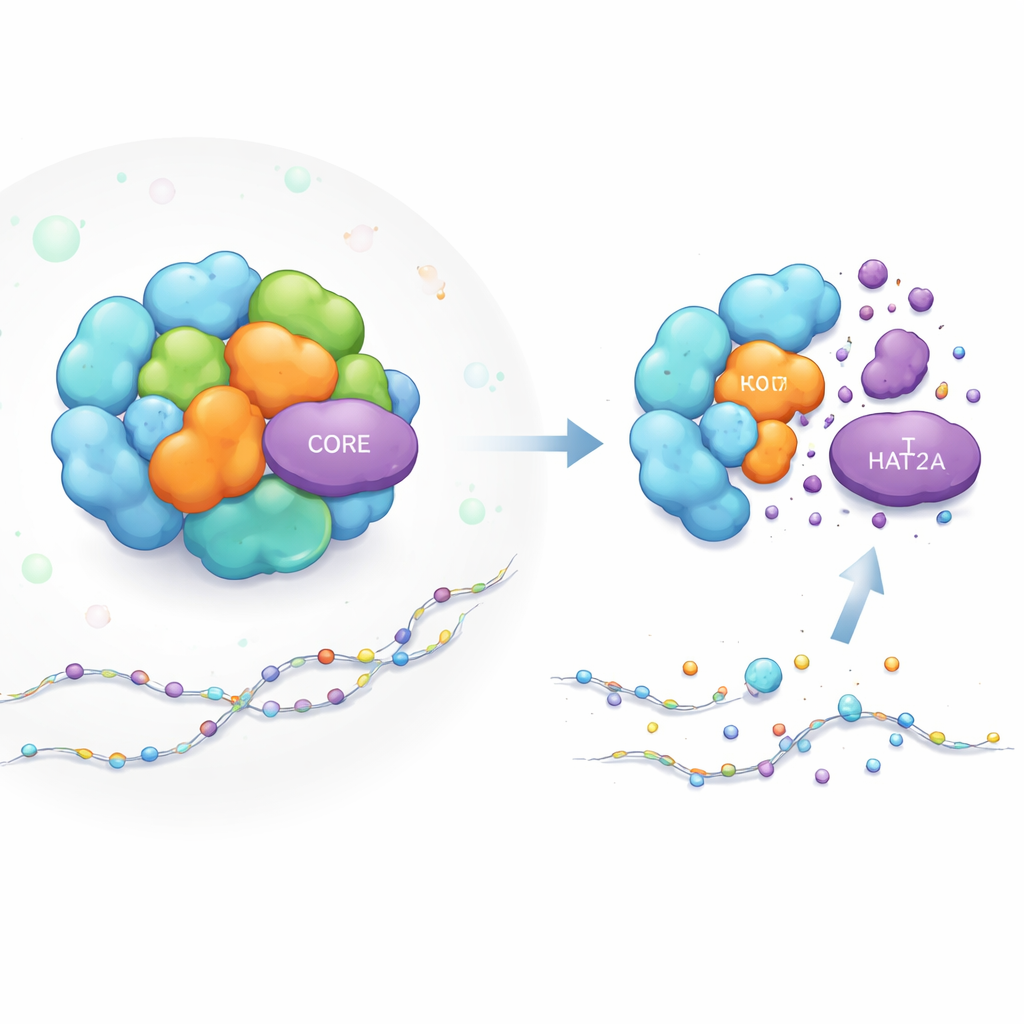
How a Gene-Switching Machine Is Built
The SAGA complex is like a modular workshop for gene activation. Some parts perform chemical tasks, such as adding acetyl “tags” to histone proteins (the spools that package DNA), which makes nearby genes easier to turn on. KAT2A is the acetyl-adding worker in one of SAGA’s key modules. Other SAGA parts act more like scaffolding, giving the whole structure its shape and holding modules in the right place near DNA. While scientists knew a lot about what SAGA does, they did not understand how the stability of individual parts like KAT2A is maintained inside this crowded, dynamic machine.
Testing What Happens When Pieces Are Removed
To probe SAGA’s inner workings, the authors built a fluorescent reporter that glows in proportion to the amount of KAT2A in living cells. Using CRISPR gene editing, they knocked out each SAGA component one by one and watched how KAT2A levels changed. Removing several nearby partners in the same acetylation module reduced KAT2A abundance, as expected. Surprisingly, knocking out a specific trio of structural CORE proteins—TADA1, TAF5L and TAF6L—also caused KAT2A protein to drop sharply, even though these parts do not directly perform any chemical reactions. The effect was seen in multiple human cell types, including leukemia cells, showing it is a general feature rather than a cell-line oddity.
From Broken Structure to Lost Gene Activity
By looking more closely at the molecular fallout, the team showed that losing these CORE proteins does more than simply trim KAT2A levels. The SAGA complex itself falls apart, and the acetylation module disengages from the larger assembly. When they separated cellular components by size, KAT2A shifted from high–molecular weight fractions, representing intact SAGA, into lighter fractions consistent with solitary protein. At the same time, KAT2A largely disappeared from DNA start sites across the genome, and the usual acetyl marks it places on histone H3 (at a position called K9) were substantially reduced. Importantly, disrupting the CORE blocked the usual backup response in which a related enzyme, KAT2B, can compensate, making the loss of acetylation more profound and harder for the cell to repair.
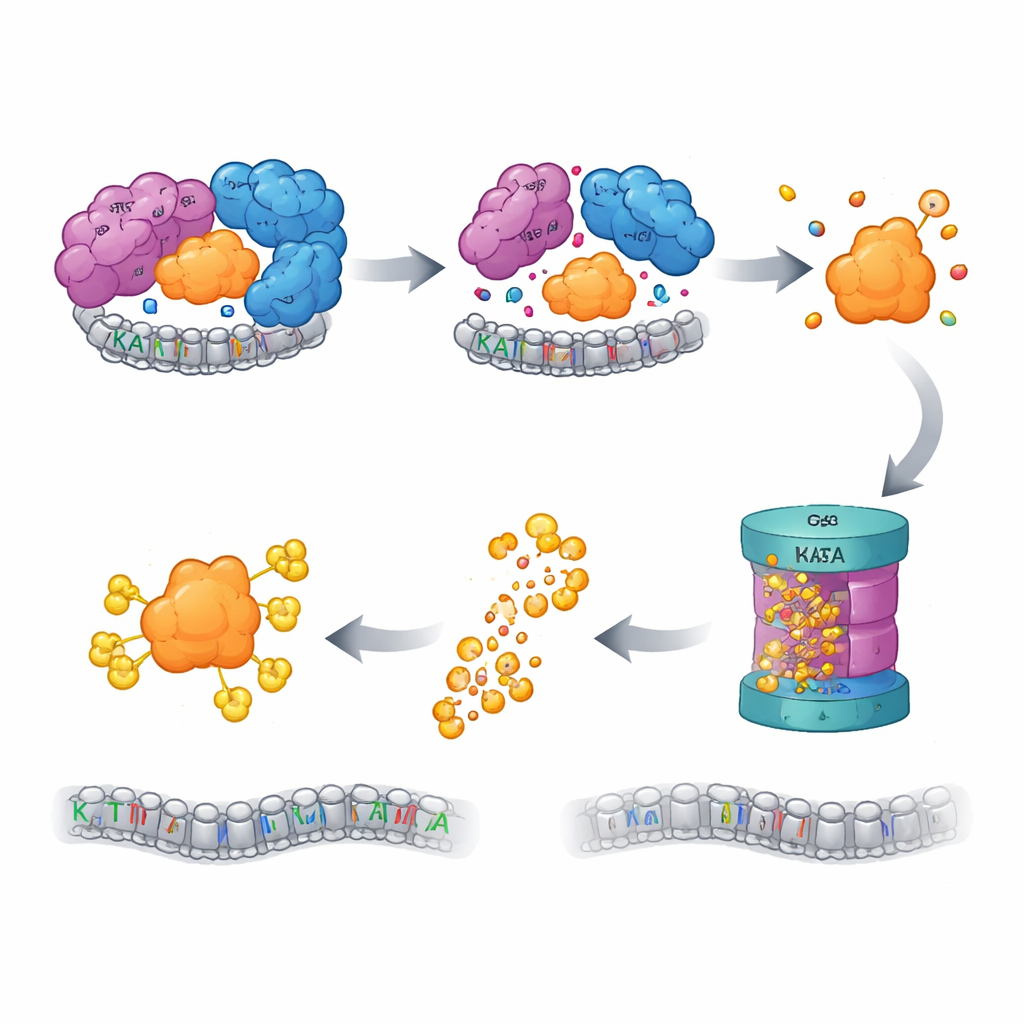
Quality Control Turns on the Shredder
Cells possess quality-control systems that watch for “orphan” proteins—components that have lost their partners and risk causing harm if left to misfold or clump. The authors found that when the SAGA CORE is disrupted, free KAT2A is tagged for destruction by the ubiquitin–proteasome system, the cell’s main protein-disposal machinery. Blocking the proteasome, or earlier steps in this tagging cascade, restored KAT2A levels. A focused CRISPR screen of more than a thousand ubiquitin-related genes pinpointed an E3 ligase called UBR5, together with its helper deubiquitinase OTUD5, as key players in recognizing and degrading orphan KAT2A. Intriguingly, KAT2A’s close cousin KAT2B remained stable under the same conditions, and mutating a short, KAT2A-specific sequence near its beginning was enough to make the protein more resistant to destruction, identifying a minimal, paralogue-specific “degron” that flags KAT2A for disposal.
A Hidden Weakness with Therapeutic Promise
Overall, the work shows that KAT2A’s stability and activity are tightly tied to the intact architecture of the SAGA complex. When structural CORE components are lost, SAGA disassembles, KAT2A is released as an unprotected orphan, and the UBR5–OTUD5 surveillance system swiftly routes it to the cellular shredder, reducing gene-activating acetyl marks across the genome. Because KAT2A is a known vulnerability in several cancers, this built-in linkage between structure, assembly, and selective degradation suggests a new strategy: instead of targeting KAT2A’s active site directly, drugs could destabilize non-enzymatic SAGA scaffold proteins, simultaneously blocking KAT2A’s access to chromatin and promoting its collateral destruction.
Citation: Batty, P., Beneder, H., Schätz, C. et al. Disruption of the SAGA CORE triggers collateral degradation of KAT2A. Nat Commun 17, 3410 (2026). https://doi.org/10.1038/s41467-026-71613-7
Keywords: SAGA complex, KAT2A, protein quality control, ubiquitin proteasome system, cancer epigenetics