Clear Sky Science · en
L-RNA aptamer-based CXCL12 inhibition combined with radiotherapy and bevacizumab in newly-diagnosed glioblastoma: expansion of the phase I/II GLORIA trial
Why this brain cancer study matters
Glioblastoma is one of the deadliest brain cancers, in part because it almost always grows back after treatment. Even when surgeons remove as much tumor as possible and doctors follow with radiation, the tumor’s blood supply rapidly rebuilds itself, allowing cancer cells to recover. This study tested a new strategy: blocking two different routes the tumor uses to regrow its blood vessels, and asking whether this dual shutdown, added to radiotherapy, can safely hold the cancer in check for longer in patients who usually do very poorly.
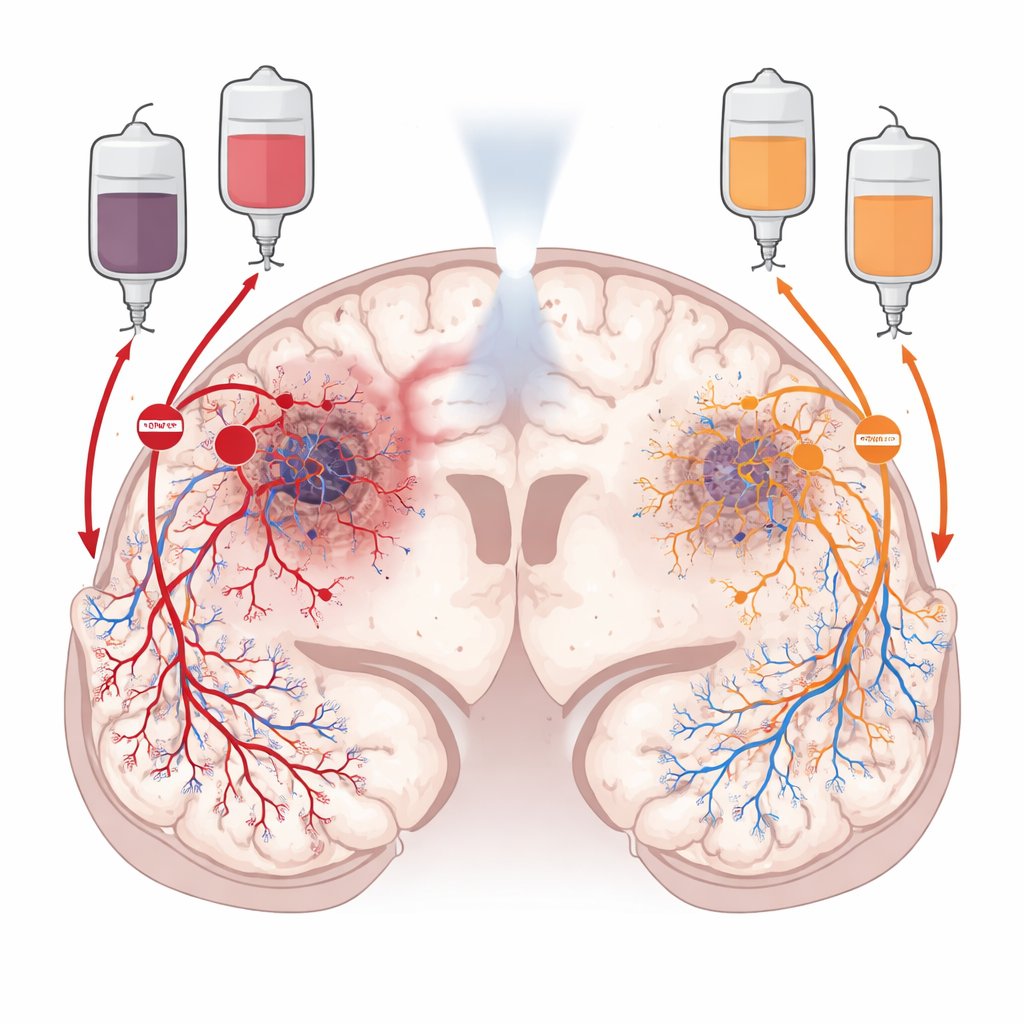
Two hidden routes that feed the tumor
After radiotherapy, glioblastoma does not simply sit still. It actively reshapes its surroundings to restore blood flow. One route, called angiogenesis, relies on a growth signal that makes nearby blood vessels sprout new branches into the tumor. The other, called vasculogenesis, recruits fresh blood-vessel–forming cells from the bone marrow, which home in on the tumor and build new vessels from scratch. The researchers focused on two chemical “traffic controllers” that guide these routes: VEGF, which drives local vessel sprouting, and CXCL12, which attracts circulating progenitor and immune cells and helps them settle in the tumor. Because these signals operate in different regions of the tumor, the team reasoned that blocking both might starve the cancer more effectively than blocking either one alone.
Mapping where the signals act inside human tumors
To see how VEGF and CXCL12 are arranged inside glioblastomas, the scientists mined large public genetic datasets and then turned to high-resolution images of real patient tumors. They found that VEGF closely tracks with low-oxygen, dying regions deep within the tumor, where damaged tissue calls urgently for new blood vessels. CXCL12, in contrast, appears mainly in the more cellular and highly vascular zones and in the outer infiltration areas where tumor cells snake into healthy brain. Using advanced staining techniques and spatial gene profiling, they showed that CXCL12 is produced not just by tumor cells, but also by the lining of blood vessels, support cells, and immune cells in and around the tumor, including normal brain vessels. This reinforced the idea that VEGF and CXCL12 are complementary, non-overlapping drivers of blood-vessel rebuilding.
A trial combining radiation with a CXCL12 trap and anti-VEGF therapy
Building on earlier work with a CXCL12-blocking drug called NOX-A12, the GLORIA trial enrolled adults with newly diagnosed glioblastoma whose tumors could not be fully removed and who lacked a DNA marker that makes standard chemotherapy effective. All patients received radiotherapy and continuous intravenous NOX-A12. In an expansion phase, a subset of six patients also received bevacizumab, an antibody that targets VEGF and is already used in recurrent glioblastoma. The main goal was to test safety; the team also tracked how long tumors stayed controlled, overall survival, changes in tumor blood flow on MRI, and patients’ neurological function and quality of life.
Safety and signs of stronger, longer responses
The triple combination of radiotherapy, NOX-A12, and bevacizumab proved well tolerated. There were no treatment-related deaths and no dose-limiting toxic side effects. Most adverse events were mild to moderate and largely linked to the tumor or to radiation itself; blood pressure elevations from bevacizumab were manageable with medication. Imaging showed that adding bevacizumab led to much deeper drops in tumor blood volume and perfusion, suggesting that vessel regrowth was strongly suppressed. Clinically, patients on the triple therapy had a median period without tumor progression of just over nine months and a median overall survival of nearly 20 months—both clearly longer than patients who received radiotherapy with NOX-A12 alone, and better than carefully matched groups treated with standard care. Remarkably, two of the six patients on triple therapy lived more than two years, an unusually long time for this high-risk population.
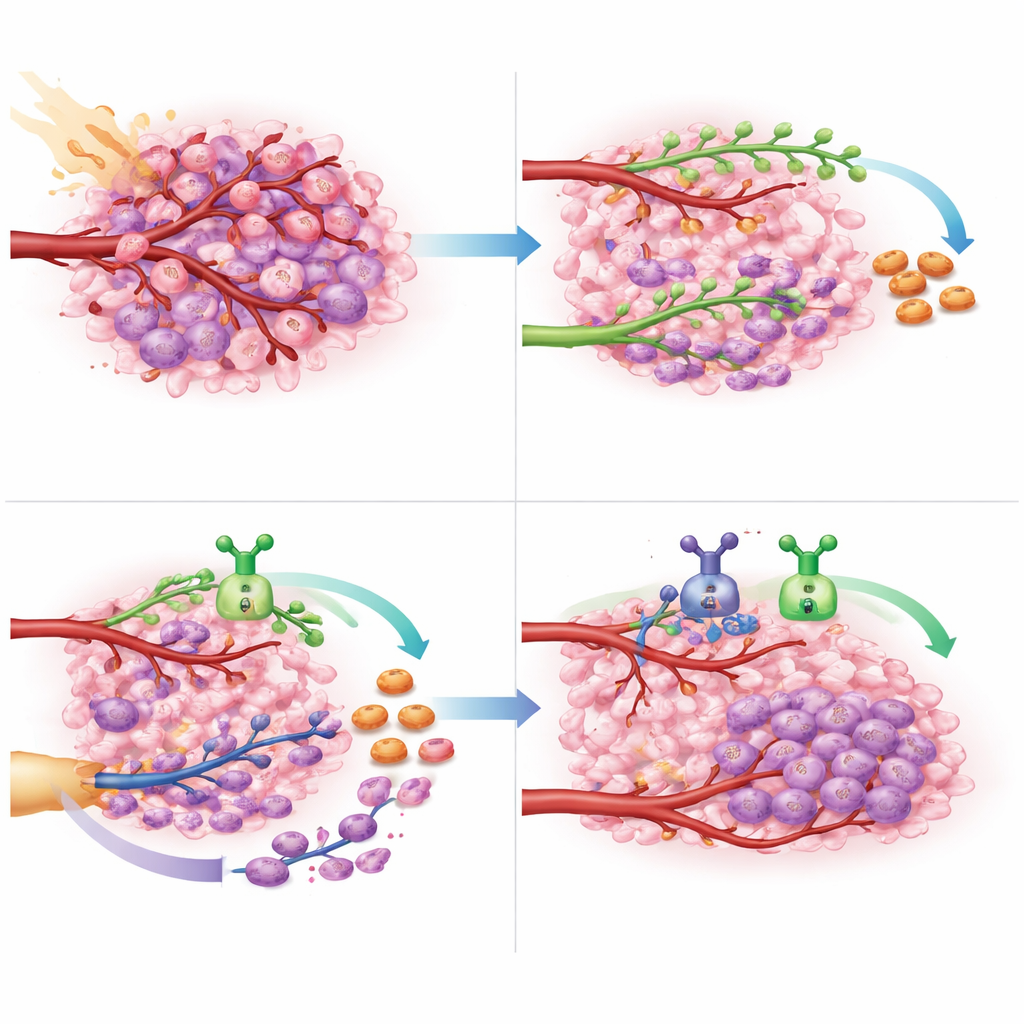
What this could mean for future brain cancer care
For patients with glioblastoma that cannot be fully removed and resists standard chemotherapy, options are scarce and outcomes are grim. This study offers proof-of-principle that shutting down both major blood-vessel repair routes—VEGF-driven sprouting and CXCL12-driven recruitment of new vessel-forming cells—during and after radiotherapy can be done safely and may substantially delay tumor regrowth. While the trial was small and designed mainly to assess safety, the survival gains are encouraging enough to warrant larger, randomized studies. If confirmed, dual targeting of these two vascular signals could become a new first-line strategy to keep this aggressive brain cancer at bay for longer.
Citation: Giordano, F.A., Layer, J.P., Turiello, R. et al. L-RNA aptamer-based CXCL12 inhibition combined with radiotherapy and bevacizumab in newly-diagnosed glioblastoma: expansion of the phase I/II GLORIA trial. Nat Commun 17, 3405 (2026). https://doi.org/10.1038/s41467-026-71362-7
Keywords: glioblastoma, brain cancer, tumor blood vessels, radiotherapy, targeted therapy