Clear Sky Science · en
Disentangling the Janus-faced effects of cations in electrocatalysis
Two-Sided Helpers in Clean Energy
Producing clean hydrogen fuel from water often relies on reactions at charged metal surfaces bathed in salty solutions. The dissolved positively charged atoms, or cations, are usually treated as passive background. This paper shows they are anything but: the same cations that sometimes speed up hydrogen production can, under slightly different conditions, slow it down. Understanding this double nature is vital for designing better electrolyzers and other electrochemical technologies for a low‑carbon future.
Why Salt Ions Matter at Electrodes
When an electric voltage is applied to a metal electrode in an alkaline solution, water molecules near the surface split to form hydrogen gas. Experiments have long shown that both the type of dissolved cation (such as lithium, sodium, or potassium) and its concentration can dramatically change how fast hydrogen is produced. Even more puzzling, the trends can flip: on gold surfaces, one ordering of cations appears at gentle voltages but reverses at stronger voltages; changing the solution pH or cation concentration can turn an apparent promoter into an inhibitor. Existing explanations focused on how strongly hydrogen sticks to the metal, or on how easily charged species move away, but these ideas could not consistently explain all the observed reversals.
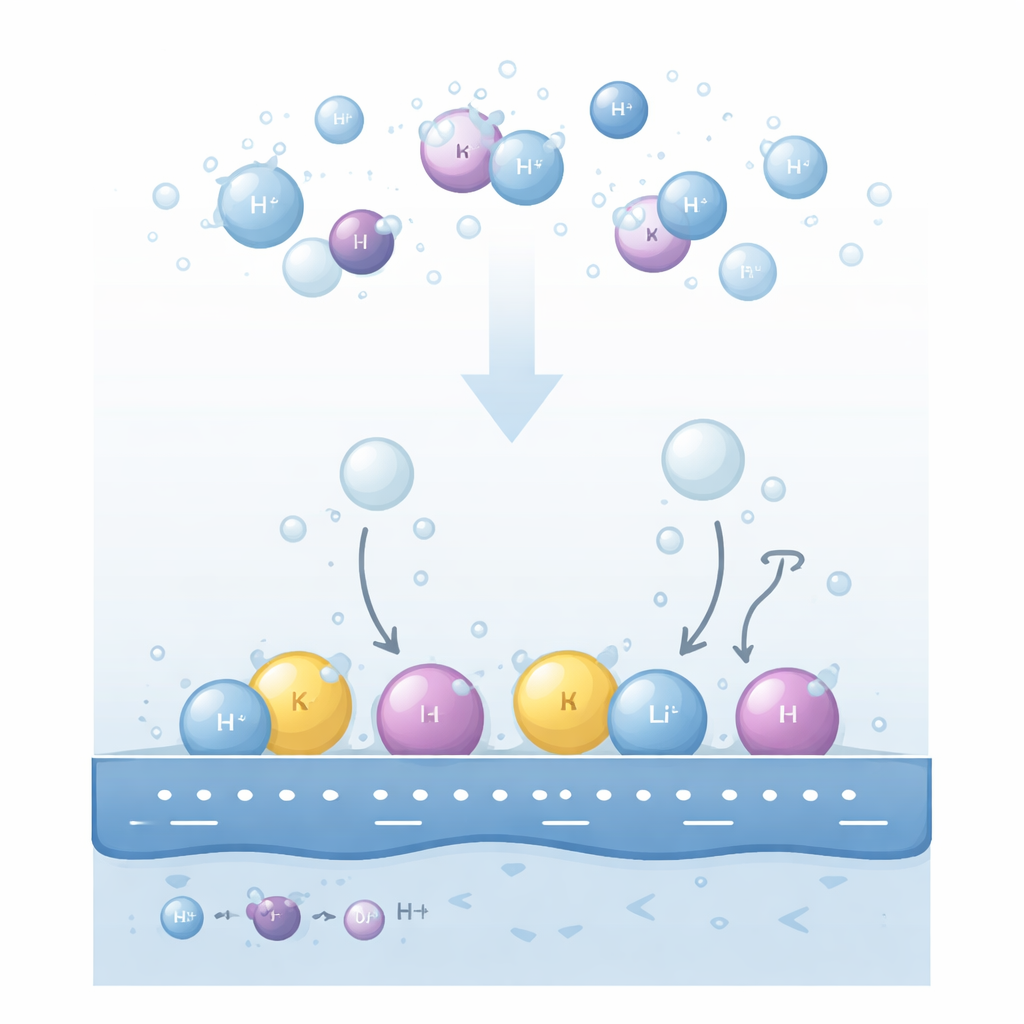
Two Neighborhoods for Cations
The authors propose that the key lies in where cations actually reside in the thin liquid layer next to the electrode. They distinguish two main neighborhoods. Some cations stay fully wrapped in water molecules and hover a bit away from the surface in a “diffuse” region. Others, driven by a strong negative charge on the metal, partially shed their water shell and snuggle right up to the first layer of interfacial water, forming a compact “specifically adsorbed” layer. These two cation populations rearrange the local electric field in opposite ways. Diffuse cations strengthen the field felt by nearby water molecules, while adsorbed cations partially screen the metal’s charge and weaken that field.
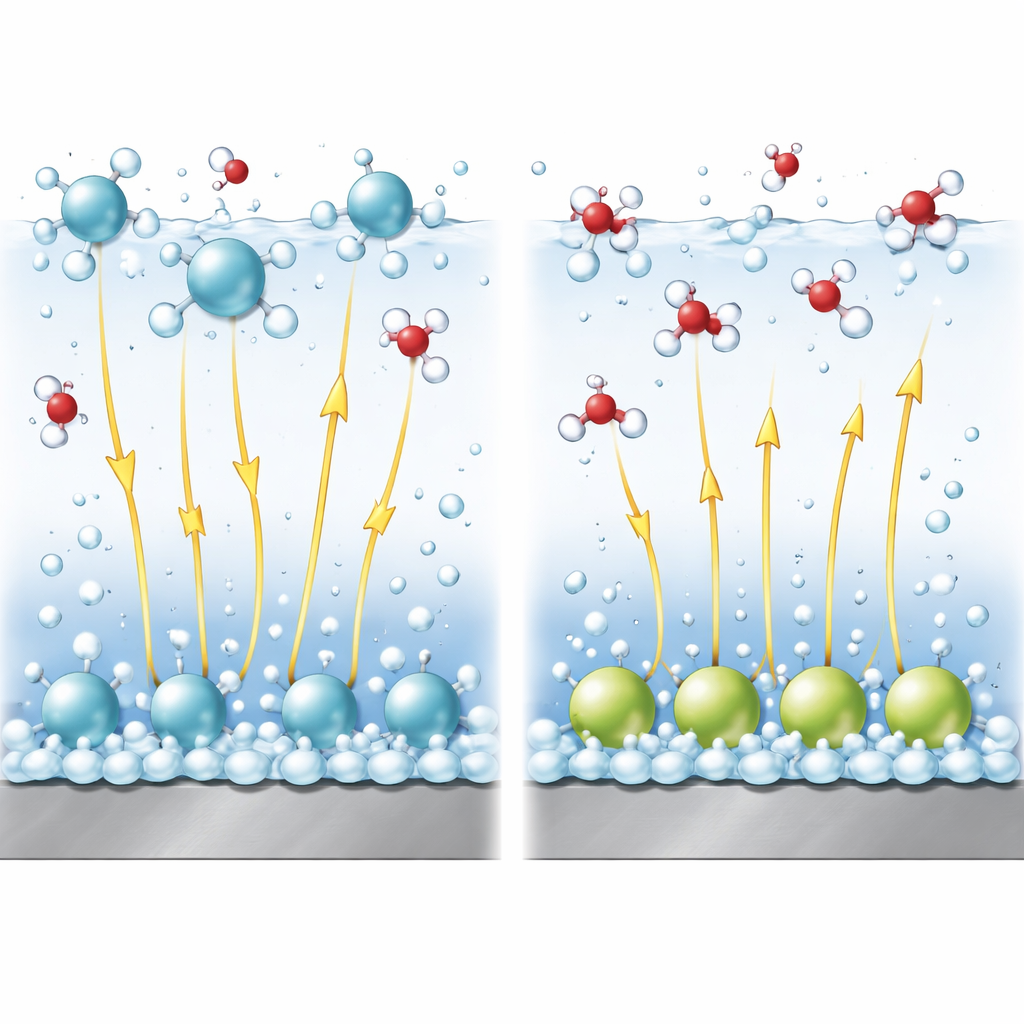
Electric Fields as Invisible Levers
In alkaline solutions, splitting water to make hydrogen is harder than reducing simple protons. A crucial part of the reaction is breaking the bond within a water molecule. Strong electric fields near the surface help stretch and break this bond, much like pulling on opposite ends of a spring. Using a refined theoretical model, the authors couple the arrangement of cations to the strength of this field and to the energy barrier for bond breaking. They show that cations held in the diffuse layer tend to raise the local potential in a way that favors the reaction and keep the electric field strong, thus promoting hydrogen formation. In contrast, specifically adsorbed cations reduce the effective field in the water layer where the reaction takes place, making bond breaking more difficult and slowing the reaction.
Explaining Confusing Experimental Trends
By allowing the balance between diffuse and adsorbed cations to shift with voltage, pH, cation size, and cation–surface affinity, the model reproduces a wide range of puzzling data. For example, potassium ions are more weakly hydrated and more prone to adsorb than lithium ions. At modest voltages, size and positioning effects make potassium seem more beneficial. At stronger voltages, however, dense adsorption of potassium screens the electric field so effectively that lithium becomes the better “helper,” reversing the activity order. Similarly, when the concentration of lithium salts is raised, diffuse cations dominate and the reaction speeds up, but increasing sodium or potassium concentration favors adsorbed cations and slows the reaction. Adjusting how strongly cations bind in the model also mimics differences between electrode materials and solution pH, matching experimental behavior on gold and platinum across various alkaline conditions.
What This Means for Future Devices
In simple terms, the study reveals that dissolved cations play a double role in hydrogen production: they can either pull on water molecules to help them break apart, or crowd the surface and muffle the very electric field that drives the reaction. Which face they show depends on their size, how tightly they hold on to water, how strongly they stick to the electrode, and how concentrated they are. This unified picture helps explain decades of confusing observations and suggests new design rules for electrolyzers and related technologies. Rather than choosing a single “best” cation, engineers may tune mixtures and conditions so that most cations sit at just the right distance from the surface—strong enough to boost the electric field, but not so attached that they smother it.
Citation: Zhu, X., Binninger, T., Koper, M.T.M. et al. Disentangling the Janus-faced effects of cations in electrocatalysis. Nat Commun 17, 3149 (2026). https://doi.org/10.1038/s41467-026-71126-3
Keywords: hydrogen evolution reaction, electric double layer, alkali metal cations, water electrolysis, electrocatalysis