Clear Sky Science · en
Auxiliary subunits reshape structural asymmetry and functional plasticity in heterotetrameric GluA1/A2 AMPA receptor core
How brain signal gates shape learning and disease
Every thought, memory, or movement you make depends on tiny protein machines that let charged atoms flow between nerve cells. This study zooms in on one of the brain’s most important of these machines—the GluA1/A2 AMPA receptor—and reveals how small helper proteins subtly change its shape and behavior. Understanding these changes may open doors to safer, more precise drugs for conditions such as epilepsy, stroke, and Alzheimer’s disease.
The brain’s fast switches
In the brain, many excitatory signals are carried by AMPA receptors, which sit in the receiving membrane of synapses. When the chemical messenger glutamate arrives, these receptors open a pore that briefly lets positively charged ions rush into the cell, producing electrical signals in just milliseconds. The exact makeup of each receptor, and which helper proteins cling to its sides, tunes how quickly it opens and closes, how much current passes, and how it recovers after heavy use. One particular combination, called the GluA1/A2 heterotetramer, is the dominant form in key learning centers like the hippocampus, yet its detailed structure without any helpers attached had remained elusive.
Capturing the receptor in three key poses
Using high-resolution cryo–electron microscopy, the authors purified GluA1/A2 receptors and trapped them in three essential functional poses: closed and ready to respond, open and conducting ions, and desensitized—a protective, temporarily unresponsive state after prolonged stimulation. They verified with electrical recordings that their slightly engineered receptors behaved much like natural ones. The images revealed a Y‑shaped architecture built from four subunits arranged as pairs, and, crucially, clearly resolved the short loop that forms part of the ion selectivity filter, a region that had been blurred in many earlier structures. These baseline views show how glutamate-induced motions in the ligand-binding “clamshells” tug on linkers and pry open the channel gate in this physiologically central receptor.
Helper proteins widen the gate
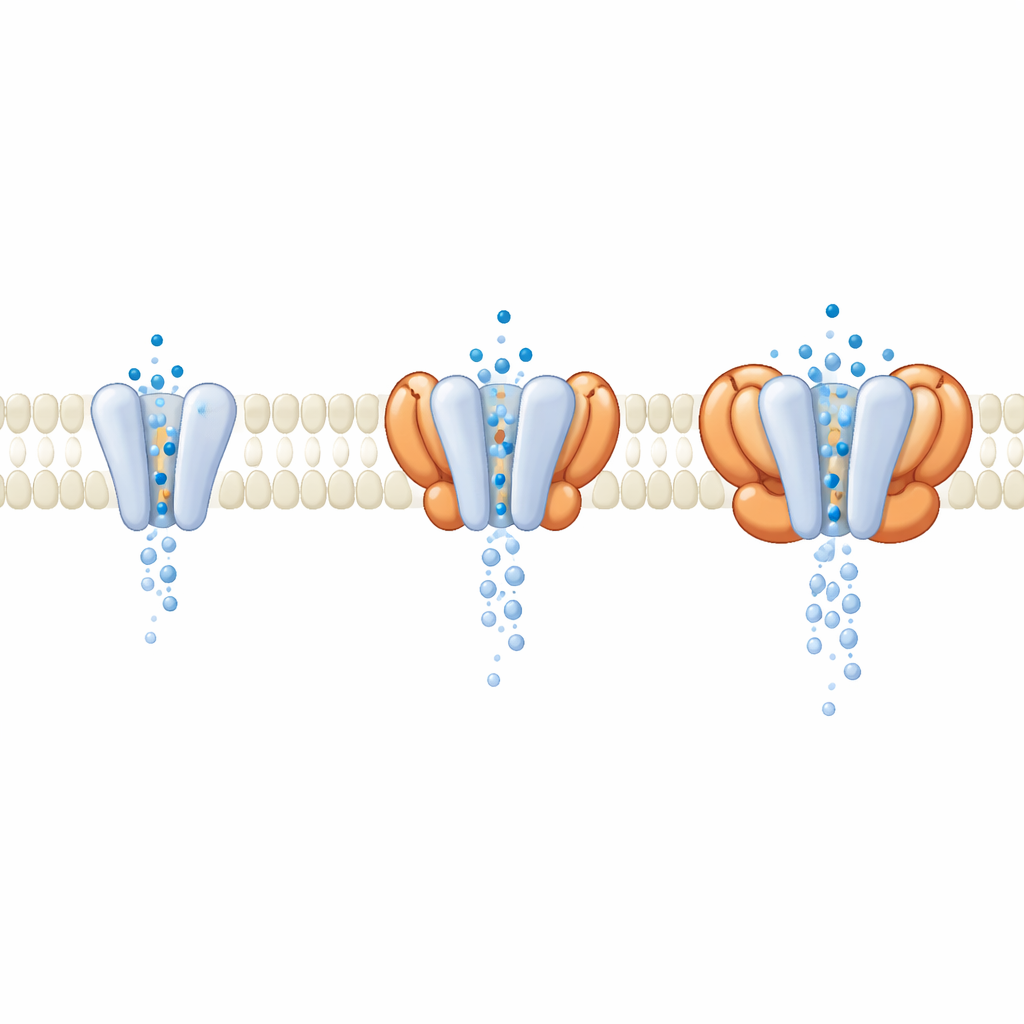
In cells, GluA1/A2 rarely acts alone; it is usually accompanied by auxiliary proteins that ride in the membrane beside it. During data processing, the researchers discovered a subset of receptors naturally bound to four cornichon (CNIH) helper units carried over from the expression cells. Comparing these helper-loaded receptors to the bare core, they found that the outer, signal-detecting parts looked almost identical when the channel was open. The key differences appeared deeper in the membrane, where CNIH and related helper families altered the geometry of the pore itself. Molecular dynamics simulations of water flow through the channel showed that complexes containing certain helpers produced a wider, better hydrated pore and higher simulated conductance, even though the glutamate-sensing domains barely changed. This suggests that many modulators act not by changing how receptors sense glutamate, but by reshaping the gate through which ions pass.
Asymmetry and the art of recovery
The team also examined what happens when the receptor becomes desensitized—still bound to glutamate but no longer conducting. In this pose, the clamshells rearrange so that the interface that holds them together in the active state cracks open, while their lower halves stay close enough to keep the pore shut. By comparing GluA1/A2 with a variety of previously solved AMPA receptor complexes containing different helper proteins, the authors quantified how much each pair of clamshells twists out of a neat two-fold symmetry. They found a strong correlation: the greater this twist, the slower the receptor recovers from desensitization and becomes ready to respond again. Some helpers keep the dimer nearly symmetric, allowing rapid recovery, while others promote distortion and thus prolonged unresponsiveness.
Implications for tuning brain circuits and therapies
Together, these structural and simulation results show that both the identity of the core subunits and the particular set of helper proteins work together to define how AMPA receptors gate electrical signals. Helper units can widen or narrow the pore and control how long receptors stay silent after heavy use, without necessarily altering how glutamate is sensed. Because AMPA receptor malfunction is linked to seizure disorders, stroke damage, and neurodegeneration, the detailed maps of the receptor–helper interfaces and the moving parts of the gate highlight promising new drug targets. Rather than broadly blocking these crucial receptors, future medicines might fine-tune specific assemblies, subtly adjusting ion flow and recovery in exactly the circuits that need it.
Citation: Yen, L.Y., Newton, T.P., Yelshanskaya, M.V. et al. Auxiliary subunits reshape structural asymmetry and functional plasticity in heterotetrameric GluA1/A2 AMPA receptor core. Nat Commun 17, 4191 (2026). https://doi.org/10.1038/s41467-026-71063-1
Keywords: AMPA receptor, synaptic plasticity, auxiliary subunits, cryo electron microscopy, ion channel gating