Clear Sky Science · en
Structural mechanism of substrate binding and inhibition of human taurine transporter
Why this tiny nutrient pump matters
Taurine is a small molecule found throughout the human body, where it helps keep the heart, brain, eyes, and muscles working properly. Our cells cannot rely on passive diffusion to get enough taurine; instead they use a specialized gate in the cell membrane called the taurine transporter. When this transporter goes wrong, it is linked to heart disease, nerve problems, vision loss, and cancer. This study reveals, in atomic detail, how the human taurine transporter grabs taurine, moves it into cells, and can be blocked or hijacked by druglike molecules, offering clues for future therapies.
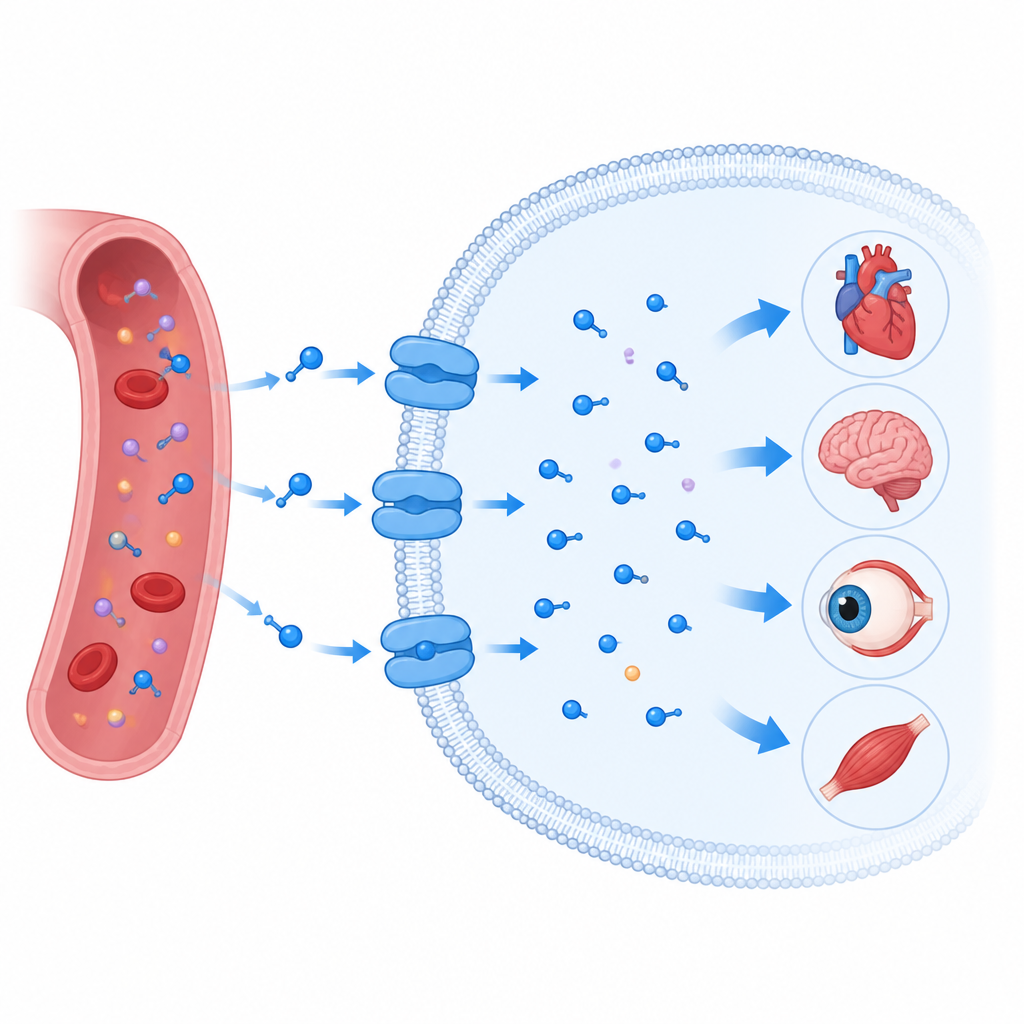
A gate that pulls taurine into cells
The taurine transporter sits in the oily membrane that surrounds every cell and belongs to a large family of transport proteins that also move brain messengers such as serotonin and dopamine. Like its relatives, it does not simply form an open channel. Instead, it works more like a revolving door: it opens to one side of the membrane, closes around its cargo, then opens to the other side. The researchers used cryo electron microscopy to determine multiple three dimensional snapshots of the human taurine transporter in action. They captured it empty, bound to taurine, bound to a related molecule called beta alanine, and bound to a ring shaped taurine mimic known as P4S, in both partially closed and more open inward facing forms.
How taurine fits into its custom pocket
The structures show that taurine docks in a central pocket deep inside the transporter, surrounded by several membrane spanning helices. Taurine has a negatively charged sulfonate group at one end and a positively charged amino group at the other, linked by a short carbon chain. The transporter has a small, glycine rich niche that cradles the bulky sulfonate group and anchors it through a web of hydrogen bonds and a coordinated sodium ion. The carbon chain nestles into a greasy patch, while the amino end reaches a negatively charged residue that forms a stabilizing salt bridge. This finely tuned arrangement explains why the transporter strongly prefers taurine over other similar molecules and how small changes in key residues can weaken transport.
Impostor molecules and how they block or ride the pump
Beta alanine, a natural compound with a carboxyl group instead of taurine's sulfonate, binds in almost the same way, using the same pocket, sodium ions, and contact points. However, it makes fewer hydrogen bonds, which matches its slightly weaker affinity for the transporter. P4S, a synthetic ring shaped taurine analog, also pushes its sulfonate group into the same niche and occupies the taurine site, but its rigid ring does not align its amino group properly with the key acidic residue. This misfit disrupts one of the strongest interactions and makes P4S a lower affinity blocker. Because all three molecules compete for the same site, taurine uptake can be slowed when beta alanine or P4S is present at high levels.
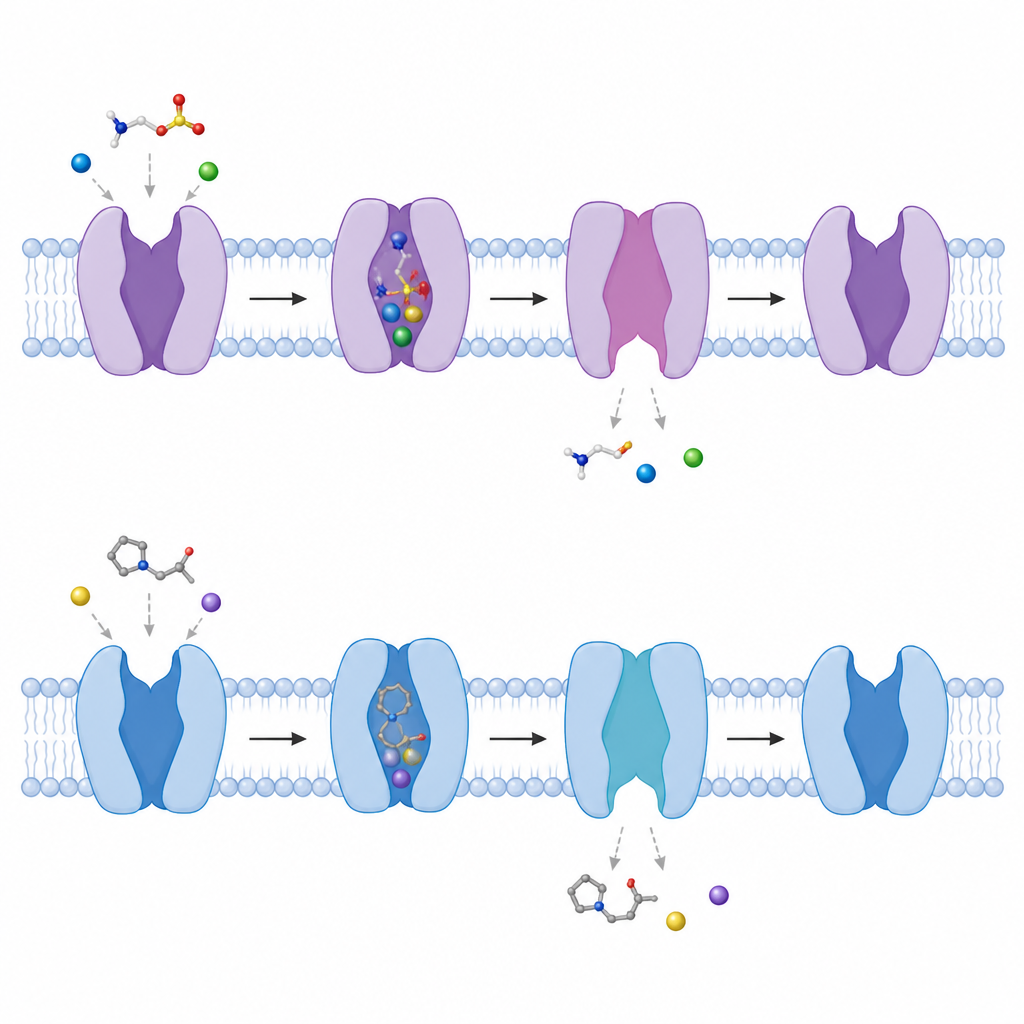
Watching the gate swing open inside the cell
By comparing structures with and without bound molecules, the authors tracked how the transporter toggles between conformations. A short helical segment near the inner side of the membrane bends outward by about 50 degrees, and another helix partly unwinds, creating a tunnel from the central pocket to the inside of the cell. As this happens, the carefully arranged sodium binding sites fall apart, making it easier for sodium and then taurine to be released into the cell interior. The team also showed that in the absence of any bound molecules the transporter naturally flips between more closed and more open inward facing shapes, hinting at a constantly shifting landscape that allows it to respond quickly when taurine appears.
Redefining an inhibitor and looking toward drugs
Surprisingly, functional tests revealed that P4S is not just a simple stopper that jams the gate. Instead, it can be moved through the transporter like taurine itself, driven by the same sodium gradient, and even trigger the release of preloaded taurine from cells. In other words, P4S behaves as a transported substrate analog that happens to compete with taurine. Together, the structural snapshots and transport measurements outline a full working cycle for the taurine transporter and pinpoint the specific pockets and movements that control each step. For non specialists, this means researchers now have a detailed blueprint for designing new molecules that either supply taurine more effectively or selectively slow its uptake in diseases such as cancer, where cells rely on excessive taurine transport.
Citation: Qi, Y., Zhang, Y., Wang, D. et al. Structural mechanism of substrate binding and inhibition of human taurine transporter. Nat Commun 17, 4257 (2026). https://doi.org/10.1038/s41467-026-70772-x
Keywords: taurine transporter, membrane transport, cryo electron microscopy, neurocardiac health, drug design