Clear Sky Science · en
Endocrine therapy reprogramming of breast cancer facilitates metastatic escape via upregulation of P-Rex1/Rac1 signalling
Why this research matters for patients
Many breast cancers are fueled by the hormone estrogen and are treated with drugs that block this signal for years after surgery. Even so, a large fraction of these cancers can quietly return long after treatment seems finished. This study uncovers how hormone-blocking therapy can push some cancer cells into a slow-growing but travel-ready state, and shows that blocking a specific cell movement pathway may help stop these late, deadly returns. 
Slow but dangerous cancer cells
Doctors have long seen that some estrogen receptor positive breast cancers come back a decade or more after initial treatment. These late recurrences often grow slowly but are hard to cure once they have spread. By examining tissue from patients, the researchers found that metastases from estrogen driven tumors frequently contained fewer actively dividing cells than the original breast tumors. This suggested that slow-growing cells, not just fast-dividing ones, could be key drivers of late spread.
Building a model of quiet escape
To study these quiet escape artists, the team treated estrogen sensitive breast cancer cells with standard hormone-blocking drugs in the lab. Over time, two kinds of survivors emerged: fast-growing resistant cells and a second group that divided slowly yet refused to die. When the slower cells were implanted into mice, they formed smaller primary breast tumors with features resembling aged or stressed cells, including large, distorted nuclei and dense collagen around them. Despite this sluggish growth at the original site, these cells spread to the lungs and bones as efficiently as the fast-growing cells, revealing that growth speed and spread are not the same thing. 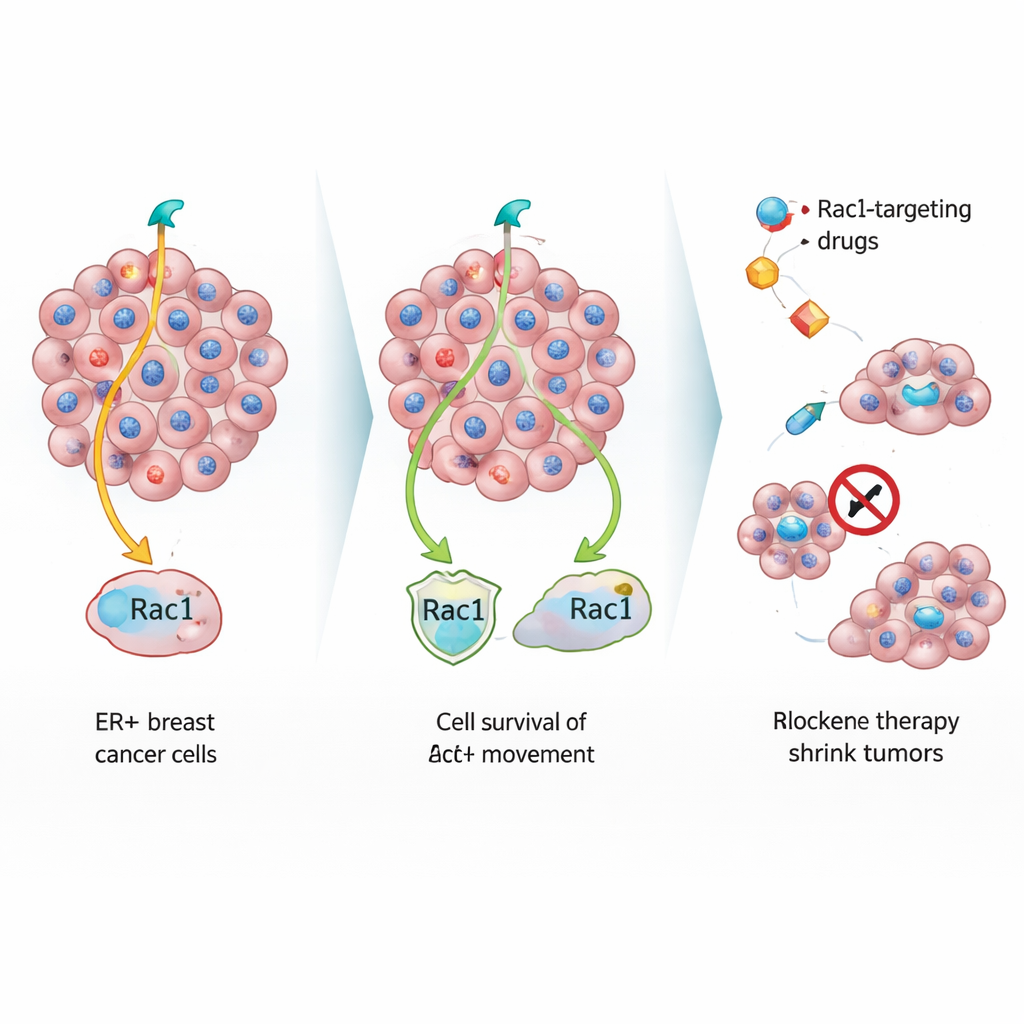
A movement switch flipped by treatment
The team then used single cell RNA sequencing, a technique that reads the activity of thousands of genes in individual cells, to see how hormone therapy was rewiring these survivors. They found that endocrine treatment pushed a subset of cells into a senescent-like state, where division slows but survival pathways stay active. In these cells, a signaling route centered on proteins called P-Rex1 and Rac1 was strongly turned up. Rac1 helps control cell shape and movement, and P-Rex1 is one of its key activators. The slow resistant cells depended on this axis for their ability to migrate, and patient samples showed that P-Rex1 levels were high in estrogen receptor positive tumors and their metastases, especially in cases that recurred late.
Testing new drug combinations
Because the P-Rex1/Rac1 route looked like a movement and survival engine for these tolerant cells, the researchers tested drugs that dampen Rac1 activity. In dishes, these inhibitors reduced colony formation and slowed wound-like closure by the slow resistant cells more than by the fast ones. In mouse models with built in fluorescent sensors for Rac1 activity, two Rac1-targeting compounds visibly lowered Rac1 signaling inside tamoxifen treated tumors. In a patient derived tumor model taken from a woman whose cancer had already resisted multiple hormone therapies, Rac1 inhibitors alone modestly slowed tumor growth and lengthened survival. When combined with tamoxifen, however, they caused tumors to shrink and significantly reduced final tumor weight.
Clues from real world painkillers
One of the Rac1-blocking agents, R-ketorolac, is the mirror image of a drug already used for pain relief after surgery. The team performed a meta-analysis of previous clinical studies that tracked breast cancer outcomes in patients who received ketorolac around the time of surgery. Across these studies, use of ketorolac was linked to a lower risk of cancer recurrence, while other painkillers without Rac1 targeting activity did not show this pattern. Although not proof on its own, this population level hint supports the idea that temporarily blocking Rac1 during key treatment windows may reduce the chance that tolerant cells seed future metastases.
What this means for the future
To a non specialist, the central message is that hormone therapy can unintentionally reshape some breast cancer cells into slow dividing but highly capable travelers. This work pinpoints the P-Rex1/Rac1 pathway as a control knob for that behavior and shows in animals that turning this knob down, especially in combination with standard hormone drugs, can shrink resistant tumors. The findings do not yet translate into a new standard treatment, but they suggest that adding safe Rac1 targeting strategies, possibly even adapted from existing pain medicines, could one day help prevent late, life threatening returns of estrogen driven breast cancer.
Citation: Fernandez, K.J., Sultani, G., Nobis, M. et al. Endocrine therapy reprogramming of breast cancer facilitates metastatic escape via upregulation of P-Rex1/Rac1 signalling. Nat Commun 17, 3042 (2026). https://doi.org/10.1038/s41467-026-70683-x
Keywords: estrogen receptor positive breast cancer, endocrine therapy resistance, metastasis, Rac1 signalling, P-Rex1