Clear Sky Science · en
INSIG1/2 succination mediated by the moonlighting function of ADSL promotes lipogenesis and liver tumorigenesis
How Sugar Feeds Liver Tumors
Why do cancer cells crave sugar, and how do they turn it into the raw materials needed to grow? This study uncovers a hidden wiring inside liver cancer cells that links high blood sugar directly to fat production and tumor growth. By tracing a chain of events from glucose outside the cell to fat-making switches deep inside, the researchers also reveal how an existing HIV drug could be repurposed to slow liver cancer.
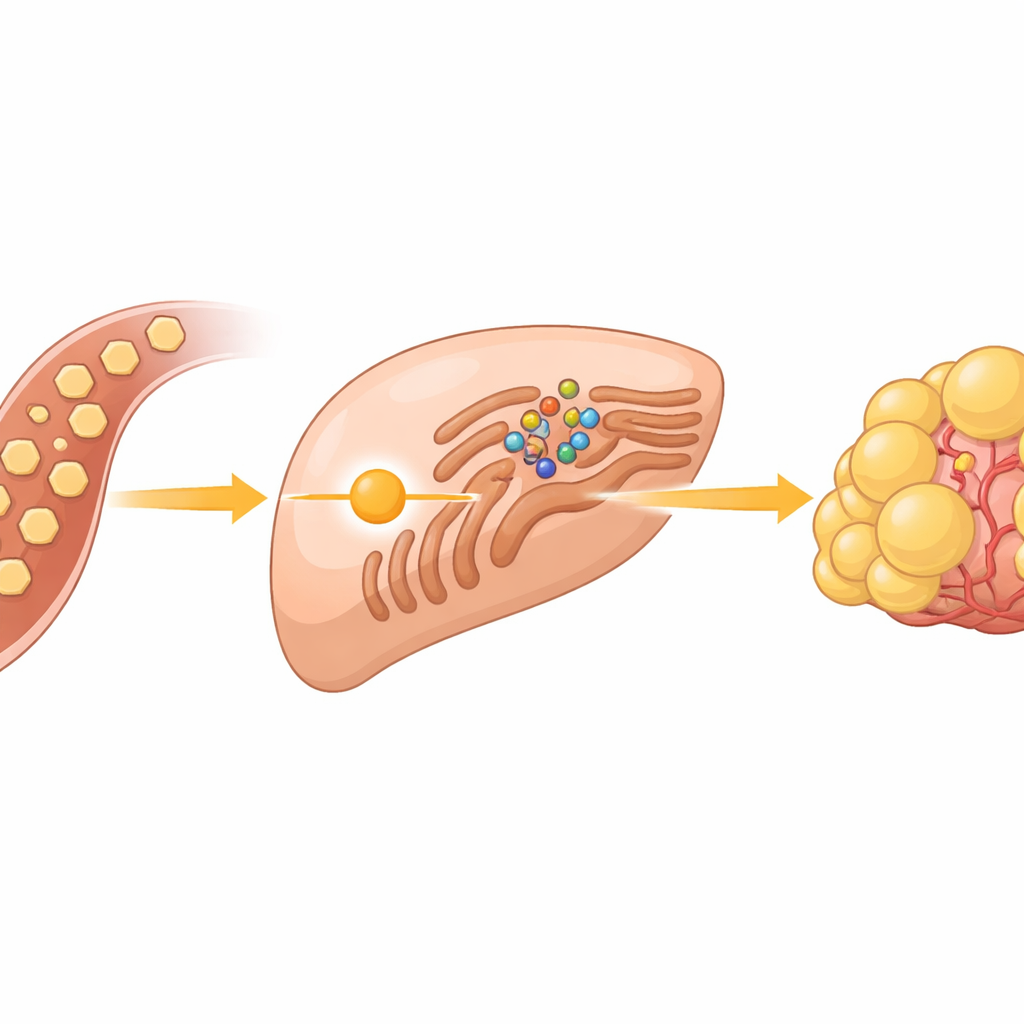
A Hidden Sugar Sensor Inside Cancer Cells
Cancer cells are famous for burning sugar rapidly, even when oxygen is plentiful, a behavior known as the Warburg effect. In liver tumors, this sugar rush does more than just provide energy; it also feeds the creation of fats that build new cell membranes. The team focused on a group of proteins called SREBPs, master switches that turn on genes for making fats and cholesterol. Under normal conditions, these switches are held in check at a cellular structure called the endoplasmic reticulum, where they are locked in place by a sensor protein named INSIG and its partner SCAP. The big mystery has been how extra glucose tells this system to release SREBPs and start fat production.
An Enzyme With a Second Job
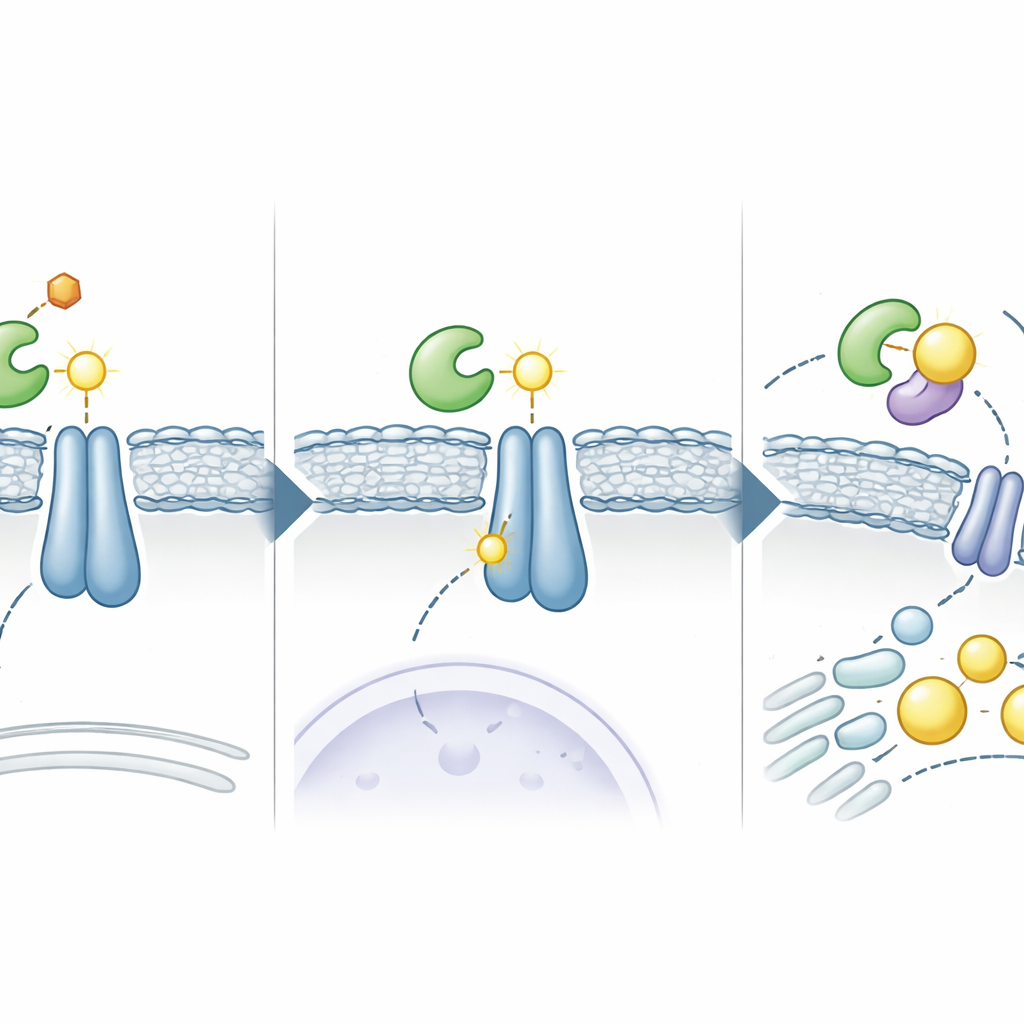
The researchers discovered that an enzyme called ADSL, best known for helping cells build DNA building blocks, has a surprising second job in liver cancer. When glucose levels are high, it activates a signaling enzyme called PKCε. PKCε then adds a phosphate tag to ADSL at a specific position, prompting ADSL to move from the watery interior of the cell to the surface of the endoplasmic reticulum. There, ADSL physically attaches to INSIG proteins. This relocation turns ADSL from a simple metabolic workhorse into a local chemical factory that can directly influence the fat-making switches.
Chemical Scarring That Unlocks Fat Production
Once ADSL is parked next to INSIG, it generates a small molecule called fumarate as part of its usual chemistry. In this new setting, fumarate reacts with a particular amino acid on INSIG, forming a permanent chemical “scar” known as succination. This scar forms at a key cysteine site (C167 in INSIG1 and C111 in INSIG2) that normally helps INSIG bind oxysterols, cholesterol-like messengers that keep the SCAP–SREBP complex anchored in place. Succination weakens INSIG’s grip on these messengers and on SCAP, allowing the SCAP–SREBP complex to leave the endoplasmic reticulum and travel to the Golgi compartment. There, SREBP is cut and its active fragment moves into the nucleus, turning on a suite of genes that drive fat and cholesterol production, fueling rapid tumor cell growth.
From Cell Circuits to Drug Repurposing
To see if this pathway could be turned against the cancer, the scientists used computer-based screening of approved drugs to find molecules that might wedge into the interface between ADSL and INSIG. They identified Elsulfavirine, an HIV medication, as a strong candidate. In laboratory experiments and computer simulations, Elsulfavirine disrupted the binding between ADSL and INSIG, reduced INSIG succination, and blocked SREBP activation in liver cancer cells. As a result, the cells made fewer fats, stored fewer lipid droplets, and grew more slowly. In mouse models of liver cancer, Elsulfavirine shrank tumors, and it worked even better when combined with the standard liver cancer drug Lenvatinib, suggesting a powerful one-two punch that targets both growth signals and metabolic demands.
What This Means for Patients
To connect these findings to real disease, the team examined human liver tumor samples. Tumors with high levels of phosphorylated ADSL, heavily succinated INSIG, and abundant SREBP in the nucleus were associated with poorer patient survival. In other words, the same sugar-sensing circuit mapped in cells and mice appears to be active in aggressive human liver cancers. The work shows that liver tumors can directly sense extra glucose and convert it into a pro-growth fat program through the “moonlighting” function of ADSL. It also highlights Elsulfavirine as a promising candidate for repurposing, opening a potential new strategy to cut off the fat supply that liver tumors need to thrive.
Citation: Duan, Y., Wang, S., Liu, J. et al. INSIG1/2 succination mediated by the moonlighting function of ADSL promotes lipogenesis and liver tumorigenesis. Nat Commun 17, 4002 (2026). https://doi.org/10.1038/s41467-026-70583-0
Keywords: liver cancer, cancer metabolism, lipogenesis, SREBP pathway, drug repurposing