Clear Sky Science · en
Extracellular vesicle engineering using a small scaffold protein
Tiny Couriers with Big Medical Promise
Many of today’s medicines struggle to reach exactly the right cells without causing side effects elsewhere. This study explores how to turn the body’s own nanosized “courier packets,” called extracellular vesicles, into smarter delivery vehicles for drugs and anti-inflammatory proteins. By discovering and refining a very small scaffold protein that helps pack cargo into these vesicles, the researchers build designer particles that calm dangerous inflammation in sepsis and protect worn joints in osteoarthritis. 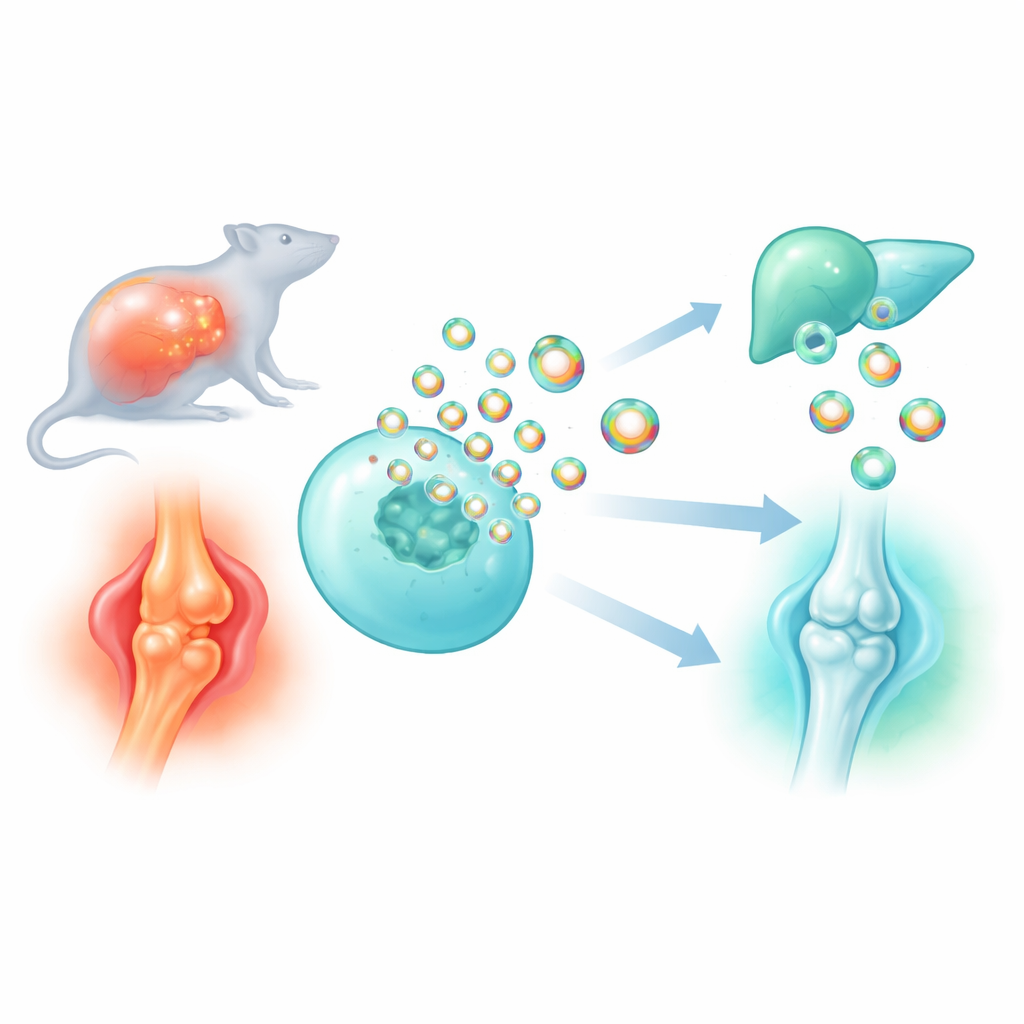
Natural Packages Turned into Targeted Carriers
Cells constantly shed tiny membrane bubbles known as extracellular vesicles (EVs). These packets naturally carry proteins and genetic material between cells and can slip through biological barriers that often block synthetic nanoparticles. That makes EVs attractive as living delivery trucks for therapy, but there is a catch: loading enough useful cargo inside them, or displaying the right molecules on their surface, has been difficult. Existing “scaffold” proteins that steer cargo into EVs can be bulky, complex, or unevenly present, limiting how much therapeutic material can be packed into each vesicle and raising safety concerns.
Finding a Small but Mighty Scaffold
To solve this, the team systematically surveyed proteins that appear on EVs produced by a commonly used human cell line. Using three independent purification methods and mass spectrometry, they shortlisted 15 candidates and fused each one to a fluorescent reporter to measure how efficiently it was sorted into vesicles. One stood out: ENPP1, which loaded cargo more effectively than widely used scaffolds such as PTGFRN and Lamp2b. The scientists then trimmed ENPP1 down to several shorter versions and discovered EN144, a fragment just 144 amino acids long, that packed three times more reporter protein into EVs than full-length ENPP1, while keeping vesicle size and structure intact. 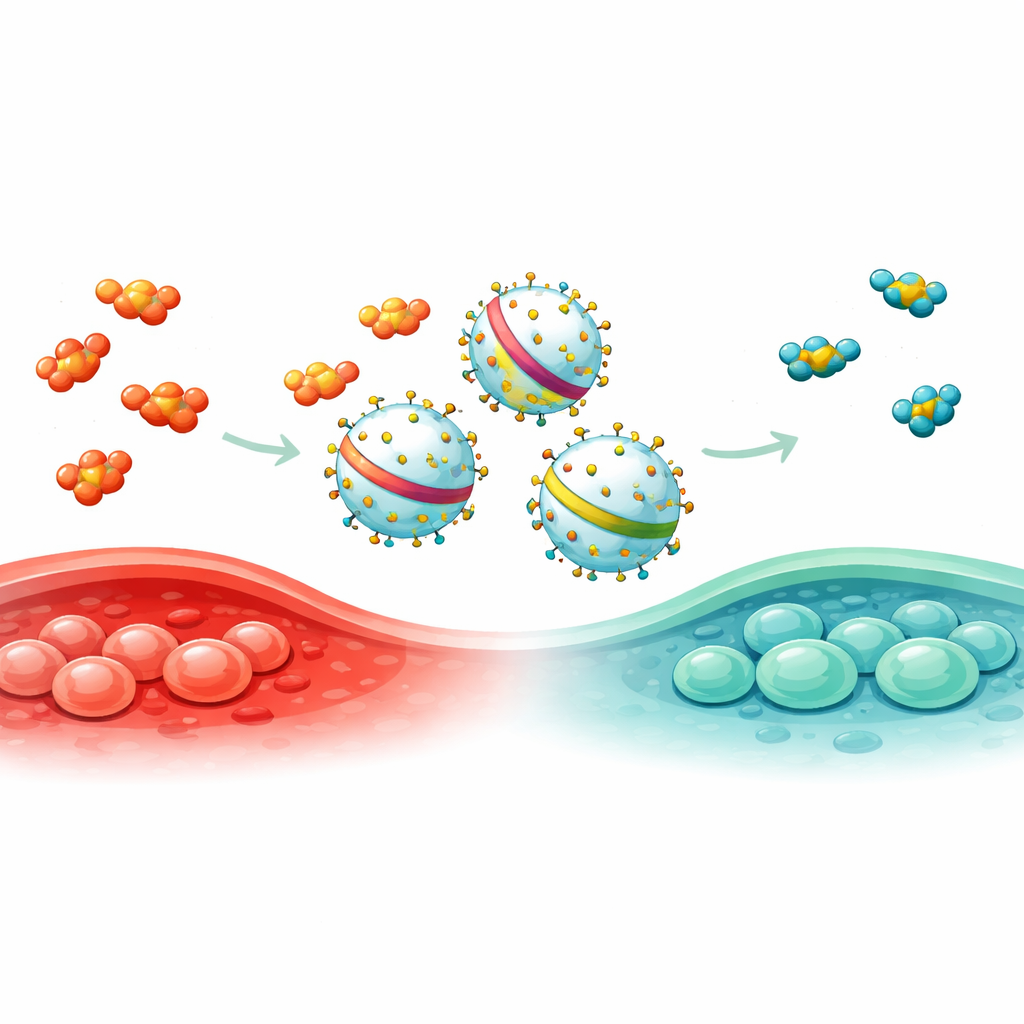
Safe, Versatile Vehicles for Genetic and Protein Cargo
Because any delivery system must be safe, the researchers tested EN144-decorated EVs in multiple cell types and in mice. The vesicles displayed typical EV markers, stayed within normal size ranges, and caused little to no toxicity, even at high or repeated doses. After injection into mice, EN144-EVs mainly accumulated in the liver and spleen, similar to standard EVs, without damaging organs or disturbing blood chemistry. The team then showed that EN144 can be used like a molecular docking station for many types of cargo. By coupling it to a cartilage-homing peptide, they created EVs that were taken up efficiently by chondrocytes, the cells that maintain joint cartilage. Using RNA-binding motifs, they enriched therapeutic mRNA inside EVs and confirmed that recipient cells translated that mRNA into protein. They also loaded CRISPR–Cas9 gene-editing complexes via engineered protein–protein pairs and demonstrated that EV-delivered editors could activate a silent reporter gene in target cells.
Decoy Vesicles that Soak Up Harmful Signals
Inflammatory diseases like sepsis and osteoarthritis are driven in part by the cytokine interleukin-6 (IL-6), which can act in a damaging “trans-signaling” mode when it forms complexes with a soluble receptor. To selectively block this harmful route without shutting down IL-6’s beneficial roles, the authors fused EN144 to gp130, a key IL-6 signal transducer, creating EVs that display gp130 as a decoy on their surface. In cell culture, these decoy vesicles sharply reduced the production of inflammatory cytokines. In a mouse model of sepsis, gp130-bearing EVs lowered IL-6 levels in organs, dampened downstream signaling, and greatly improved survival, outperforming doses of soluble gp130 protein. The vesicles specifically bound IL-6–receptor complexes, curbing the pro-inflammatory pathway while leaving classic IL-6 signaling, important for normal responses, largely intact.
Protecting Worn Joints by Aiming at Cartilage
To address osteoarthritis, the team combined targeting and decoy strategies. They added the cartilage-homing peptide to gp130-bearing EN144 EVs, generating particles that both home to chondrocytes and neutralize IL-6 trans-signaling. In cultured chondrocytes exposed to inflammatory cues, these targeted decoy EVs boosted cell survival and movement while reducing cell death. In rats with surgically induced osteoarthritis, repeated injections into the joint cavity led to strong retention of the EVs in cartilage, less breakdown of the joint surface, and healthier cartilage under the microscope. Markers of tissue damage fell, while structural proteins that maintain cartilage increased, and the balance of immune cells in the joint lining shifted toward a more healing, less inflammatory profile.
From Tiny Scaffold to Broad Therapeutic Platform
By carving out EN144 as a minimal, high-performance scaffold, this work turns naturally occurring extracellular vesicles into a flexible therapeutic platform. The same small anchor can display targeting tags and decoy receptors on the vesicle surface while loading genetic or protein cargo inside, all with a safety profile similar to unmodified vesicles. In animal models, EN144-based “decoy EVs” successfully tamed life-threatening inflammation and protected damaged joints. For a layperson, the implication is that we may be moving toward medicines built from the body’s own delivery packets, precisely engineered to carry the right tools to the right place, and to quiet harmful signals without silencing the body’s normal defenses.
Citation: Yan, W., Wang, S., Hao, H. et al. Extracellular vesicle engineering using a small scaffold protein. Nat Commun 17, 3726 (2026). https://doi.org/10.1038/s41467-026-70451-x
Keywords: extracellular vesicles, drug delivery, inflammation, sepsis, osteoarthritis