Clear Sky Science · en
Candesartan cilexetil disrupts methicillin-resistant Staphylococcus aureus membrane and potentiates gentamicin and polymyxin B activity
Turning a Blood-Pressure Pill into a Germ-Fighting Ally
Methicillin-resistant Staphylococcus aureus (MRSA) is a tough hospital and community bug that shrugs off many of our best antibiotics. This study asks a surprising question: can a common blood-pressure drug, candesartan cilexetil, be repurposed to weaken MRSA and make standard antibiotics work better? By probing MRSA cells from multiple angles—chemistry, genetics, imaging, and animal models—the researchers show how this pill can punch holes in bacterial defenses and revive older drugs.
Why Drug-Resistant Staph Is So Hard to Treat
MRSA causes hundreds of thousands of serious infections and thousands of deaths each year. One reason it is so difficult to treat is that the bacterium’s outer membrane and cell wall act as a smart shield. The fatty molecules in this shield can be rearranged to become harder, less permeable, and more resistant to standard antibiotics. Some MRSA cells also enter slow-growing “persister” states or hide in sticky biofilms, where they tolerate drugs that normally kill active bacteria. These features make it critical to find new ways to attack the bacterial surface rather than designing yet another traditional antibiotic from scratch.
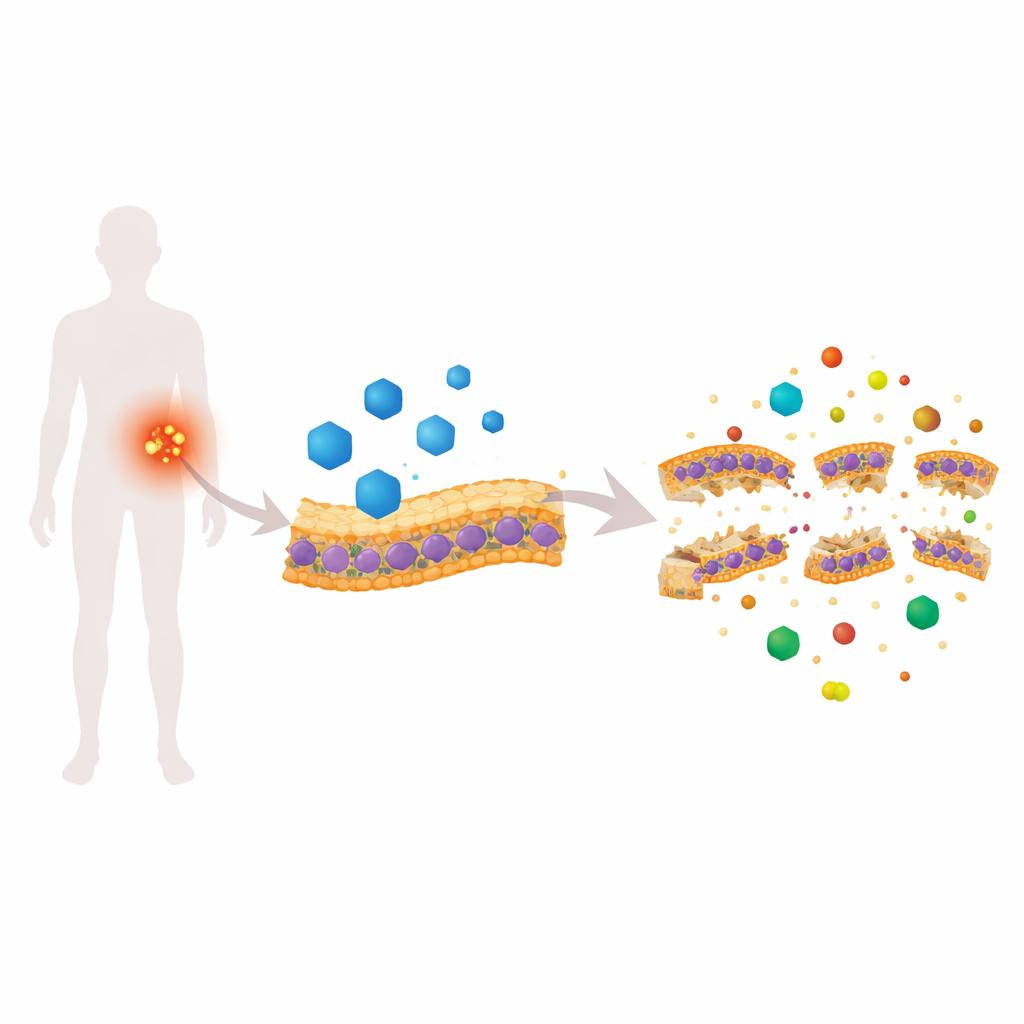
A Heart Medicine that Attacks Bacterial Skins
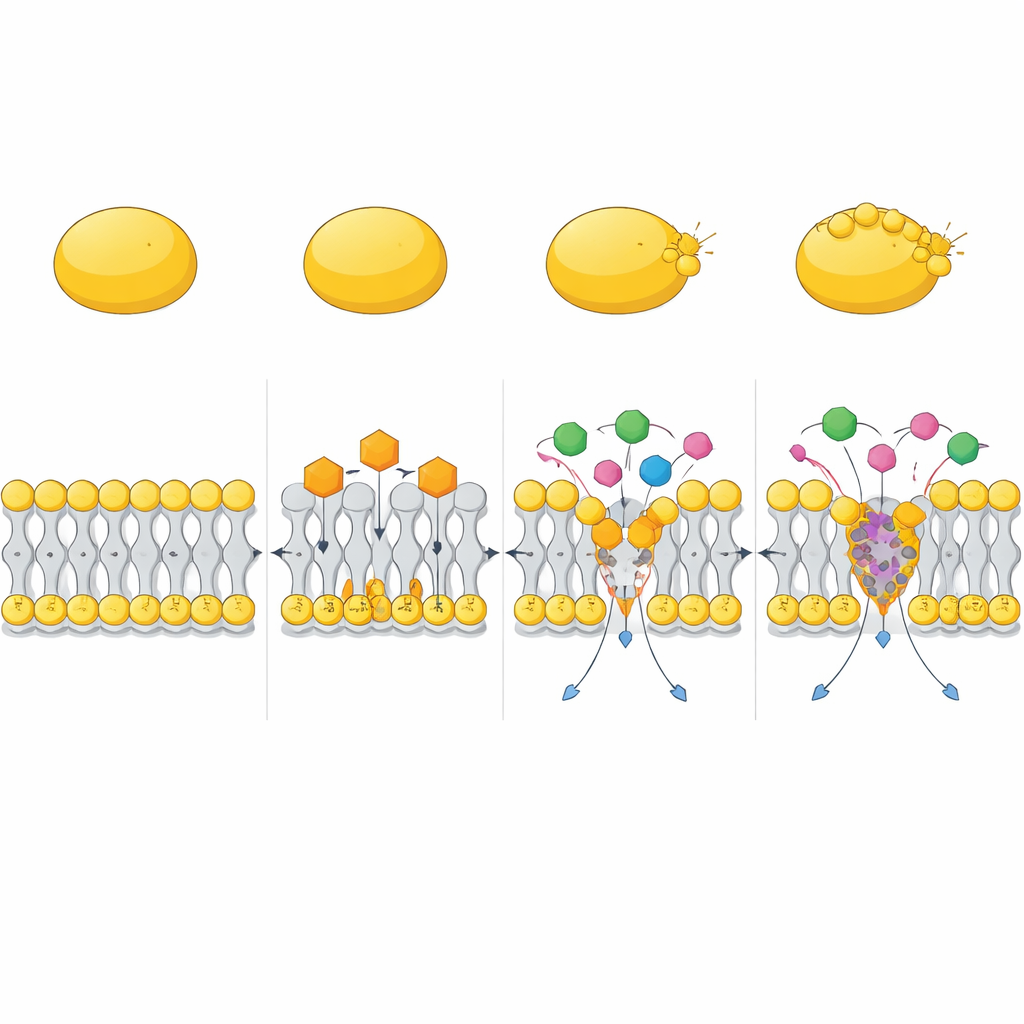
Candesartan cilexetil (CC) is approved to treat high blood pressure, but in a worm-based drug screen it unexpectedly showed strong activity against MRSA. In lab tests, CC killed actively dividing MRSA and the more stubborn stationary-phase cells, and it reduced biofilm formation and viability. Detailed experiments with fluorescent dyes revealed that CC quickly makes the MRSA membrane leaky, collapses the electrical gradient across it, and causes the inside of the cell to become unusually alkaline. High-resolution imaging—scanning electron microscopy, atomic force microscopy, transmission electron microscopy, and 3D cryo-tomography—showed blistered surfaces, thinning of the membrane, and visible gaps, all signs that the bacterial “skin” is physically compromised.
Rewiring the Bug’s Chemistry and Fatty Coat
To understand what CC is doing under the hood, the team combined RNA sequencing, metabolite profiling, and lipid analysis. They found that genes and small molecules involved in building the cell wall, making membrane lipids, and generating energy were broadly dialed down after CC exposure, even at doses too low to kill outright. Several key phospholipids that help keep the membrane intact dropped in abundance. A striking change involved a particular group of long, 20-carbon fatty acids (called C20:0). When the researchers used another compound, cerulenin, to deplete these fatty acids, MRSA became less sensitive to CC. Adding back C20:0 fatty acids restored much of the drug’s punch, and separate binding assays showed CC directly interacting with C20:0-linked lipids and related surface components. Measurements of membrane “fluidity” indicated that CC actually stiffens the MRSA membrane, locking lipids into a more rigid state that ultimately undermines its function.
Teaming Up with Existing Antibiotics
The study also tested how CC behaves alongside existing drugs. In checkerboard and time-kill experiments, CC dramatically boosted the activity of aminoglycoside antibiotics such as gentamicin, as well as polymyxin B. These combinations rapidly wiped out both actively growing MRSA and hard-to-kill persister cells, whereas the individual drugs often left survivors. Computer simulations suggested one reason: CC can form compact complexes with polymyxin B that insert into the membrane and distort it more powerfully than either molecule alone, creating deep, far-reaching deformations in the lipid layer. This helps other antibiotics cross the membrane barrier and reach their targets more efficiently.
From Petri Dish to Living Animals
Encouragingly, CC’s antibacterial and helper effects extended beyond the test tube. In tiny worms and wax moth larvae infected with MRSA, CC improved survival. In a mouse thigh infection model using a virulent MRSA strain, CC alone reduced bacterial counts, and its combination with gentamicin cut the bacterial burden by roughly a thousand-fold compared with untreated controls. Blood measurements showed that drug levels similar to those effective in the lab could be reached in mice without obvious damage to liver, kidney, or heart tissues over the course of the experiment.
What This Could Mean for Future Treatments
In plain terms, this work reveals that a long-used blood-pressure drug can double as a membrane-targeting weapon against MRSA. By latching onto specific fatty building blocks in the bacterial envelope, stiffening the membrane, and poking holes in it, candesartan cilexetil both kills MRSA outright and opens the door for older antibiotics to work again. While more safety and dosing studies are needed—especially because this medicine also acts on human blood vessels—the findings suggest a promising shortcut: instead of inventing entirely new antibiotics, we might retool existing drugs to weaken bacterial defenses and restore the power of treatments we already have.
Citation: Tharmalingam, N., Kovacs, R.W., Scarpa de Mello, S. et al. Candesartan cilexetil disrupts methicillin-resistant Staphylococcus aureus membrane and potentiates gentamicin and polymyxin B activity. Nat Commun 17, 4012 (2026). https://doi.org/10.1038/s41467-026-70173-0
Keywords: MRSA, antibiotic resistance, drug repurposing, bacterial membrane, candesartan cilexetil