Clear Sky Science · en
Asymmetric synthesis of Heteroatom-bridged [3.2.1]Octane scaffolds via enantioselective β-H elimination reaction
Why these tiny ring-shaped molecules matter
Many important medicines, especially those that act on the brain, are built around a compact, ring-shaped structure called a tropane. These molecular “skeletons” help drugs latch onto targets involved in diseases such as Alzheimer’s and depression. Yet making tropane-based drug candidates in a single, preferred mirror-image form has been slow, costly, and technically demanding. This study introduces a more direct and efficient way to build these complex frameworks from a cheap starting material, opening the door to faster discovery of new treatments.
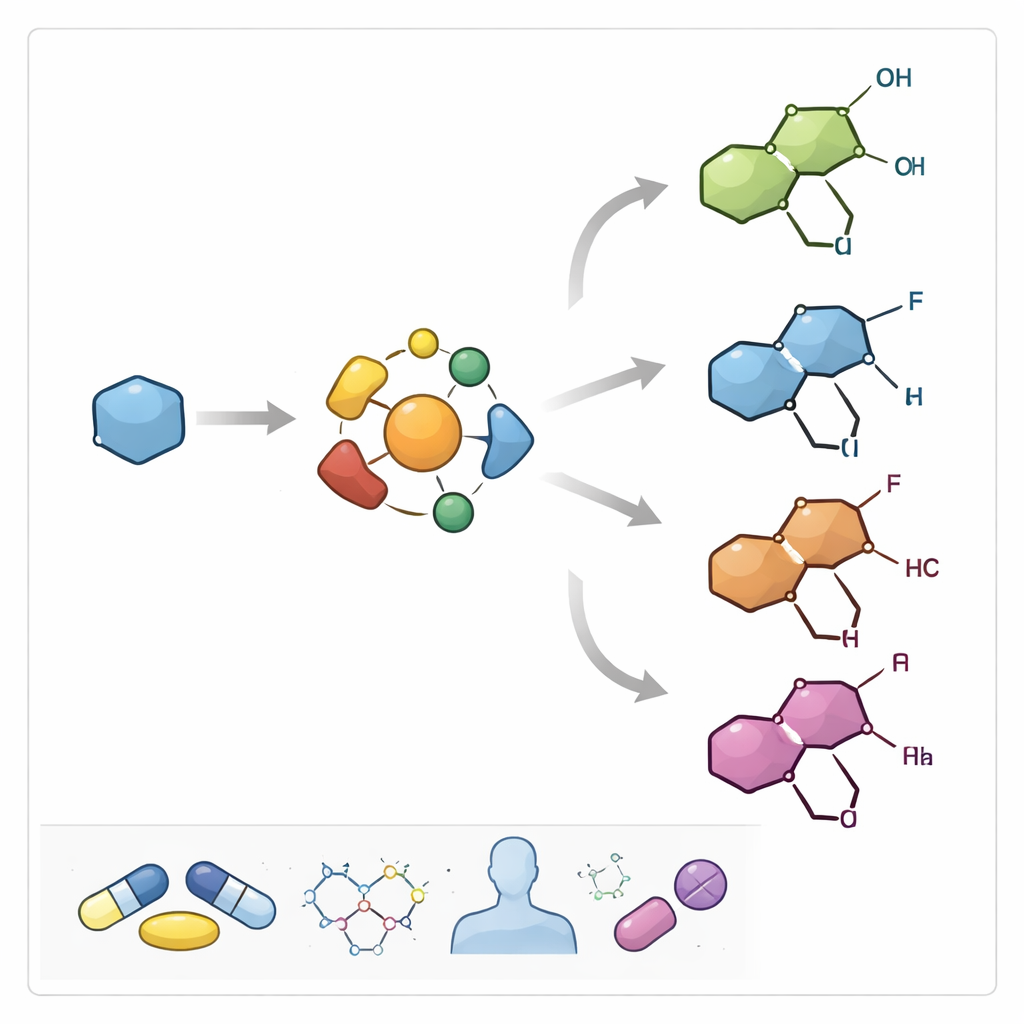
Turning a simple starting block into a rich family of drugs
The researchers begin with tropinone, an inexpensive compound that already contains the basic ring layout found in many natural tropane alkaloids. From this simple starting block, they convert it into highly versatile building units called N-arylsulfonylhydrazones. These are then coupled with a wide range of aryl bromides—molecules that carry different aromatic “decorations”—to assemble a broad family of N-bridged [3.2.1]octane and related scaffolds. The key advance is that this transformation happens in a way that strongly favors one mirror-image form over the other, giving access to chiral tropanes that are particularly valuable in drug design.
A designer catalyst that steers molecular traffic
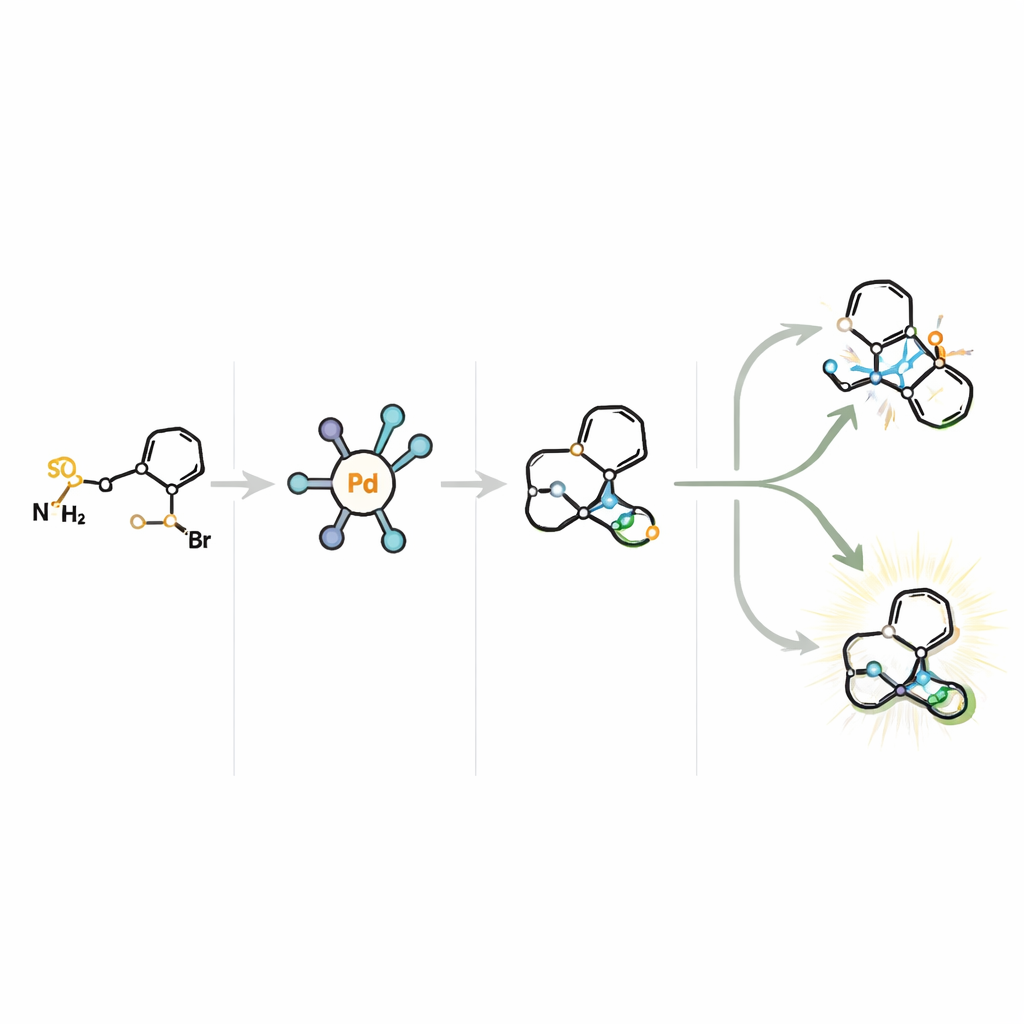
At the heart of the method is a tailor-made palladium catalyst partnered with a chiral ligand called Ming-Phos. The reaction proceeds through a short‑lived, highly reactive carbene stage followed by a subtle step in which a hydrogen atom is removed from a neighboring carbon atom—known as β‑H elimination. In this system, two distinct sites can undergo this elimination, leading in principle to four different pathways. The catalyst must therefore control not only which face of the molecule reacts (its handedness) but also which hydrogen is removed. By carefully shaping the Ming-Phos ligand—tuning its substituents and positioning key groups so they can form a hydrogen bond to the reacting partner—the authors create a catalyst “pocket” that guides the molecule along a single, favored route, delivering products in high yield and excellent enantiomeric purity.
From reaction scope to real medicinal targets
Under optimized conditions, the method tolerates a wide array of functional groups on the aryl bromides, including electron‑rich and electron‑poor rings, bulky substituents, and heterocycles such as pyridines. It also works when the bridgehead nitrogen is swapped for oxygen, giving oxa‑tropane analogs. The team demonstrates that these products are not just laboratory curiosities: they adapt the reaction to build advanced intermediates for pharmaceuticals aimed at treating Alzheimer’s disease and for ligands that bind monoamine transporters, key proteins involved in mood and cognition. Compared with older routes that required harsh conditions, toxic gases, or laborious chiral resolution, the new strategy delivers these targets in fewer steps, under milder conditions, and with much better control of molecular handedness.
Peering under the hood of the new reaction
To understand how the process works, the authors conduct detailed mechanistic studies. By varying the electronic nature of the aryl bromide partners and measuring how this affects reaction rates, they show that the initial bond‑forming step with the aryl bromide is unlikely to be the slow, controlling step. Isotope labeling experiments further suggest that the β‑H elimination itself is not rate‑limiting. Instead, the data point to the formation of the palladium–carbene intermediate, derived from the sulfonylhydrazone, as the bottleneck. Additional tests rule out a pathway involving free radicals and support a picture in which hydrogen bonding between the Ming‑Phos ligand and the reacting partner helps organize the transition state, leading to precise control over which β‑hydrogen is removed and which mirror-image product is formed.
New routes to brain drugs and beyond
Overall, this work shows that a carefully crafted catalyst can tame a notoriously hard‑to‑control step and turn it into a powerful tool for building intricate molecular frameworks. By enabling efficient, scalable, and highly selective access to chiral tropane and oxa‑tropane scaffolds from a cheap feedstock, the method streamlines the synthesis of potential Alzheimer’s drugs, monoamine transporter ligands, and other bioactive molecules. For non‑specialists, the key message is that chemists now have a more precise and economical way to construct a crucial class of drug backbones, which could accelerate the development of new treatments for brain disorders and possibly cancer.
Citation: Fang, C., Ai, J., Wang, Q. et al. Asymmetric synthesis of Heteroatom-bridged [3.2.1]Octane scaffolds via enantioselective β-H elimination reaction. Nat Commun 17, 3315 (2026). https://doi.org/10.1038/s41467-026-69960-6
Keywords: tropane scaffolds, enantioselective catalysis, palladium chemistry, drug discovery, beta-hydrogen elimination