Clear Sky Science · en
A gated hydrophobic funnel within BAX binds bioactive lipids to potentiate pro-apoptotic function
How Fats Help Cells Decide When to Die
Every day, millions of our cells quietly self‑destruct to keep tissues healthy and remove damaged or dangerous cells. This built‑in suicide program, called apoptosis, depends on proteins that can punch holes in the membranes of mitochondria, the cell’s power plants. This study uncovers how a particular fat‑like molecule helps flip one of these proteins, BAX, from a quiet bystander into an efficient executioner—revealing a hidden “funnel” in BAX that could one day be targeted by new medicines.
A Cell’s Point of No Return
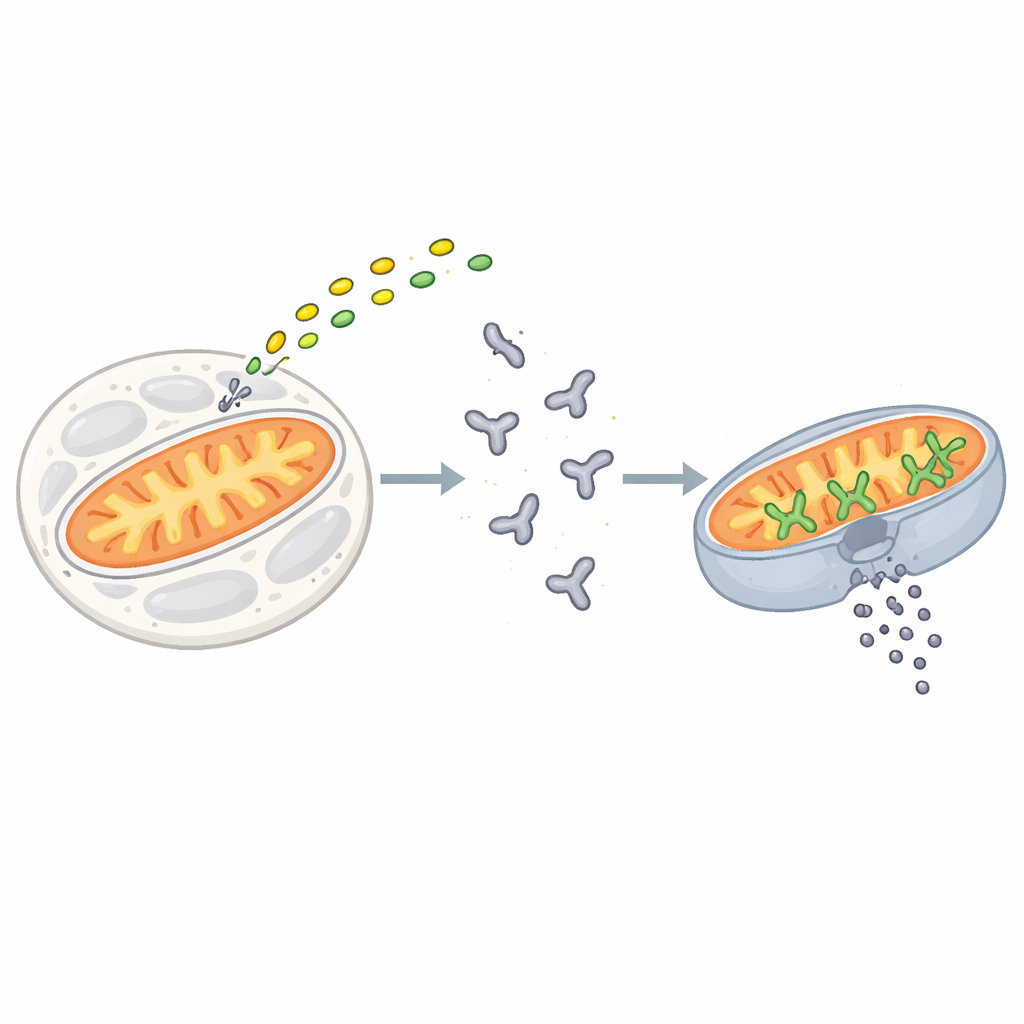
Apoptosis is triggered when stress or damage signals converge on a family of proteins known as BCL‑2 proteins. BAX is one of the key enforcers in this family. In resting cells, BAX drifts in the watery interior as a single, folded unit. When death signals arrive, short “activator” proteins such as BIM latch onto BAX and start a series of shape‑changes that move BAX to the outer surface of mitochondria. There, BAX assembles into clusters that open large pores in the membrane, allowing internal contents to spill out. This irreversible event, called mitochondrial outer membrane permeabilization, marks the point of no return for a cell.
A Reactive Lipid Partner Enters the Scene
Earlier work suggested that a breakdown product of the signaling molecule sphingosine‑1‑phosphate, called 2‑trans‑hexadecenal, is required for BAX to form pores efficiently. But it was unclear whether this oily molecule simply altered the mitochondrial membrane or whether it touched BAX directly. The authors first tested how added 2‑trans‑hexadecenal affected living mouse cells. By tracking cell death in real time, they found that the lipid by itself caused limited apoptosis, but in the presence of a drug that disables BAX’s anti‑death partners, cell death rose sharply—and only when BAX and its close relative BAK were present. In purified systems using artificial membrane bubbles, 2‑trans‑hexadecenal did not damage membranes on its own, but strongly boosted the ability of BAX, especially when BIM was also present, to punch holes.
A Hidden Funnel Inside BAX
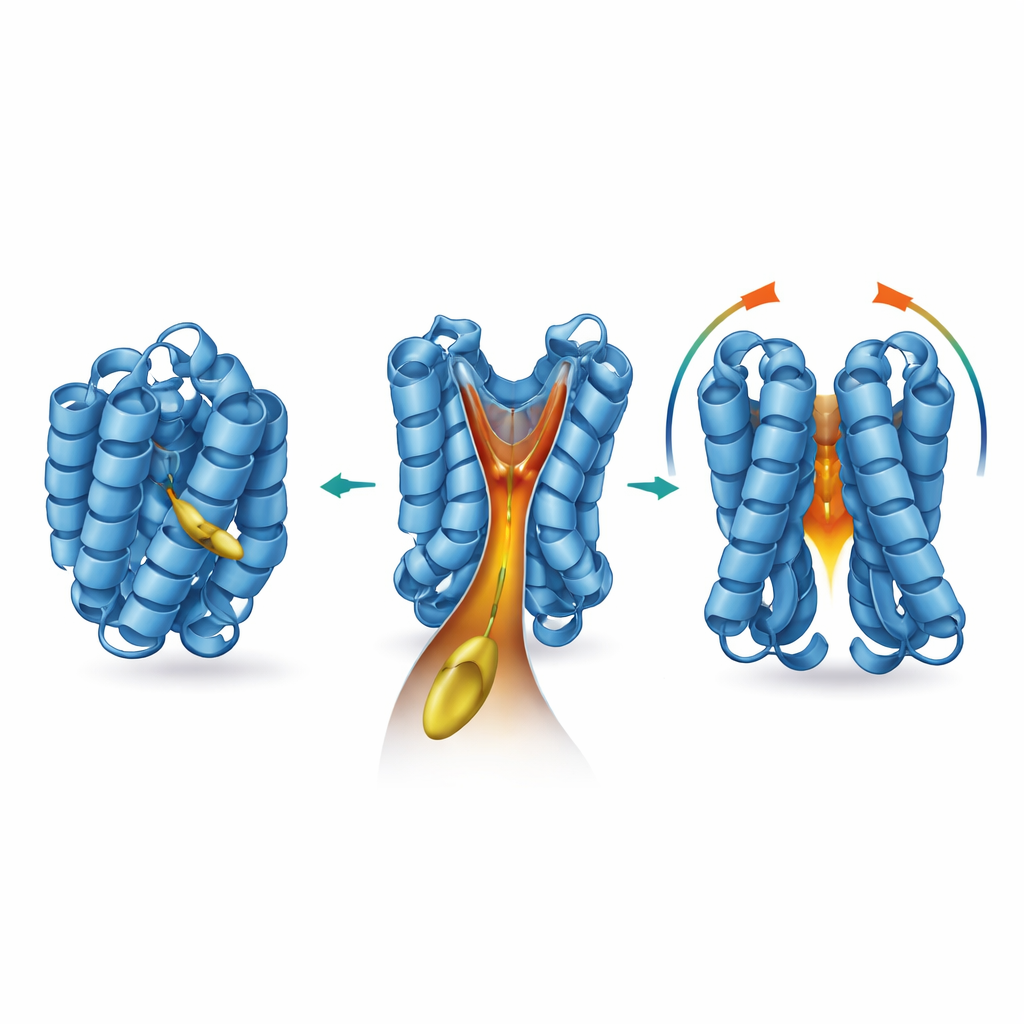
To find out where and how this lipid acts, the team turned to a battery of structural and biophysical tools. They showed that 2‑trans‑hexadecenal binds to BAX without forming permanent chemical bonds, and that removing BAX’s natural cysteine “handles” does not blunt the effect. Nuclear magnetic resonance experiments revealed that binding causes subtle shifts in specific amino acids buried inside BAX, especially around helices known as α5, α6, and α8. Computer models based on these changes uncovered a previously overlooked, funnel‑shaped cavity at the core of BAX, capped by the flexible α8 helix. When activator BIM engages BAX, α8 moves, the funnel widens, and docking simulations predict that the lipid chain of 2‑trans‑hexadecenal fits snugly into the deep neck of this hydrophobic tunnel. The authors name this pocket the “BAX actuating funnel,” or BAF.
Tuning the Funnel with Chemistry and Mutations
The researchers then asked what features of the lipid and of BAX itself are crucial for this partnership. Shorter or chemically altered versions of the lipid could still brush against BAX but were far less able to trigger its activation or pore‑forming activity. In contrast, long‑chain versions most similar to 2‑trans‑hexadecenal were best at switching BAX on, indicating that chain length and head‑group structure must match the funnel’s shape. When the team introduced precise mutations that slightly filled in or distorted the funnel walls, BAX still responded to BIM but lost its sensitivity to 2‑trans‑hexadecenal. A naturally important residue, proline 168, which sits in a hinge between two helices, turned out to control the funnel at a distance. Changing this single amino acid reshaped the cavity, stabilized BAX in its resting form, and made both purified proteins and cells far less responsive to the lipid co‑factor.
How Lipids and Proteins Team Up to End a Cell
Taken together, the work supports a stepwise view of how BAX is activated. First, activator proteins like BIM bind BAX and start early rearrangements, including loosening of the α8 “gate” helix. This motion exposes the BAX actuating funnel, into which 2‑trans‑hexadecenal can insert like a wedge. Occupying this internal tunnel destabilizes BAX’s tightly packed core, favors exposure of its interaction surface, and promotes the formation of dimers and larger clusters that perforate the mitochondrial membrane. Rather than acting as a simple membrane solvent, this bioactive lipid emerges as a true molecular partner that tunes BAX’s readiness to kill. By defining the BAF as a critical control site, the study opens the door to designing small molecules that either boost BAX activity—potentially useful against cancer—or dampen it to protect healthy tissues in degenerative or inflammatory diseases.
Citation: Gelles, J.D., Chen, Y., Luna-Vargas, M.P.A. et al. A gated hydrophobic funnel within BAX binds bioactive lipids to potentiate pro-apoptotic function. Nat Commun 17, 3180 (2026). https://doi.org/10.1038/s41467-026-69836-9
Keywords: BAX, apoptosis, bioactive lipids, mitochondria, protein-lipid interaction