Clear Sky Science · en
High efficiency CRISPR knock-in demonstrates that TCF1 is insufficient to reverse T cell exhaustion
Why Tired Immune Cells Matter
Our immune system relies on killer T cells to hunt down virus-infected and cancerous cells. But when these threats linger for months, T cells can become “exhausted,” losing much of their firepower. A special subset of these cells behaves more like stem cells, continually renewing the response and helping modern cancer immunotherapies work. This study asks a deceptively simple question with big treatment implications: can we push worn-out T cells back into this youthful, stem-like state by turning on a single master control gene called TCF1?
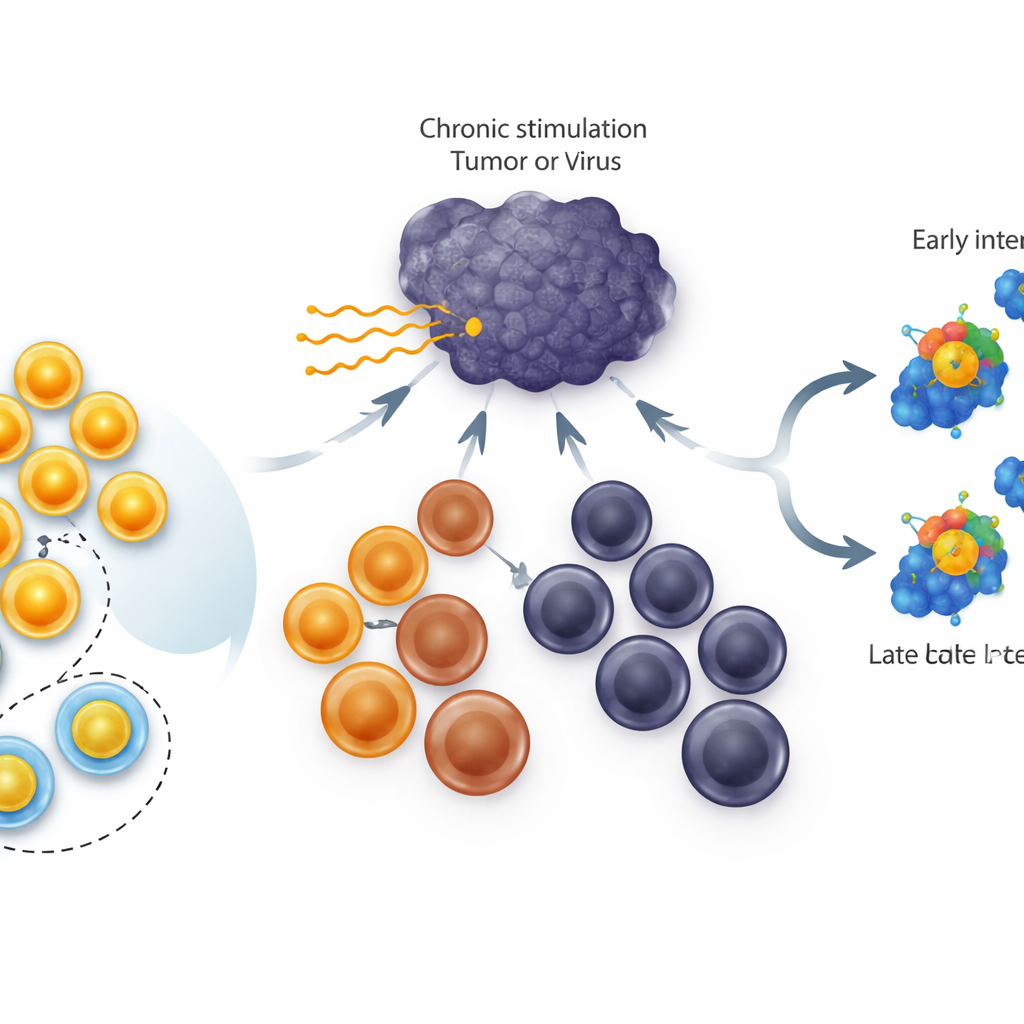
Different Flavors of Tired T Cells
In chronic infections and tumors, killer T cells do not just switch off; they pass through a ladder of states. At the top are stem-like cells that divide well and can spawn new fighters. In the middle sit “intermediate” cells that still contribute to control of disease, and at the bottom are terminally exhausted cells that have little flexibility left. The gene TCF1 is highly active in the stem-like group and is known to be essential for their formation and survival. Previous work showed that constantly boosting TCF1 in all exhausted cells increases the size of the stem-like pool. What was unknown is whether TCF1 can actually rewind more advanced, exhausted cells back up the ladder into that stem-like state.
Building a Precise Gene-Editing Toolkit in Mice
To tackle this, the researchers first needed a highly efficient way to insert new DNA into mouse T cells without disrupting their natural behavior inside the body. They optimized a homology-directed repair form of CRISPR editing that uses an AAV6 viral template to “knock in” new genetic cargo at precise spots in the genome. By carefully activating T cells, delivering CRISPR components, briefly resting the cells in the lab, and then transferring them into mice, they could reach knock-in rates of 40–80 percent—levels comparable to or better than traditional viral overexpression. They also showed how to insert genes without breaking important host genes, and how to target specific genetic addresses so that a new gene turns on only in particular T cell subsets.
Switching TCF1 On Early Versus Late
The heart of the study compares two strategies for turning on TCF1. In one, the team inserted TCF1 into a location that is active in all T cells, so TCF1 was high from the start of the response. In the other, they inserted TCF1 into the Cx3cr1 gene, which becomes active only when stem-like cells mature into the intermediate exhausted state. This second design acts like a conditional switch, flipping TCF1 on only after the cells have already moved down the exhaustion path. When the modified T cells were tested in a well-established chronic viral infection model, constant TCF1 expression clearly expanded the stem-like pool. In contrast, turning TCF1 on only in intermediate cells did not increase the number of stem-like cells, even though those intermediate cells produced as much or more TCF1 per cell as in the constant-expression setup.
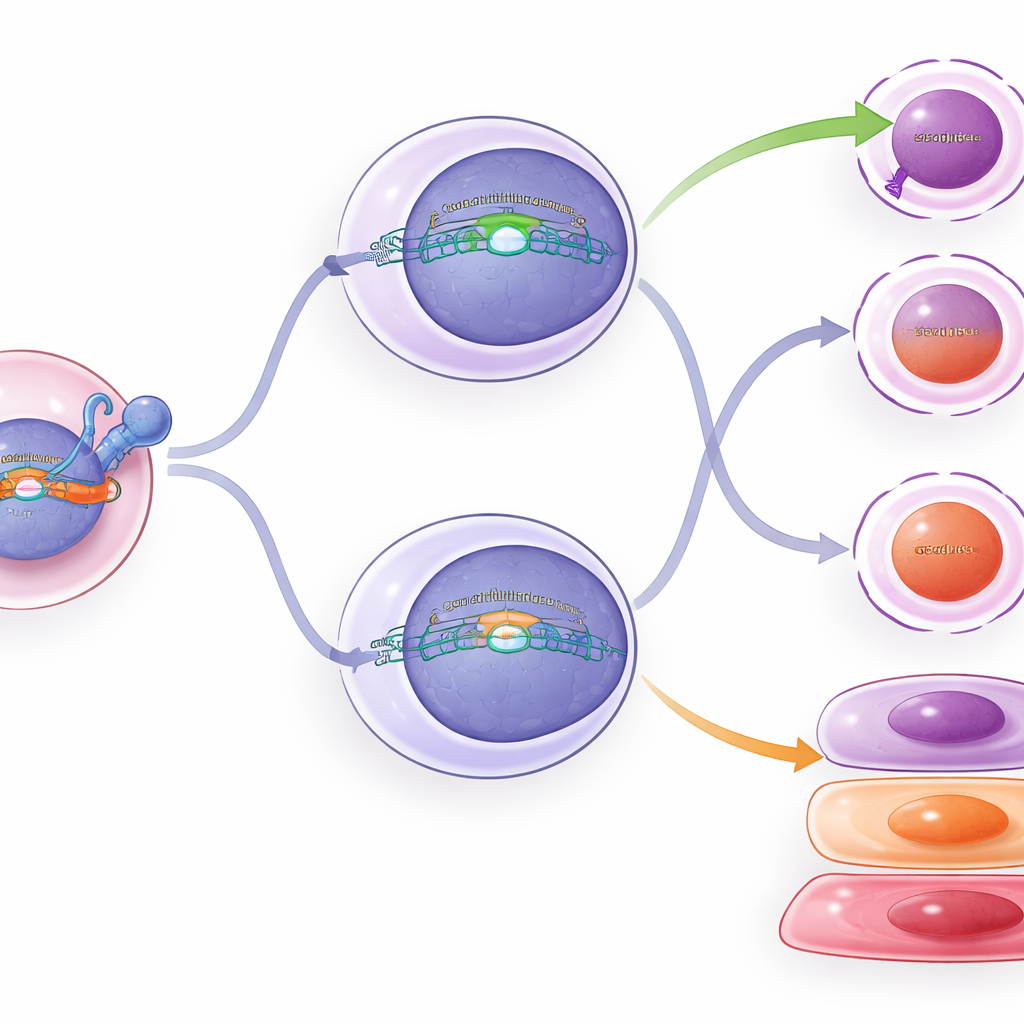
What Happens Inside the Rewired Cells
To see beyond simple cell counts, the team profiled gene activity and chromatin (the DNA packaging that controls which genes can turn on) in thousands of individual T cells. Cells with constant TCF1 showed more stem-like clusters and early effector-like cells, consistent with TCF1 slowing the march toward deep exhaustion. Conditional TCF1 in intermediate cells caused only subtle shifts: some gene patterns and chromatin features nudged toward those of stem-like cells, and markers of exhaustion dipped slightly. Flow cytometry confirmed these mild changes—intermediate cells with added TCF1 had a bit less of certain killing and exhaustion proteins, and a bit more of a marker associated with stem-like cells—but they did not fully revert in identity.
Why One Gene Cannot Turn Back the Clock
For a non-specialist, the key message is that once T cells have travelled far enough down the exhaustion road, simply reactivating TCF1 is not enough to send them back to a youthful, stem-like state. Constant TCF1 helps preserve stem-like cells if it is present from early on, effectively slowing aging, but it cannot undo exhaustion after the fact. This has important implications for cancer immunotherapy: strategies that try to rejuvenate heavily exhausted T cells by boosting TCF1 alone are unlikely to work. Instead, successful therapies may need to combine several factors or focus on protecting and expanding the stem-like pool before cells become too exhausted to rescue.
Citation: de Menezes, M.N., Chen, A.X.Y., Kulkarni, N. et al. High efficiency CRISPR knock-in demonstrates that TCF1 is insufficient to reverse T cell exhaustion. Nat Commun 17, 2857 (2026). https://doi.org/10.1038/s41467-026-69671-y
Keywords: T cell exhaustion, TCF1, CRISPR knock-in, cancer immunotherapy, stem-like T cells