Clear Sky Science · en
TGFβ-activated PDHB promotes mitochondrial pyruvate metabolism and contributes to human endoderm differentiation via ATP-dependent BRG1
How Fuel Use Shapes Early Human Development
Our bodies begin as tiny balls of stem cells that must decide what kinds of tissues to become. This study asks a surprisingly down-to-earth question: how does the way these early cells burn sugar influence their decision to form organs like the liver, lungs and pancreas? By tracking energy use and DNA regulation side by side, the researchers show that a specific shift in sugar metabolism is not just a backdrop but an active driver of early human development.
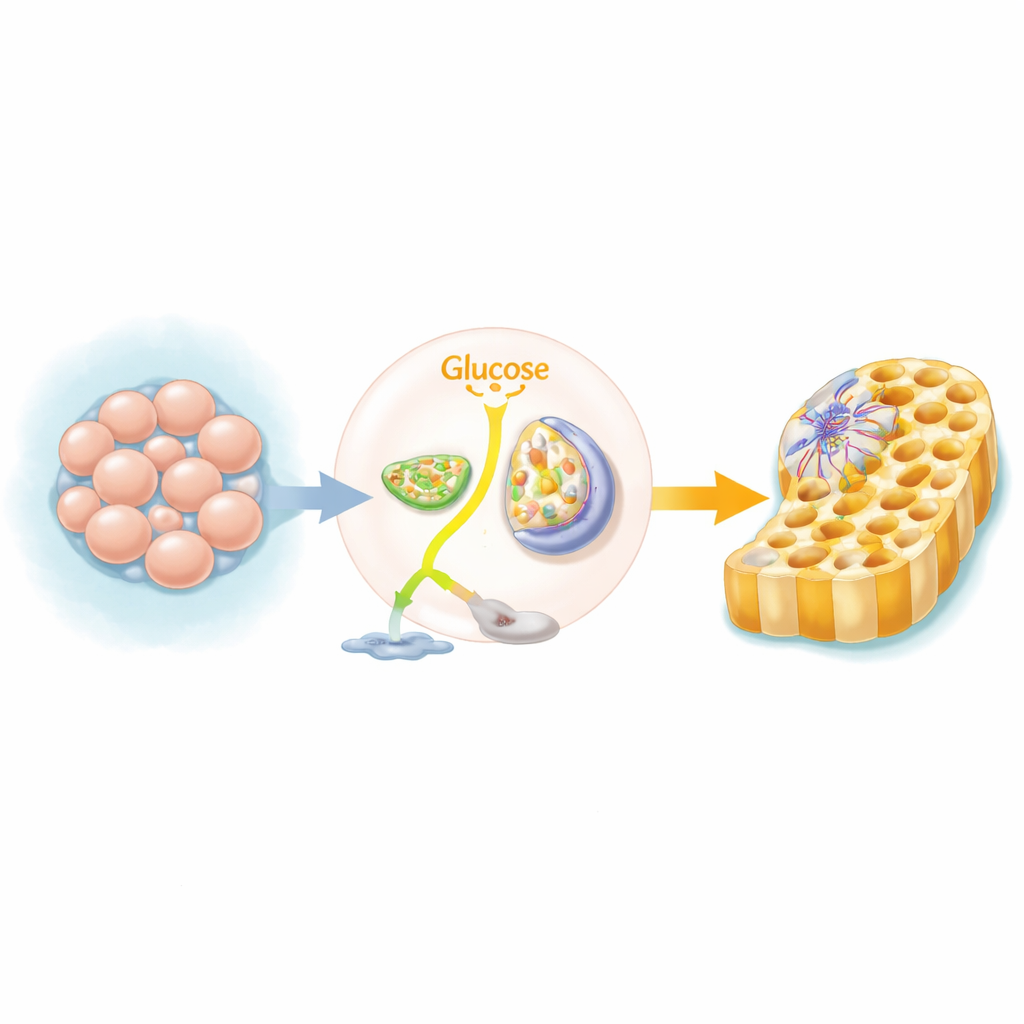
A Metabolic Switch in Budding Tissues
Human pluripotent stem cells, which can form almost any tissue, normally rely on a fast but inefficient way of using sugar, breaking glucose down to lactate even when oxygen is available. As these cells begin to form the “definitive endoderm” layer—the precursor of gut, liver and pancreas—they flip their main fuel route. Instead of shunting sugar to lactate, they send more of it into mitochondria, the cell’s power stations, to run the tricarboxylic acid (TCA) cycle and oxidative phosphorylation. The team used genome-wide CRISPR screens and a battery of chemical blockers to show that when this switch is blocked, far fewer cells successfully adopt endoderm identity.
Turning Sugar into Mitochondrial Power
At the heart of this switch lies pyruvate, a key breakdown product of glucose. Pyruvate can either be converted to lactate or imported into mitochondria and turned into acetyl-CoA, the entry ticket to the TCA cycle. The researchers found that steering pyruvate toward mitochondria strongly favors endoderm formation. Drugs that inhibit lactate-making enzymes push more pyruvate into mitochondria and boost differentiation, while compounds that prevent pyruvate from entering mitochondria or being processed there sharply reduce endoderm markers. Supplementing cells with pyruvate or glutamine, another fuel for the TCA cycle, helps restore this developmental path when early steps of sugar use are blocked.
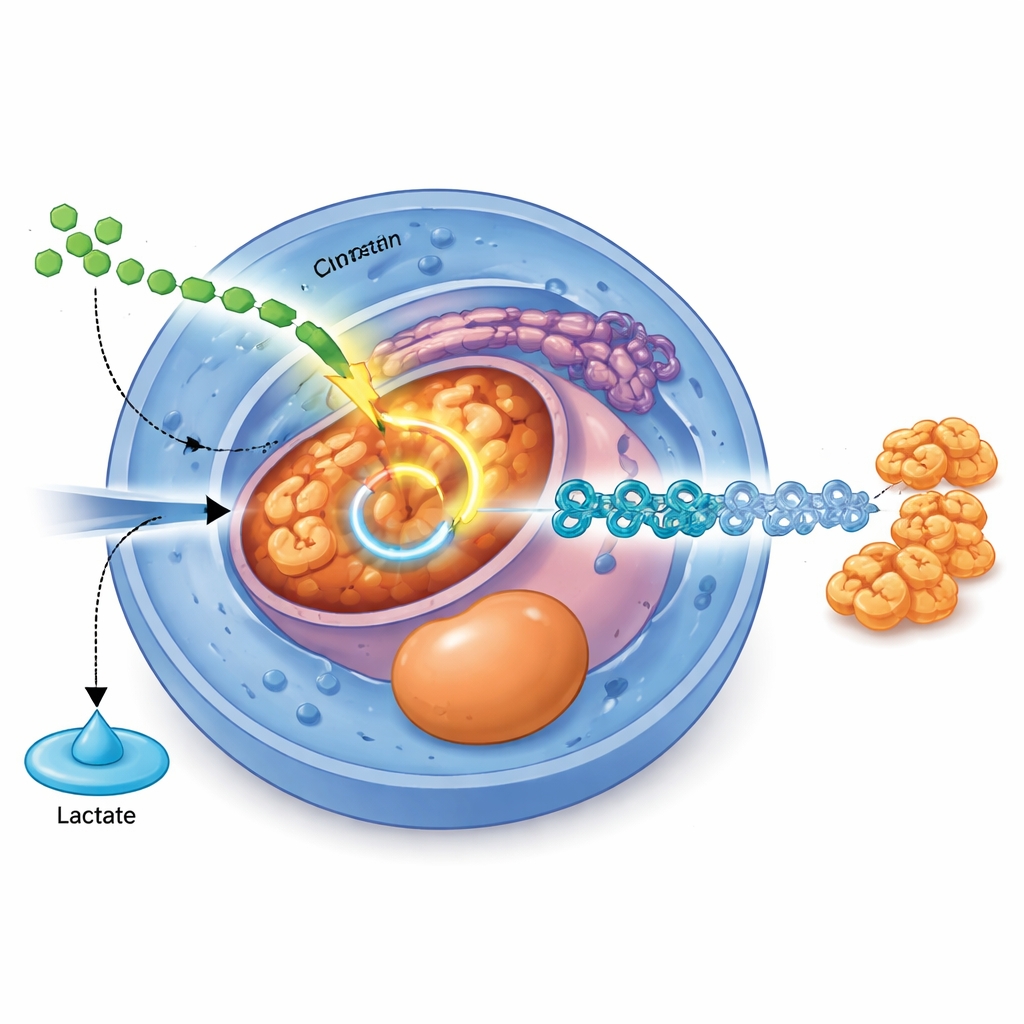
A Signal Pathway That Rewires Metabolism
The study identifies a specific molecular switch that links outside cues to this inner fuel rerouting. A well-known growth signal called TGFβ, acting through its messenger proteins SMAD2 and SMAD3, directly boosts production of PDHB, a core component of the pyruvate dehydrogenase complex that feeds pyruvate into the TCA cycle. When PDHB levels are reduced using CRISPR editing, cells produce more lactate, less acetyl-CoA, and struggle to progress from stem cells toward endoderm and later pancreatic stages. Conversely, raising PDHB levels enhances mitochondrial activity, increases cellular energy, and improves the efficiency of endoderm differentiation, especially when the TGFβ signal itself is weak.
Energy Levels Control Access to the Genetic Script
Why does this fuel choice matter so much for identity? The authors trace the effect to ATP, the cell’s main energy currency. Blocking glucose breakdown or mitochondrial entry lowers ATP levels, and simply supplying extra ATP from outside can rescue failed endoderm differentiation in several settings. The key ATP consumer is the BAF complex, a large “chromatin remodeling” machine centered on the protein BRG1 that uses ATP to open up folded DNA. When BAF is chemically inhibited or BRG1 is degraded or mutated so that its ATP-driven motor no longer works, endoderm formation falters and extra ATP can no longer help. Genome-wide mapping of chromatin accessibility shows that, when PDHB or mitochondrial pyruvate use is reduced, many regions near endoderm-related genes become less accessible, especially enhancer regions where BRG1 normally binds.
What This Means for Building Organs from Cells
Taken together, the work outlines a clear chain of events: TGFβ signals raise PDHB levels, PDHB routes pyruvate into mitochondria, mitochondrial metabolism raises ATP, and ATP powers BRG1-driven opening of key DNA regions so that endoderm genes can turn on. In simple terms, the way early human cells burn sugar determines how much “energy budget” they have to unlock the parts of the genome needed to become gut- and organ-forming tissue. Understanding and controlling this fuel switch could improve methods for growing specific human tissues from stem cells for research, disease modeling, and eventual cell-based therapies.
Citation: Meng, L., Lv, J., Yi, Y. et al. TGFβ-activated PDHB promotes mitochondrial pyruvate metabolism and contributes to human endoderm differentiation via ATP-dependent BRG1. Nat Commun 17, 2846 (2026). https://doi.org/10.1038/s41467-026-69510-0
Keywords: stem cell metabolism, endoderm differentiation, mitochondrial pyruvate, chromatin remodeling, TGF beta signaling