Clear Sky Science · en
A neuron type-specific microexon in Ank3/ankyrin-G modulates calcium activity and neuronal excitability
How tiny genetic edits shape brain cell behavior
Brains depend on a delicate balance between nerve cells that excite circuits and those that calm them down. This study shows that a tiny genetic fragment—just nine building blocks in a single protein—helps set how strongly some inhibitory brain cells fire and how they handle calcium signals. Because the same gene is tied to bipolar disorder and other brain conditions, understanding this microscopic switch offers clues to how subtle molecular tweaks can ripple up to behavior and disease risk.
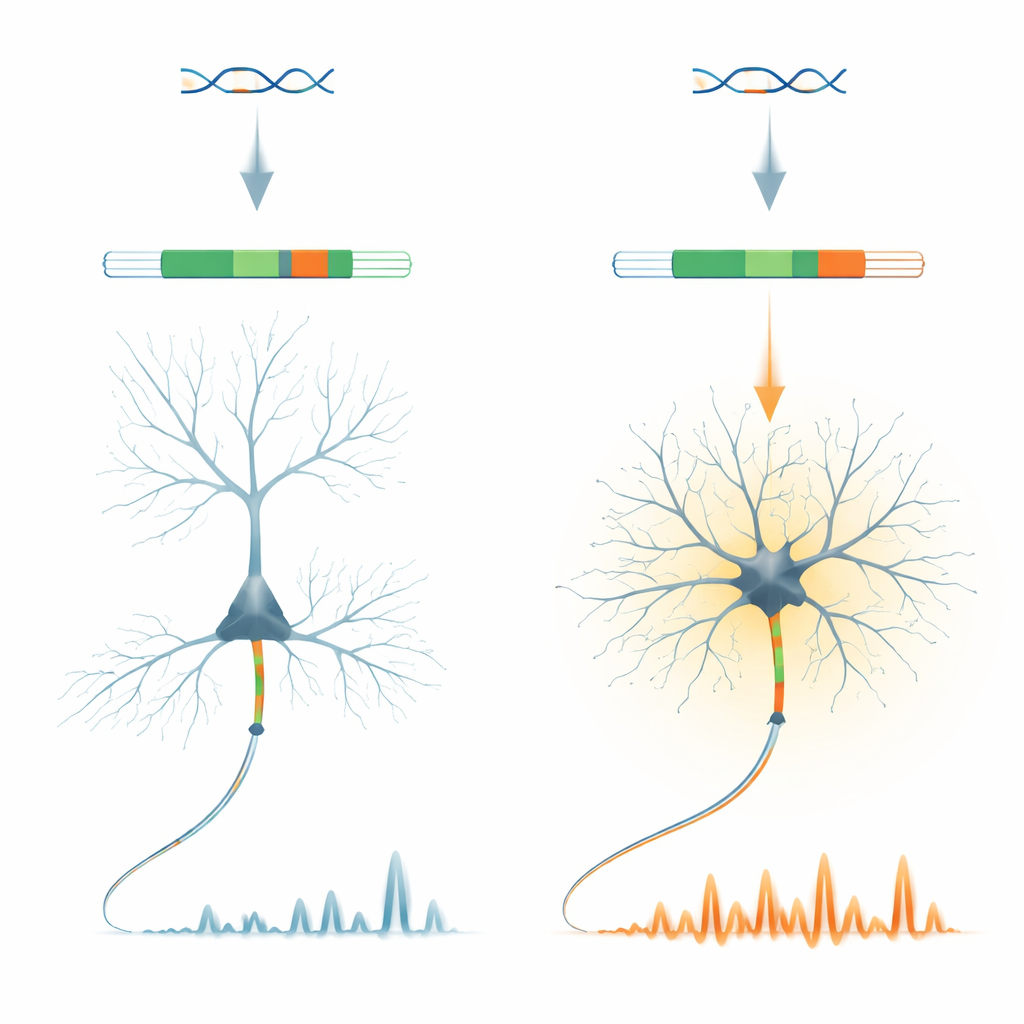
A small genetic switch with a big job
The researchers focus on a protein called ankyrin-G, a key organizer of the region where nerve signals are first generated in an axon. Within the Ank3 gene that encodes ankyrin-G, they identify a very short "microexon" called E35a. This snippet is used very differently across neuron types: it is mostly left out in excitatory pyramidal neurons of the cortex, but commonly included in many inhibitory GABA-producing neurons and in cerebellar neurons. Remarkably, this pattern is conserved across vertebrates, suggesting that evolution has strongly preserved this neuron-type-specific on/off switch.
Different brain cells, different splicing choices
Cells decide whether to include E35a through a coordinated network of RNA-binding proteins that control how raw RNA is cut and stitched. By reanalyzing large datasets and testing a simplified “minigene” reporter in cells, the authors show that several regulators either promote or block E35a inclusion. Some of these regulators are more active in excitatory neurons, others in inhibitory neurons, and their combined action yields the characteristic pattern: high inclusion in many inhibitory cells, low inclusion in most excitatory cells. The inclusion level can also shift with neuronal activity, indicating that this tiny segment is part of an activity-responsive program that adjusts protein variants as circuits mature and fire.
Removing the microexon changes firing and calcium signals
To probe what E35a actually does, the team engineered mice in which this microexon is completely deleted, forcing all ankyrin-G molecules into the "short" form. These animals grow and appear generally normal, but their inhibitory neurons show striking changes. In brain slices, fast-spiking interneurons in the frontal cortex fire more readily and at higher rates, even though the basic architecture of their axon initial segment remains intact. In awake mice, two-photon imaging reveals that inhibitory neurons have roughly double the somatic calcium activity compared with controls, while nearby excitatory neurons show only minor changes. This indicates that the microexon fine-tunes how certain inhibitory cells integrate inputs and control their internal calcium levels.
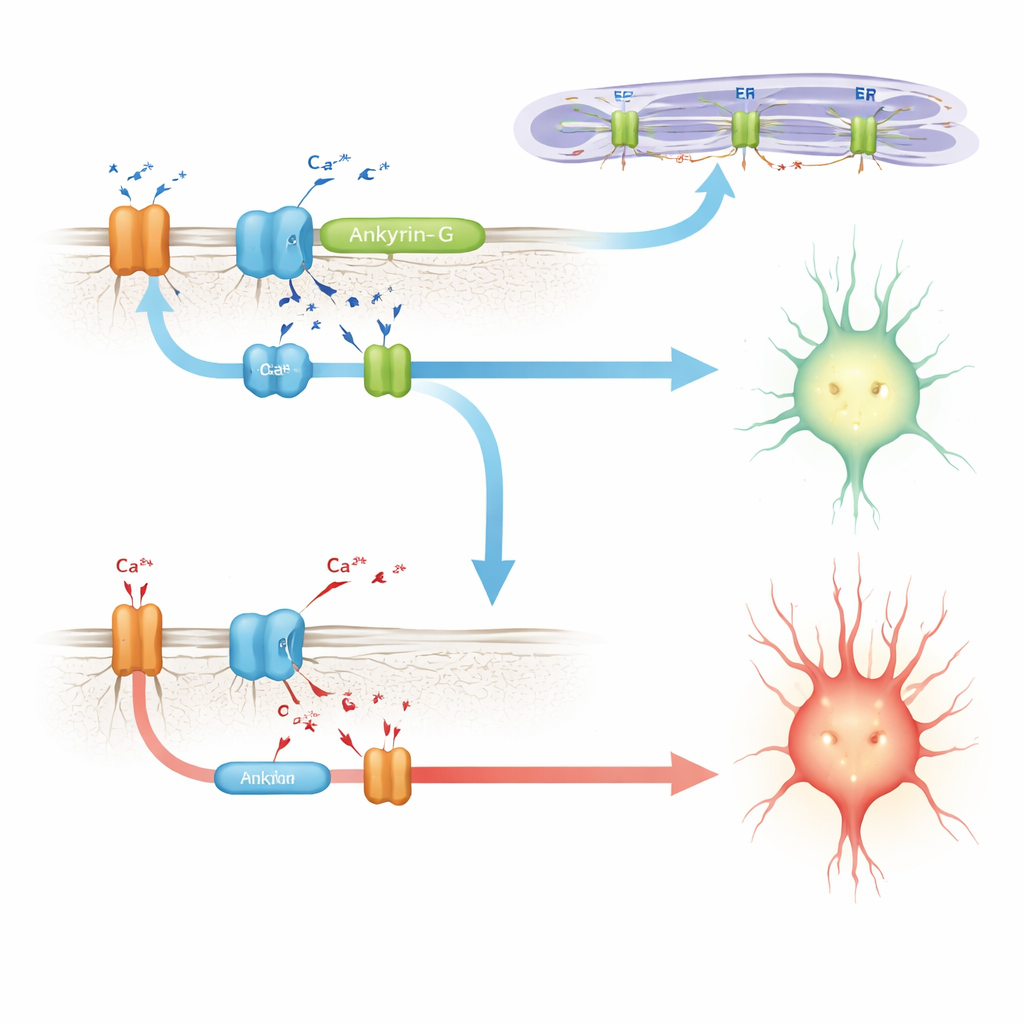
Connecting ankyrin-G to calcium-handling machinery
Seeking a mechanism, the authors turn to earlier work in heart cells, where a related protein, ankyrin-B, helps assemble a cluster of calcium-handling components: the sodium–potassium pump, the sodium–calcium exchanger, and calcium-release channels on internal membranes. A disease mutation in that region disrupts the complex and alters cardiac calcium rhythms. Here, the scientists show that including E35a in ankyrin-G strengthens its interaction with a similar set of partners in the brain, including an inositol trisphosphate receptor and the sodium–potassium pump. In mice lacking E35a, these interactions are markedly weaker. In cultured cells, a version of ankyrin-G that contains the E35a-encoded peptide pulls down much more of the calcium-release receptor than a version lacking it, supporting the idea that this microexon helps create or stabilize calcium-regulating microdomains in neurons.
From molecular fine-tuning to behavior
Despite clear changes at the cellular level, the behavioral impact in the mutant mice is subtle. They do not show major deficits in coordination, anxiety-like behavior, memory in simple tests, or social interaction. However, gait analysis and activity measures suggest mildly faster, more energetic movement. The authors propose that broader brain networks may compensate for the altered excitability of inhibitory neurons, masking more dramatic behavioral effects in the assays used. Even so, the findings illustrate how a small, neuron-type-specific exon can add an extra layer of control on top of ankyrin-G’s general structural role, adjusting how inhibition and calcium dynamics are tuned in particular circuits.
Why this matters for understanding the brain
For non-specialists, the key message is that the brain’s diversity is not only about which genes are present, but also about how flexibly those genes are edited into slightly different versions in different cells. This study shows that a nine–amino acid insert in ankyrin-G acts as a precision dial for the excitability and calcium signaling of specific inhibitory neurons, without disturbing the core architecture of their signal-launching region. Because the Ank3 gene is linked to psychiatric conditions such as bipolar disorder, these results offer a mechanistic glimpse of how minute changes in RNA splicing could shift circuit balance and influence vulnerability to disease, while also highlighting the evolutionary reuse of a calcium-control strategy seen previously in heart muscle.
Citation: Alam, S., Dermentzaki, G., Cabrera-Garcia, D. et al. A neuron type-specific microexon in Ank3/ankyrin-G modulates calcium activity and neuronal excitability. Nat Commun 17, 3173 (2026). https://doi.org/10.1038/s41467-026-69486-x
Keywords: alternative splicing, ankyrin-G, GABAergic interneurons, calcium signaling, neuronal excitability