Clear Sky Science · en
TGM2-mediated serotonylation of GPX4 confers ferroptosis resistance to promote gastric tumorigenesis
How Nerve Chemicals Can Help Stomach Tumors Survive
Most people know serotonin as the “feel-good” chemical that affects mood, but this same molecule also circulates in the gut and blood. This study reveals that in patients with gastric (stomach) cancer, serotonin does far more than send signals between nerve cells. It can directly alter key proteins inside tumor cells, helping them resist a form of cell death and grow more aggressively. Understanding this hidden chemistry between the nervous system and cancer cells may open new ways to diagnose and treat stomach cancer.
A Hidden Link Between Gut Chemistry and Stomach Cancer
The researchers began by measuring hundreds of small molecules in blood samples from people with and without gastric cancer. They found that serotonin levels were consistently higher in patients with cancer. When they added serotonin to gastric cancer cells grown in the lab, the cells multiplied faster, and tumors grew more quickly in mice given extra serotonin. Blocking a particular enzyme called TGM2 erased this tumor-boosting effect, suggesting that serotonin’s influence did not depend only on its usual surface receptors, but on a more direct chemical modification inside cells.
Serotonin as a Protein-Tagging Chemical
Serotonin can be covalently attached to proteins in a process known as “serotonylation,” catalyzed by TGM2. To see which proteins were tagged this way in gastric cancer cells, the team used a specially designed serotonin-like probe that clicks onto proteins and can be fished out and identified by mass spectrometry. This chemoproteomic screen uncovered more than 800 candidate proteins, many involved in metabolism, DNA repair, and a type of iron-driven cell death called ferroptosis. Among these, one enzyme stood out: GPX4, a master defender that protects cell membranes from being destroyed by oxidative damage.
How Tumor Cells Block a Self-Destruct Pathway
Ferroptosis is a built-in self-destruct program that kills cells when fat molecules in their membranes become heavily oxidized. GPX4 normally keeps this in check; when GPX4 is lost or disabled, ferroptosis is triggered and cancer cells can die. The study shows that TGM2 chemically attaches serotonin to GPX4 at two precise positions. This serotonylation makes GPX4 more stable, shielding it from being tagged with another molecule, ubiquitin, that would send it to the cell’s protein shredder. Without serotonylation, GPX4 is more easily recognized by an enzyme called TRIM25 that marks it for destruction. With serotonylation, GPX4 persists, membranes stay protected, and ferroptosis is held at bay.
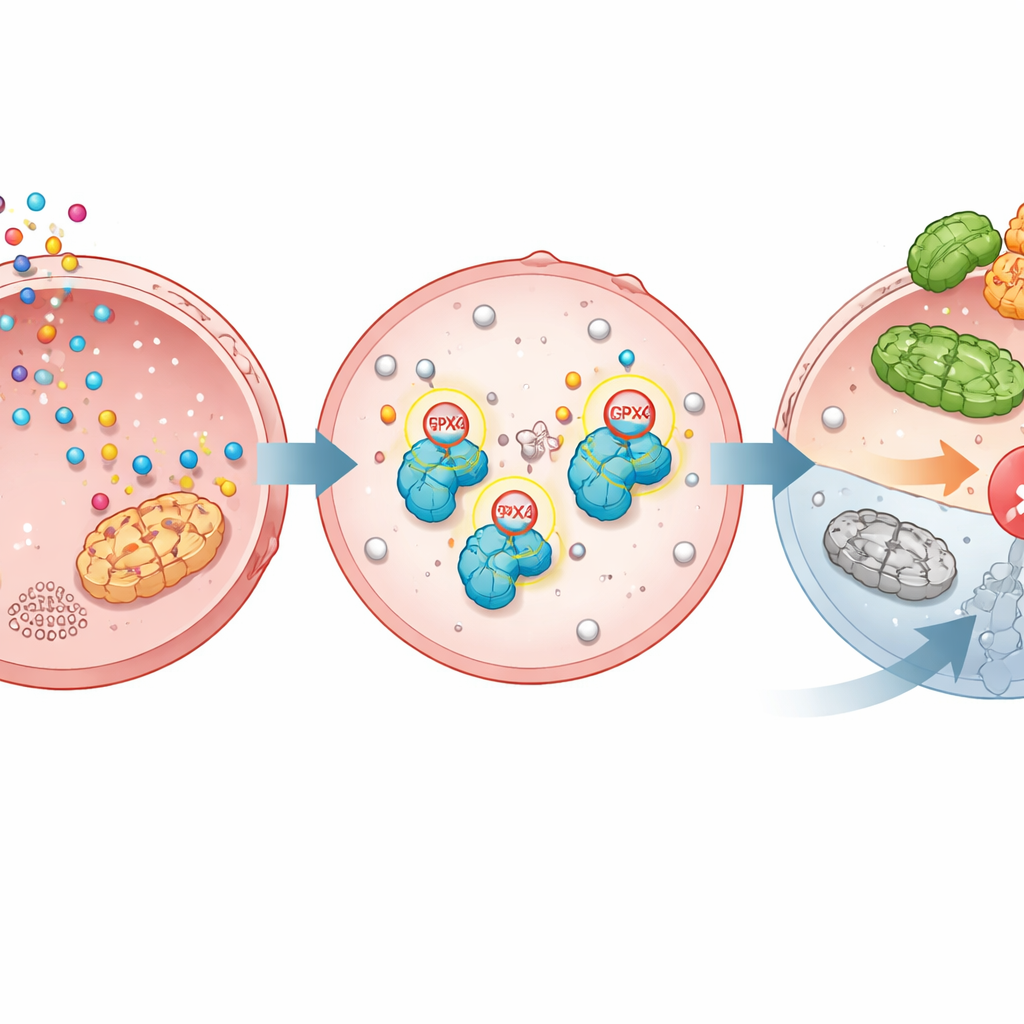
Proof from Engineered Cells, Mice, and Patient Samples
To test how crucial this modification is, the scientists engineered gastric cancer cells in which GPX4 was removed and then replaced either with the normal version or with a mutant form that cannot be serotonylated at the two key positions. Serotonin boosted growth and protected cells from ferroptosis-inducing drugs only when normal GPX4 was present, not when the non-modifiable version was used. In mouse tumor models, giving serotonin sped up growth of tumors carrying normal GPX4, but had little effect on tumors with the mutant enzyme. In patient data sets and tissue samples, the enzyme TGM2 and GPX4 were both elevated in tumors compared with nearby normal stomach tissue, and higher levels of either were linked to poorer survival, underscoring the clinical importance of this pathway.
What This Means for Patients and Future Therapies
In accessible terms, this work shows that stomach tumors can hijack a nerve chemical usually associated with mood to armor themselves against a powerful death program. By attaching serotonin directly to GPX4, the tumor uses a chemical “lock” that keeps this protective enzyme from being broken down, allowing cancer cells to survive conditions that should kill them. This discovery suggests that measuring serotonin-driven GPX4 modification could help identify high-risk gastric cancers and that drugs targeting TGM2, serotonin transport into cells, or GPX4 stability might restore ferroptosis and make tumors more vulnerable. In short, disrupting this serotonin–GPX4 safety shield could become a new strategy to treat stomach cancer.
Citation: Bai, J., Geng, D., Chen, X. et al. TGM2-mediated serotonylation of GPX4 confers ferroptosis resistance to promote gastric tumorigenesis. Cell Discov 12, 30 (2026). https://doi.org/10.1038/s41421-026-00885-6
Keywords: gastric cancer, serotonin, ferroptosis, GPX4, protein modification