Clear Sky Science · en
Inflammatory macrophage-derived plasminogen activator inhibitor-1 exacerbates inflammation through efferocytosis inhibition
When Cleanup Crews Go Wrong
Our bodies rely on immune “cleanup crews” to clear away dead and damaged cells after injury. This study reveals how one small protein, called PAI-1, can turn those helpful cleanup cells into drivers of lingering inflammation. By blocking the safe disposal of cellular debris, PAI-1 keeps injured tissue stuck in a state of slow healing and chronic irritation—an insight that could open new ways to treat muscle damage and other inflammatory diseases.
From Helpful Inflammation to Harmful Swelling
Inflammation is meant to be temporary. When tissue is injured, immune cells rush in, remove debris, and then help rebuild healthy structure. A key player in this process is the macrophage, a versatile cell that can both spark inflammation and help resolve it. Macrophages “eat” dying cells in a process known as efferocytosis, guided by surface signals that mark which cells should be removed and which should be left alone. If this clearance step fails, dead cells and their contents pile up, bathing tissues in danger signals that can fuel long-lasting inflammation linked to heart disease, diabetes, autoimmune illness, and even cancer.
A Closer Look at Muscle Injury
To understand how PAI-1 fits into this story, the researchers used a well-established mouse model of skeletal muscle injury. A toxin was injected into leg muscles to cause controlled damage followed by synchronized repair. Early after injury, a wave of inflammatory macrophages carrying markers called CCR2 and Ly6C flooded into the muscle. These particular macrophages turned out to be prolific producers of PAI-1 right at the injury site. When the PAI-1 gene was knocked out in mice, or when PAI-1’s activity was blocked with a small-molecule drug, damaged muscle fibers cleared faster, inflammatory signals dropped, and the normal honeycomb-like architecture of muscle was restored more quickly. These benefits appeared even though other sources of PAI-1 in the body were still present, pointing to a strong local effect.
How a Signal Jammer Blocks Cellular Cleanup
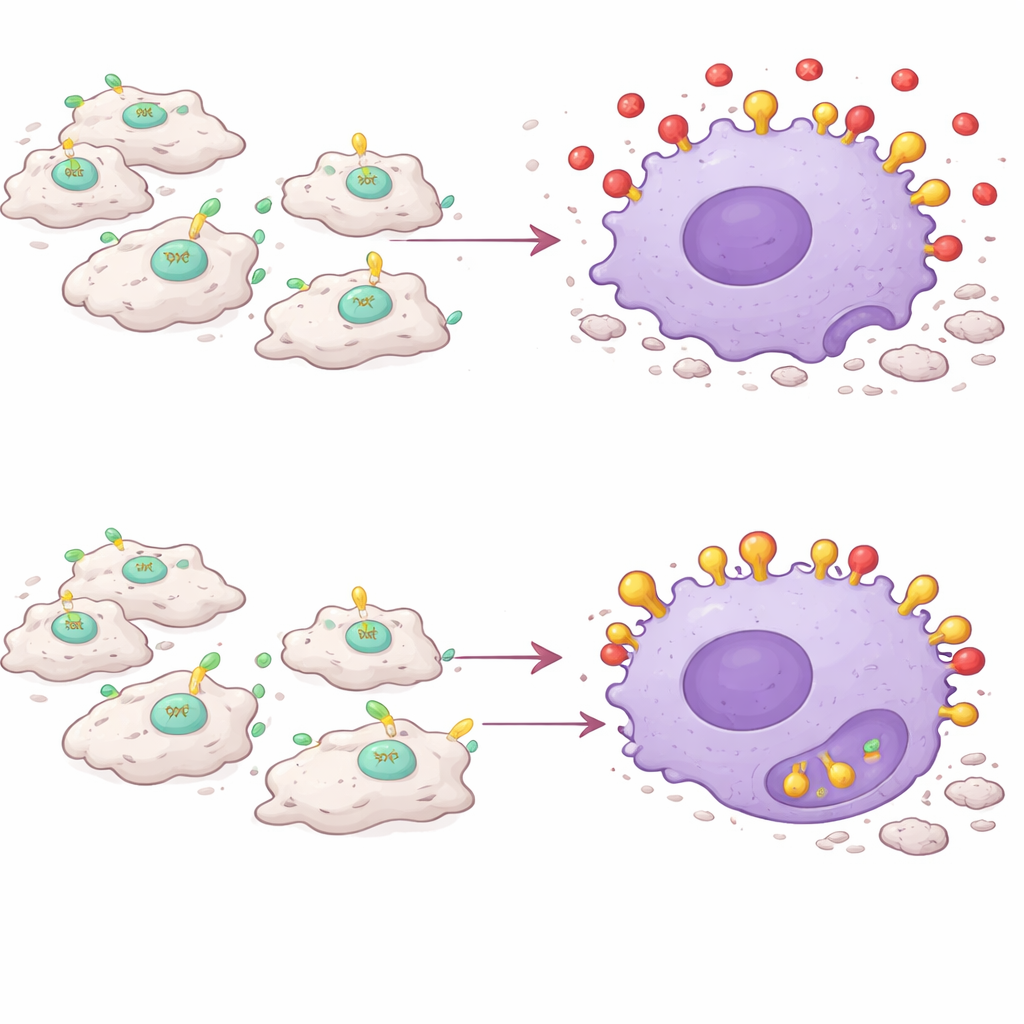
The key discovery is that PAI-1 acts as a kind of signal jammer for efferocytosis. Dying cells expose a molecule called calreticulin on their surface, which serves as an “eat me” flag. Macrophages carry a receptor called LRP-1 that recognizes calreticulin and triggers engulfment of the dying cell. The team found that PAI-1 also binds to LRP-1—and does so with higher affinity than calreticulin. In cell culture experiments, adding PAI-1 reduced the ability of macrophages to swallow dead cells, while removing PAI-1 or blocking its binding to LRP-1 boosted this cleanup. A mutant form of PAI-1 that could no longer attach to LRP-1 failed to block efferocytosis, confirming that the interference happens at this receptor. In effect, PAI-1 outcompetes the “eat me” signals, leaving dead cells stranded and uncleared.
Macrophages Turn Against Their Own Task
Strikingly, it is not just any PAI-1 that matters, but the PAI-1 made by inflammatory macrophages themselves. When the researchers selectively deleted the PAI-1 gene only in CCR2+Ly6C+ macrophages, injured muscles healed much like those in full PAI-1 knockout animals: less staining of damaged fibers, lower levels of pro-inflammatory messengers, and more regenerated muscle cells. Transplanting PAI-1–rich inflammatory macrophages into PAI-1–deficient mice was enough to restore prolonged inflammation and delayed healing, even though the rest of the body lacked PAI-1. In living animals, both genetic deletion and drug inhibition of PAI-1 increased macrophage uptake of tracer particles, indicating more active engulfment behavior during inflammation.
Opening a New Path for Treatment
By showing that macrophage-derived PAI-1 blocks the clearance of dead cells, this study links a well-known clotting regulator to the failure of inflammation to shut itself off. When PAI-1 crowds out the normal “eat me” signals at macrophage receptors, debris lingers and tissue repair stalls. A drug that inhibits PAI-1, given by mouth, restored cleanup and sped muscle regeneration in mice, hinting at a practical way to help tissues recover after injury. Because poor efferocytosis and high PAI-1 levels are common features of many chronic inflammatory and age-related conditions, targeting this pathway may one day offer a new class of anti-inflammatory therapies that work not by simply dampening immune responses, but by helping the body finish the job of healing.
Citation: Ibrahim, A.A., Miura, H., Terada, T. et al. Inflammatory macrophage-derived plasminogen activator inhibitor-1 exacerbates inflammation through efferocytosis inhibition. Cell Death Discov. 12, 195 (2026). https://doi.org/10.1038/s41420-026-03076-0
Keywords: inflammation, macrophages, muscle repair, cell clearance, PAI-1 inhibitor