Clear Sky Science · en
A novel PAAoptosis-inducing ERRα-targeting compound for combating hematopoietic and solid cancers
Why this new cancer study matters
Cancer drugs usually try to shut down a single weak spot in tumor cells, but cancers often adapt and become resistant. This study describes a new experimental drug, called PAMT‑001, that attacks cancer cells in a very different way: by overwhelming their energy factories and triggering several kinds of cell death at once. The work suggests a fresh strategy for tackling both blood cancers and solid tumors, including forms that no longer respond well to standard chemotherapy.
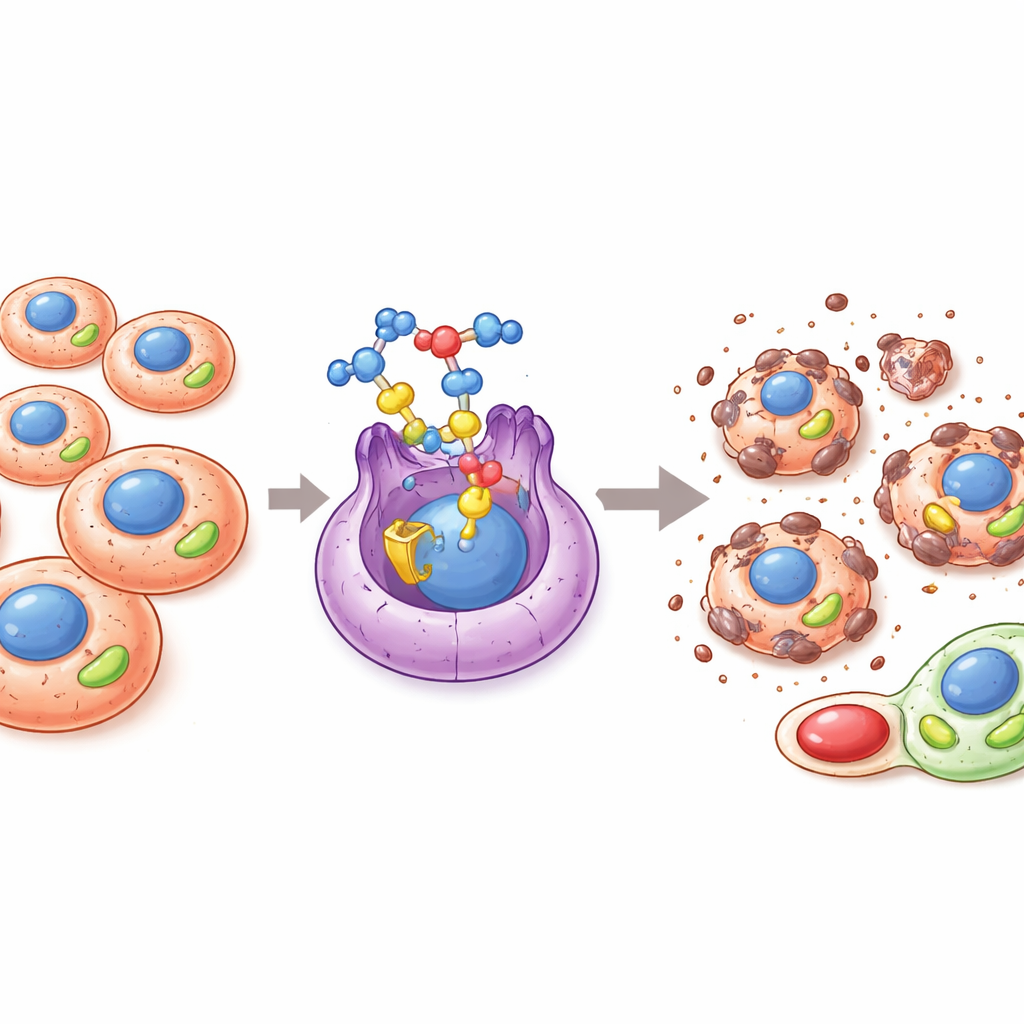
A hidden switch that fuels cancer growth
Many cancers rewire how they make and use energy in order to grow, spread, and resist treatment. A key player in this metabolic reshaping is a protein known as ERRα, a type of molecular switch inside the cell nucleus that controls genes involved in mitochondrial energy production and redox balance. When ERRα is overly active, tumors can boost their mitochondrial output, mop up harmful oxygen by‑products, maintain stem‑like cancer cells, and better survive harsh conditions such as low oxygen or drug exposure. Because of this, ERRα has attracted attention as a promising target for new cancer therapies that aim to cut off the tumor’s energy advantages.
Designing a smarter cancer blocker
The researchers started from an older ERRα‑blocking molecule, XCT‑790, and chemically redesigned it to create a family of related compounds. Out of twelve candidates, PAMT‑001 emerged as the most powerful at killing cancer cells, even though it actually binds ERRα somewhat less strongly than XCT‑790 in lab tests. Computer modeling and reporter assays showed that PAMT‑001 can nestle into the same region of ERRα that normally interacts with a co‑activator protein called PGC1α, which turbocharges ERRα’s control over mitochondrial genes. By interfering with this partnership, PAMT‑001 more effectively shuts down ERRα‑driven metabolic programs, especially in cells that lean heavily on this axis for survival.
Starving the cell’s power plants
Once inside leukemia and solid tumor cells, PAMT‑001 strongly disrupts mitochondrial function. Gene‑expression analyses revealed broad suppression of oxidative phosphorylation genes, and live measurements of oxygen consumption showed a marked drop in basic and peak respiration as well as ATP production. Under the microscope, mitochondria in treated cells became swollen and misshapen, with cristae—inner folds where energy reactions occur—widened or lost. This damage leads to excess production of reactive oxygen species, which are chemically aggressive molecules that strain both mitochondria and the neighboring endoplasmic reticulum, the cell’s protein‑folding hub. Antioxidants partially rescued cell survival, confirming that this oxidative burst is a key driver of the drug’s effects.
Three death routes instead of one
Rather than relying on a single kill switch, PAMT‑001 activates several regulated death programs in parallel. It triggers classic intrinsic apoptosis: mitochondria release cytochrome c, executioner enzymes called caspases are cut and activated, and cell membranes show characteristic blebbing; pan‑caspase inhibitors substantially blunt this effect. At the same time, the drug induces strong autophagy, in which double‑membrane sacs form and engulf damaged mitochondria and other cell contents. Blocking this process with autophagy inhibitors or by disabling a key autophagy gene reduces cell death and lowers stress signals, indicating that, in this context, autophagy becomes overactivated and harmful rather than protective. Finally, PAMT‑001 drives a fiery form of death called pyroptosis: pores open in the cell membrane, contents leak out, and a protein named gasdermin E is cleaved into a pore‑forming fragment. Together, pyroptosis, apoptosis, and autophagic cell death combine into what the authors call “PAAoptosis,” a coordinated, multi‑pathway collapse of the cancer cell.

Promise for hard‑to‑treat cancers
PAMT‑001 did not just work in dishes. In mouse models of colon cancer and acute myeloid leukemia, the drug slowed tumor growth, reduced leukemia cells in bone marrow, and lowered overall cancer burden as measured by imaging, all without obvious harm to body weight or normal cells. Samples from patients with acute myeloid leukemia were generally more sensitive to PAMT‑001 than healthy blood cells, and leukemias carrying mutations in a gene called NPM1—often associated with high mitochondrial activity—were especially vulnerable. The drug also killed leukemia cells resistant to cytarabine and lung cancer cells resistant to an EGFR‑targeted drug, hinting that its broad attack on mitochondrial metabolism and multi‑pathway cell death could help overcome treatment resistance.
What this means for future cancer care
This work introduces PAMT‑001 as a prototype of a new kind of cancer drug that targets an energy‑controlling switch and forces tumors into PAAoptosis, a blend of three destructive death programs. By undermining the power supply and stressing several vital systems at once, such drugs may leave cancer cells less room to evolve resistance. Although PAMT‑001 itself will need more testing for safety, dosing, and long‑term effects, the study points toward therapies that exploit the unique energy demands of cancer cells and use multi‑route cell death as a way to keep stubborn tumors from bouncing back.
Citation: Seo, W., Heo, Y., Tran, K.V. et al. A novel PAAoptosis-inducing ERRα-targeting compound for combating hematopoietic and solid cancers. Cell Death Discov. 12, 188 (2026). https://doi.org/10.1038/s41420-026-03010-4
Keywords: cancer metabolism, mitochondria, ERR alpha, drug resistance, cell death pathways