Clear Sky Science · en
PMEPA1 modulates YAP1 nuclear translocation to disrupt EMT subtypes and promote metastasis in Biliary tract cancer
Why this research matters
Biliary tract cancer is a rare but deadly cancer of the bile ducts and gallbladder, with few effective treatment options and a high chance of spreading to distant organs. This study uses cutting edge single cell analysis to uncover how certain tumor cells change their identity to become more mobile and invasive, and it pinpoints a specific molecule that helps drive this switch and could be targeted by an existing chemotherapy drug.
Looking closely at bile duct tumors one cell at a time
Instead of averaging signals across whole tumors, the researchers examined nearly 160,000 individual cells from 47 human biliary tract cancer and normal tissue samples. By reading the RNA of each cell, they built a detailed map of the tumor ecosystem, including immune cells, support cells, blood vessel cells, and especially the tumor epithelial cells that give rise to the cancer. They found that tumor tissues contained many more epithelial cells than normal tissue and that these epithelial cells could be divided into five distinct groups, each with its own job, such as rapid growth, altered energy use, inflammation, or fat processing.
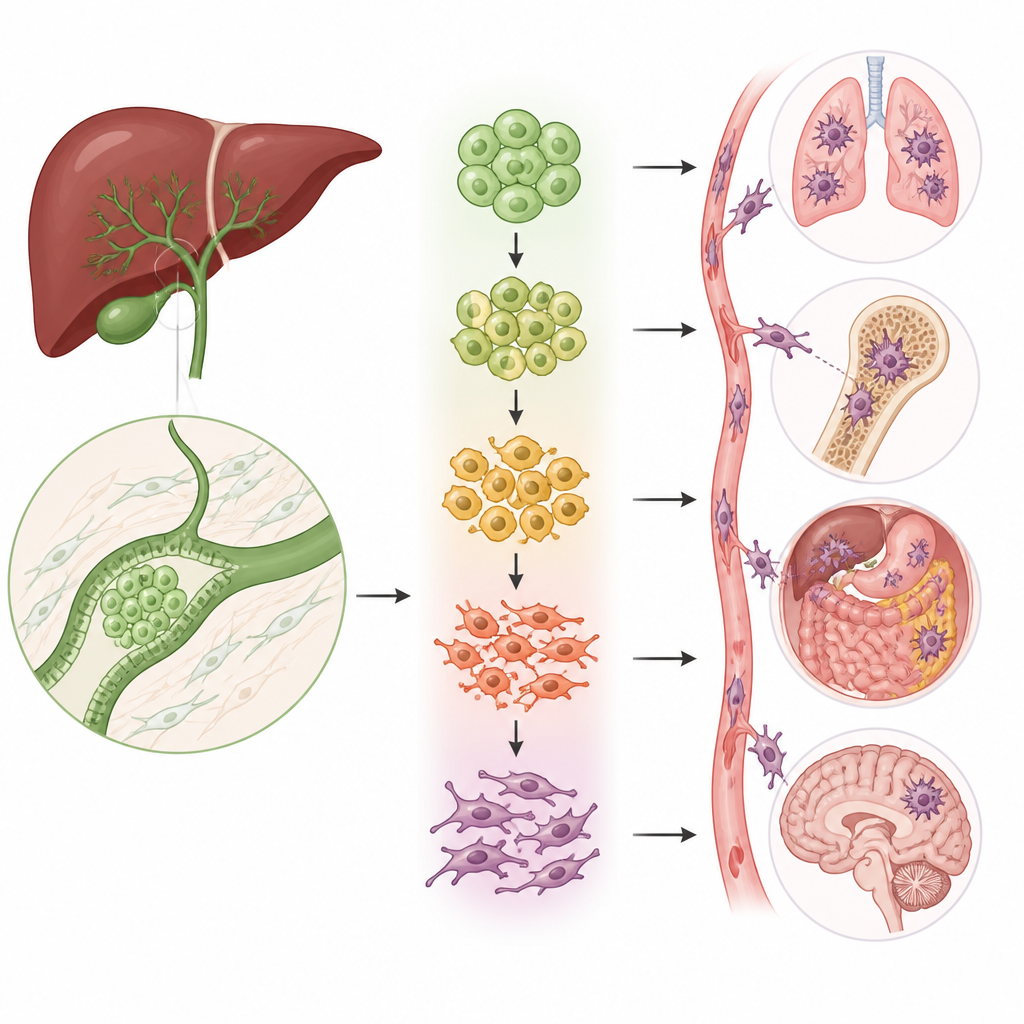
The shape shifting cells that power spread
A key process in cancer spread is called epithelial mesenchymal transition, in which compact, orderly cells loosen their bonds, change shape, and start to move. The team discovered that two of the five epithelial cell groups were strongly enriched for this transition program. Using several types of analysis, they showed that cells in these groups sit along a path from normal like, tightly connected cells toward highly mobile, invasive cells. As cells progress along this path, classic markers of stationary cells fade while markers of mobile, fibrous cells rise, linking these subgroups directly to the capacity of the tumor to invade and metastasize.
PMEPA1 as a central switch in aggressive behavior
To understand which genes actually drive this change, the scientists assembled a set of 51 transition related genes specific to biliary tract cancer and used them to score both single cells and patient tumors. Patients whose tumors had higher scores had worse survival, tying the cell level program to clinical outcome. Among the genes enriched in transition positive cells, one called PMEPA1 stood out. High PMEPA1 levels were linked to poorer prognosis and were higher in lymph node metastases than in the original bile duct tumors. When the researchers reduced PMEPA1 in bile duct cancer cell lines, the cells grew more slowly, formed fewer colonies, and lost much of their ability to move and invade through artificial membranes and to seed tumors in the lungs of mice. Boosting PMEPA1 had the opposite effect.
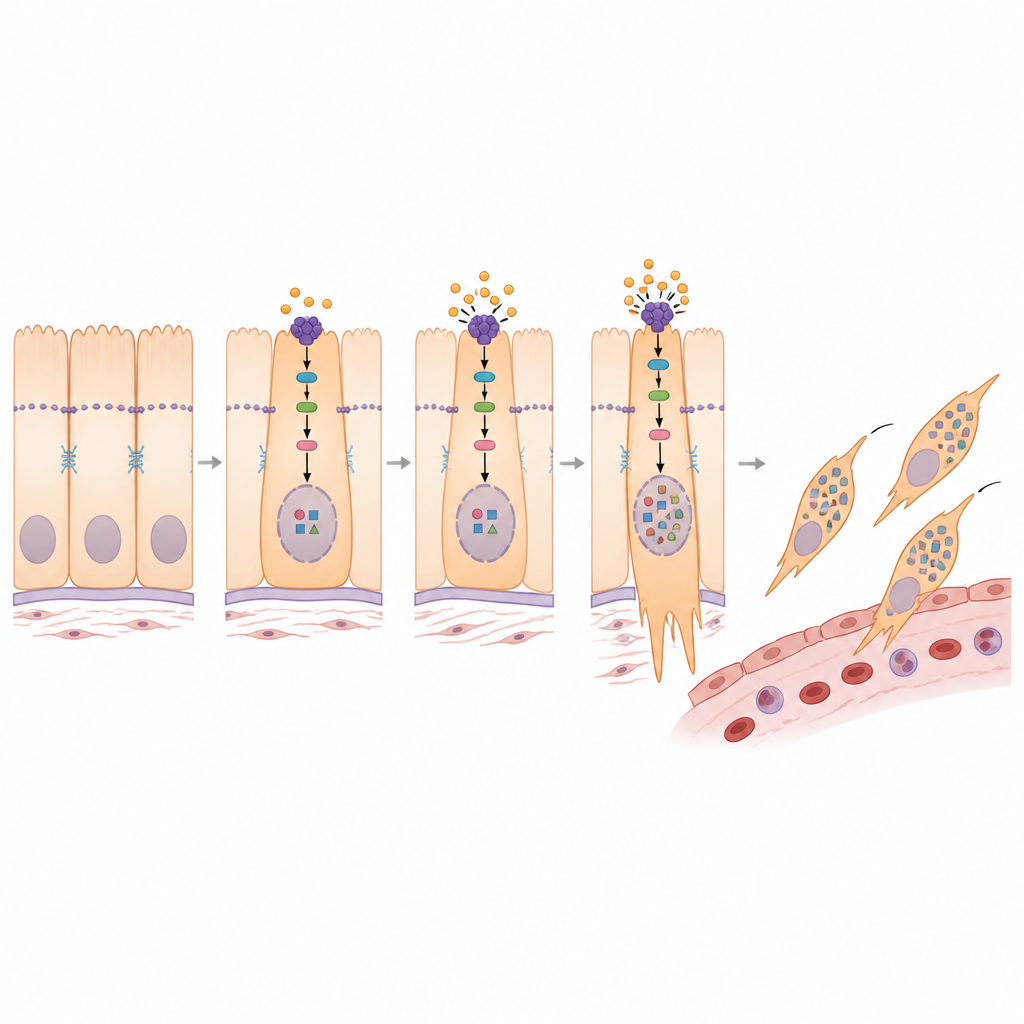
How PMEPA1 rewires signals inside the cell
Digging deeper, the study connected PMEPA1 to a known growth control pathway centered on a protein called YAP1. In healthy tissues, a safety circuit keeps YAP1 mostly outside the cell nucleus, limiting its ability to turn on growth and invasion genes. The researchers found that PMEPA1 helps to weaken this safety circuit, allowing more YAP1 to enter the nucleus. When PMEPA1 was silenced, upstream enzymes became more active, YAP1 was more heavily modified, and it remained trapped in the cell fluid instead of the nucleus. This shift was accompanied by a more stationary, less invasive profile. A chemical that blocks the safety enzymes reversed many of the changes caused by PMEPA1 loss, confirming that PMEPA1 promotes the transition program by steering YAP1 into the nucleus.
A potential drug to slow spread
Because the transition program is closely tied to treatment resistance, the team next asked whether any existing drugs might selectively hit these aggressive cells. By combining drug response databases with their gene signatures, they flagged several chemotherapy agents, including SN 38, the active form of irinotecan, as candidates. In cell experiments, SN 38 reduced markers of the mobile cell state, shrank the cells’ ability to migrate and invade, and lowered both PMEPA1 and YAP1 levels. Further work suggested that SN 38 interferes with a transcription factor called FOS that normally boosts PMEPA1 production. In mouse models of lung metastasis driven by PMEPA1 overexpression, SN 38 treatment reduced metastatic burden and PMEPA1 levels in tumor deposits.
What this means for patients
In plain terms, this study shows that a specific protein, PMEPA1, acts like a master switch that helps bile duct cancer cells loosen up, move away, and colonize new organs. It does so by freeing another protein, YAP1, to enter the nucleus and activate genes that favor invasion. Because tumors with stronger versions of this program fare worse, PMEPA1 and its partners could serve as warning markers and treatment targets. The finding that SN 38, already used in other cancers, can dampen this switch and slow metastasis in models raises the possibility that refining or combining such therapies might one day offer more precise and effective options for people with biliary tract cancer.
Citation: Xu, W., Ma, C., Li, P. et al. PMEPA1 modulates YAP1 nuclear translocation to disrupt EMT subtypes and promote metastasis in Biliary tract cancer. Cell Death Dis 17, 449 (2026). https://doi.org/10.1038/s41419-026-08684-3
Keywords: biliary tract cancer, epithelial mesenchymal transition, PMEPA1, YAP1 signaling, SN-38