Clear Sky Science · en
Ndfip2 in TrkA-expressing sensory neurons regulates noxious mechanosensation through control of TrkA signaling and protein levels
Why pain needs careful fine-tuning
Pain may feel like a nuisance, but it is one of the body’s most important warning systems. When this alarm misfires—either too loud in chronic pain, or too quiet in people who hardly feel injuries—the results can be devastating. This study uncovers a previously unknown molecule, called Ndfip2, that helps set the sensitivity of certain pain-sensing nerve cells. By learning how this internal dial works, researchers hope to point the way toward new treatments that ease pain without shutting down vital defenses.
Key players in the pain alarm
Pain-detecting nerve cells, known as nociceptors, sit in clusters beside the spinal cord and send messages to the brain when they encounter harmful stimuli such as a pinprick or extreme heat. Many of these cells depend on a growth signal called nerve growth factor (NGF) and its partner on the cell surface, a receptor called TrkA. When NGF binds to TrkA, it launches a cascade of signals that help these neurons develop and later determine how strongly they respond to painful stimuli. Too much TrkA activity can amplify pain, while too little can blunt it. Because blocking NGF directly has caused side effects in clinical trials, scientists are now searching for safer ways to tune this pathway from within the cell.
A hidden helper inside sensory neurons
The researchers focused on Ndfip2, a protein previously known to assist enzymes that tag other proteins for degradation. They first confirmed that Ndfip2 is present in adult pain-sensing neurons in the dorsal root ganglia and is especially enriched in cells that carry TrkA. Within these neurons, Ndfip2 clusters in the cell’s internal shipping centers—the endoplasmic reticulum and the Golgi apparatus—rather than at the cell surface. There it physically associates with TrkA itself. Intriguingly, male mice showed higher baseline levels of Ndfip2 in these neurons than females, hinting that this molecule might contribute to known sex differences in pain perception.
Turning down mechanical pain without numbing everything
To probe Ndfip2’s role in living animals, the team engineered mice in which Ndfip2 was removed only from cells that express TrkA. These animals grew normally and showed no differences in movement or anxiety-like behavior compared with their littermates. But when their paws were tested with calibrated mechanical pressure, both male and female mutant mice needed stronger force before withdrawing, revealing a selective reduction in sensitivity to noxious mechanical stimuli. In contrast, responses to heat and most cold tests were unchanged, and inflammatory pain behavior was only modestly heightened in females during the earliest minutes of a formalin injection. This pattern suggests that Ndfip2 fine-tunes a specific channel of pain—harmful pressure—rather than broadly dulling all forms of sensation.
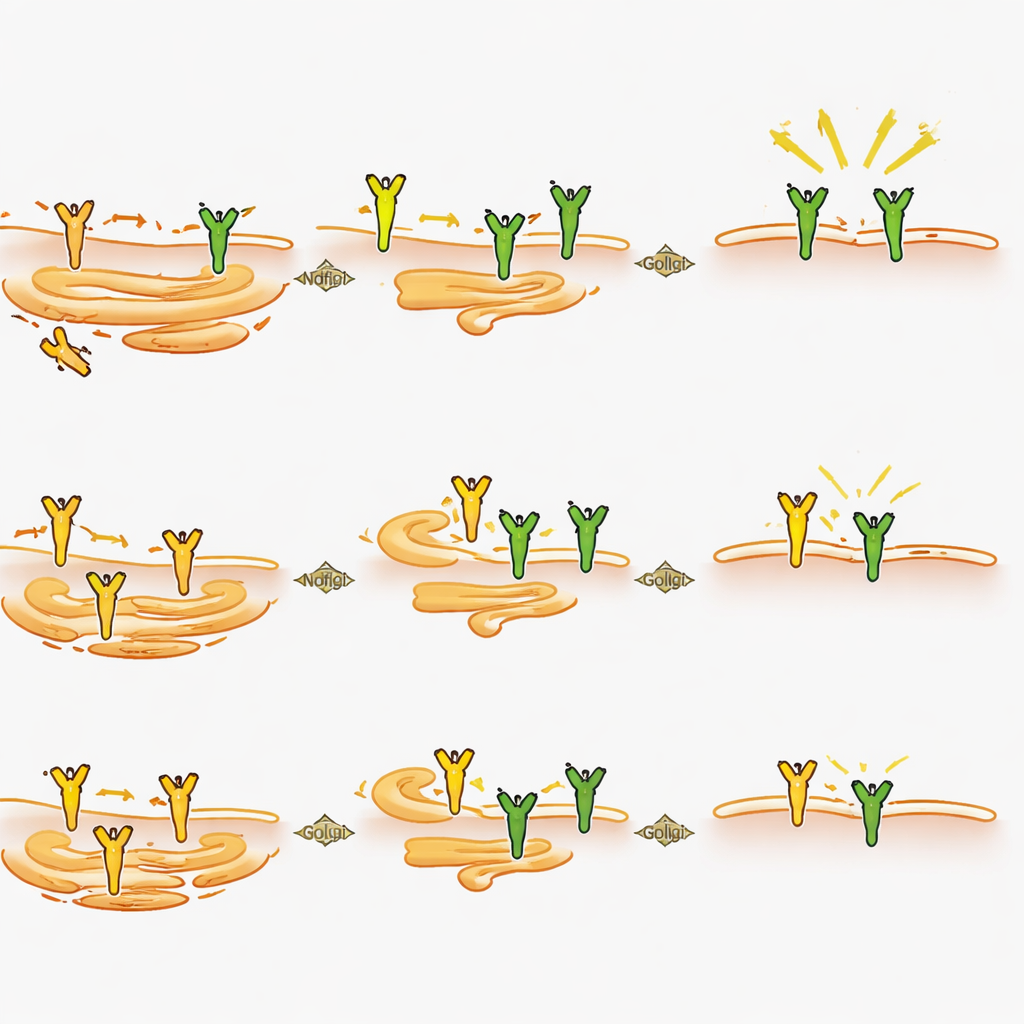
How Ndfip2 shapes the pain signal from inside the cell
Looking closer at the neurons, the scientists found that deleting Ndfip2 lowered the overall amount of TrkA protein in adult dorsal root ganglia, particularly in males, even though the gene for TrkA was still expressed at normal levels. In cultured neurons, loss of Ndfip2 reduced both TrkA levels and the strength of NGF-triggered signaling, again more markedly in males. Surprisingly, this effect did not stem from changes in the usual tagging of TrkA for degradation. Instead, experiments in cells revealed that extra Ndfip2 causes TrkA to accumulate in an immature, non-glycosylated form while depleting the fully processed versions that normally reach the cell surface. Consistent with this, knocking down Ndfip2 in neurons increased the fraction of TrkA present at the membrane in males. Together, these findings point to Ndfip2 as a regulator of how TrkA is chemically matured and routed inside the cell, thereby controlling how much functional receptor is available to sense NGF.
What this means for future pain therapies
By uncovering Ndfip2 as an internal controller of the NGF–TrkA system, this work adds a new layer of understanding to how the body calibrates pain sensitivity, especially for harmful mechanical stimuli. Rather than blocking NGF outright, influencing Ndfip2 or related steps in TrkA maturation might allow clinicians to subtly reduce excessive signaling in specific pain pathways while preserving the protective function of pain overall. The observed sex differences in Ndfip2 levels and effects also underscore the importance of tailoring future pain treatments to biological context rather than assuming one-size-fits-all solutions.
Citation: Cañada-García, D., Hernández-García, A., Vicente-García, C. et al. Ndfip2 in TrkA-expressing sensory neurons regulates noxious mechanosensation through control of TrkA signaling and protein levels. Cell Death Dis 17, 437 (2026). https://doi.org/10.1038/s41419-026-08670-9
Keywords: mechanical pain, sensory neurons, nerve growth factor, TrkA receptor, post-translational regulation