Clear Sky Science · en
BAP1 dysregulation impairs trophoblast differentiation and contributes to placental dysfunction in preeclampsia
When Pregnancy’s Support Organ Goes Awry
Preeclampsia is a dangerous complication of pregnancy marked by high blood pressure and damage to organs such as the liver and kidneys. At the heart of this condition lies the placenta, the temporary organ that nourishes the baby. This study uncovers how mismanagement of a single regulatory protein, called BAP1, can derail the development of key placental cells and help drive the severe, early-onset form of preeclampsia.
The Placenta’s Delicate Balancing Act
The placenta is built from specialized cells known as trophoblasts. Early in pregnancy, stem-like trophoblasts must choose between two main jobs. Some fuse together to form a continuous surface layer that handles nutrient and gas exchange. Others transform into invasive cells that tunnel into the mother’s uterus and remodel spiral arteries, widening them to deliver ample blood to the growing baby. This careful balance between staying in a stem-like state, changing identity, and invading just far enough is essential. When the process fails, as in early-onset preeclampsia, the placenta becomes shallow, stressed, and less able to support a healthy pregnancy.
A Placental Protein Turned Up Too High
The researchers focused on BAP1, a protein better known for its role in cancer biology. In healthy placentas early in pregnancy, BAP1 is abundant in stem-like trophoblasts but normally drops as these cells mature. Using placental samples from women with and without pregnancy complications, the team found that BAP1 and its partner proteins ASXL2 and ASXL3 were specifically increased in placentas from early-onset preeclampsia, but not in the later-onset, more maternal-driven form of the disease. This pattern suggests that excessive BAP1 activity is closely tied to placental, rather than purely maternal, causes of the disorder.
Freezing Placental Cells in an Immature State
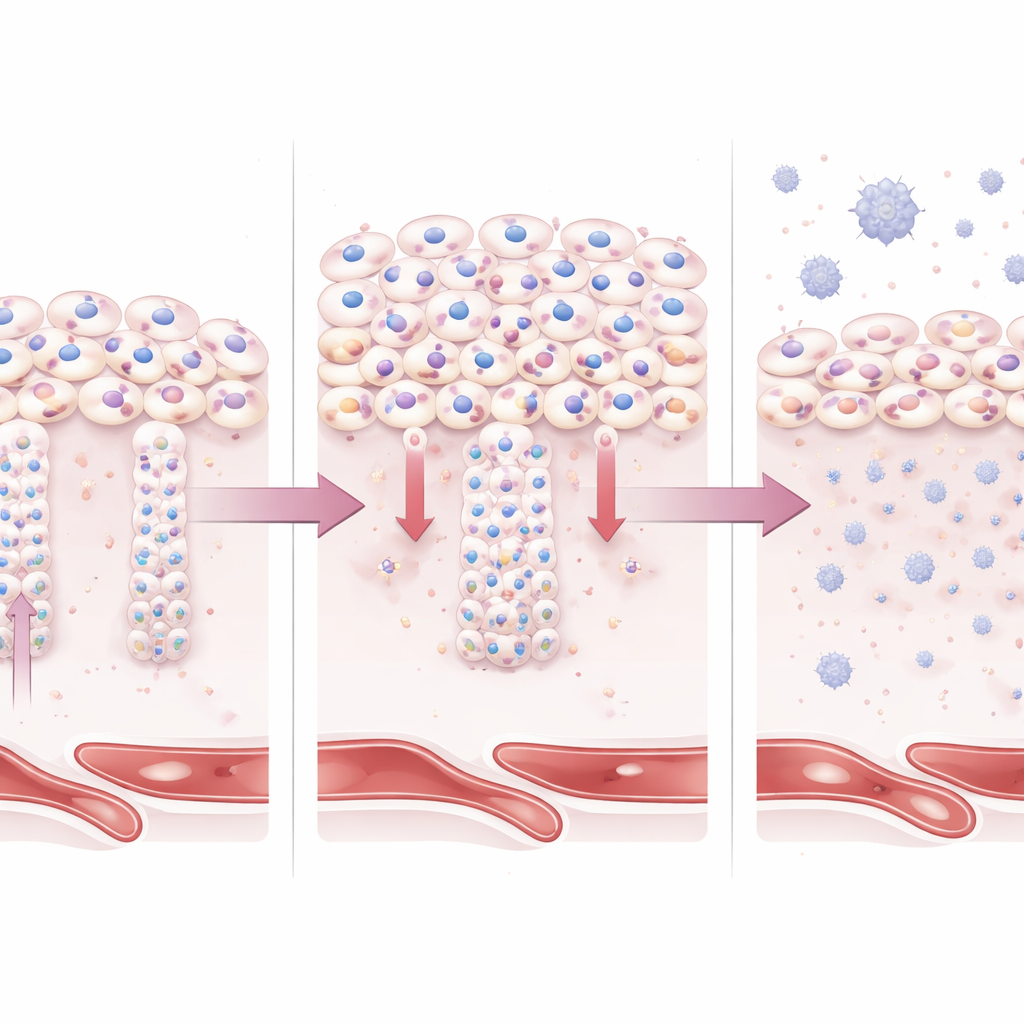
To see what BAP1 does inside human placental cells, the scientists used human trophoblast stem cells and 3D placental organoids grown in the lab. When they artificially boosted BAP1 levels, the cells clung tightly to one another and strongly maintained an epithelial, or sheet-like, identity. These cells were less able to transform into the invasive trophoblasts that normally dig into the uterus, and they produced fewer of the enzymes needed to remodel the mother’s blood vessels. Likewise, cells that should have fused into the hormone-producing surface layer instead remained as separate, unfused units. Molecular analyses showed that genes and proteins tied to cell adhesion and structural junctions were turned up, while those linked to invasion, hormone production, metabolism, and stress adaptation were dialed down.
From Faulty Cell Choices to Placental Stress
In organoid models that mimic early human placental tissue, excess BAP1 led to smaller or poorly formed regions of fused surface cells and an overrepresentation of stem-like trophoblasts. At the same time, the organoids activated innate immune and inflammatory pathways, including interferon-related signals that are also seen in early-onset preeclampsia placentas. By combining RNA sequencing and protein measurements, the researchers built a BAP1-driven molecular “signature” and showed that it strongly matched patterns found specifically in early-onset preeclampsia, but not in other pregnancy complications such as gestational diabetes or placental effects of COVID-19. This ties BAP1 overactivity to both the structural malfunction and the inflammatory atmosphere of the diseased placenta.
What This Means for Mothers and Babies
Taken together, the work paints BAP1 as a gatekeeper that must be carefully tuned during pregnancy. When BAP1 stays too high, trophoblasts fail to grow into the specialized cell types needed for deep uterine invasion and robust hormone-secreting surfaces. The result is a placenta that is shallow, stressed, and inflamed—hallmarks of early-onset preeclampsia. While more research is needed to learn whether BAP1 changes are a trigger or a response to early stress, this study provides a clear mechanistic link between misregulated cell identity programs in the placenta and a life-threatening pregnancy disorder, opening the door to new biomarkers and, eventually, targeted therapies.
Citation: Doria-Borrell, P., Ferrero-Micó, A., Navarro-Serna, S. et al. BAP1 dysregulation impairs trophoblast differentiation and contributes to placental dysfunction in preeclampsia. Cell Death Dis 17, 410 (2026). https://doi.org/10.1038/s41419-026-08650-z
Keywords: preeclampsia, placenta, trophoblast, BAP1, pregnancy complications