Clear Sky Science · en
Liver kinase B1 maintains natural killer cell survival by regulating redox homeostasis
Why Our Immune “First Responders” Need Protection Too
Natural killer (NK) cells are the body’s rapid‑response soldiers, patrolling for virus‑infected and cancerous cells before other immune forces arrive. This study asks a deceptively simple question with big implications: what keeps these powerful cells healthy and calm until they are needed, and what happens when that internal safety system fails? By uncovering how a single protein, called liver kinase B1 (Lkb1), shields NK cells from internal chemical damage, the work points to new ways to strengthen early cancer surveillance without burning out these crucial defenders.
A Guardian Switch Inside Immune Cells
NK cells must walk a fine line. They need enough energy to spring into action, but not so much that they become overactive, exhausted, or die off. The authors show that Lkb1 acts as a key internal “guardian switch” that helps NK cells stay in a quiet, ready state. Using several engineered mouse models, they selectively removed Lkb1 from NK cells at different stages of their development. No matter how they did it, the result was striking: NK cells became scarce in many organs, got stuck in immature stages, and lost the surface markers that normally signal full readiness to fight. These effects were cell‑intrinsic, meaning they arose from problems inside NK cells themselves rather than from their environment.
When Cellular Housekeeping Breaks Down
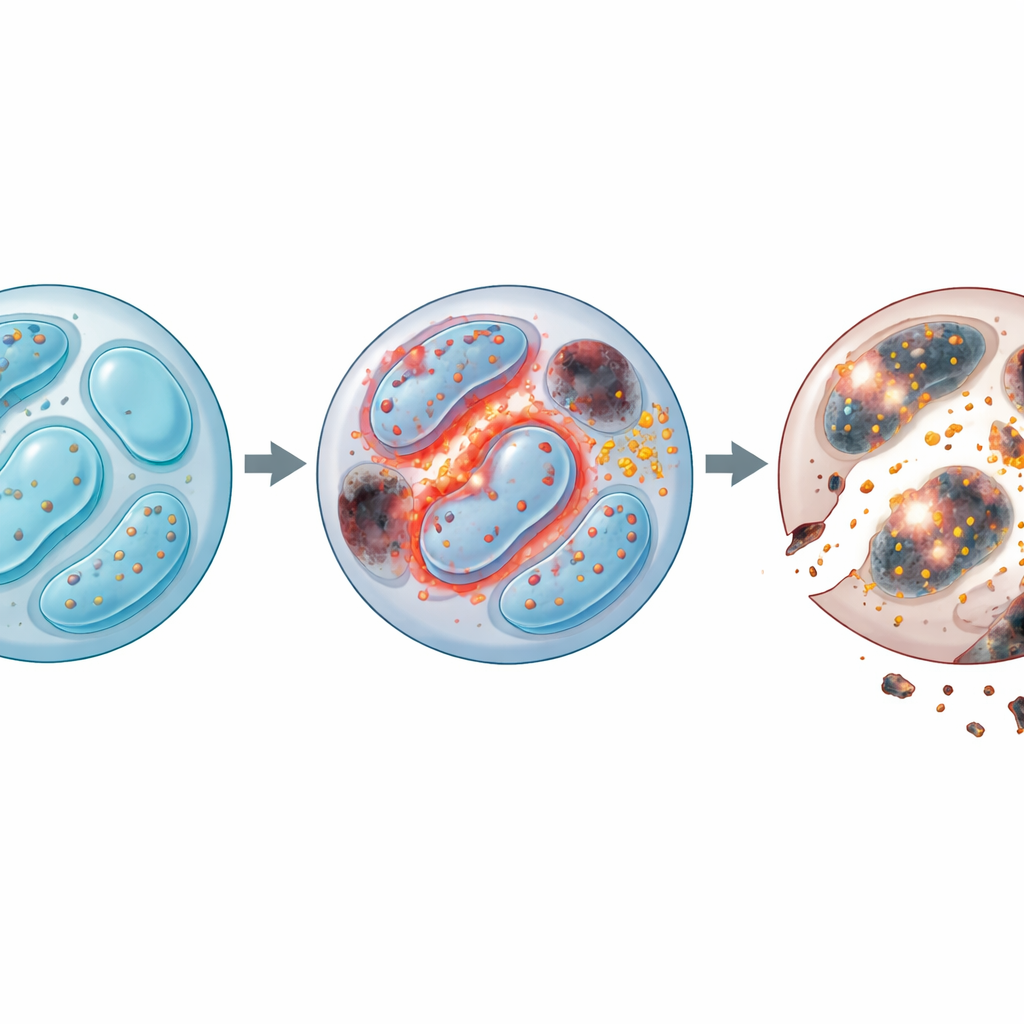
Digging deeper, the team examined which genes changed when Lkb1 was missing. Thousands of genes linked to energy use, cell division, waste recycling, and cell death were altered. In particular, NK cells without Lkb1 showed a breakdown in autophagy, the cell’s housekeeping system that clears worn‑out parts such as damaged mitochondria. Under normal conditions, this cleanup prevents harmful by‑products from piling up. Without Lkb1, autophagy stalled: cells built up extra mitochondria with abnormally high membrane potential, and these faulty structures spewed large amounts of reactive oxygen species (ROS) – unstable molecules that can damage proteins, fats, and DNA. Even though Lkb1‑deficient NK cells tried to compensate by dividing more, they died at higher rates, revealing that survival, not growth, was the core problem.
Iron Overload and a Double Hit of Cell Death
The trouble did not stop with ROS. The researchers found that NK cells lacking Lkb1 also lost control of iron balance. These cells hoarded iron and ramped up a key iron‑import receptor, which in turn fueled the formation of toxic “lipid ROS” that attack cell membranes. This pattern is characteristic of ferroptosis, an iron‑dependent form of cell death. By treating cells with an iron‑binding drug or a ferroptosis blocker, the team could rescue much of the survival defect. At the same time, classical programmed cell death pathways were also overactive: protective proteins dropped, executioner enzymes rose, and blocking caspases (the enzymes that drive apoptosis) improved viability. Combining iron removal and caspase inhibition gave an additive benefit, indicating that NK cells without Lkb1 are struck by two independent but converging death routes—iron‑driven membrane damage and mitochondrial‑driven apoptosis.
Exhausted Soldiers and Weakened Tumor Patrol
Beyond staying alive, NK cells must function effectively. Lkb1‑deficient NK cells produced less of the key defense molecule interferon‑γ and displayed higher levels of inhibitory “brakes” on their surface, such as PD‑1 and TIGIT—features associated with exhausted immune cells seen in chronic infections and cancer. Antioxidant treatments that reduced ROS or blocked ferroptosis lowered these exhaustion markers and partially restored NK cell activity. In living mice, the consequences were clear: animals lacking Lkb1 in NK cells were much worse at rejecting cells that lacked normal “self” signals and were less able to clear NK‑sensitive tumor cells or prevent the spread of melanoma to the lungs. Removing Lkb1 earlier in NK cell development caused even more severe failures, underscoring its importance from the earliest stages onward.
What This Means for Future Therapies
To many, Lkb1 is best known as a tumor‑suppressor protein in cancer cells, but this work highlights its equally critical role inside immune cells. Lkb1 keeps NK cells metabolically balanced, maintains their internal cleanup systems, prevents iron and ROS overload, and protects them from dual death pathways and exhaustion. Interestingly, common energy‑sensing routes linked to Lkb1 in other cells—AMPK activation and mTORC1 inhibition—could not fix the damage when pharmacologically tweaked, hinting that NK cells rely on distinct downstream circuits. For a lay reader, the takeaway is that NK cells themselves need protection from oxidative and iron‑driven stress to remain effective cancer sentinels. Rather than directly altering Lkb1, which might carry cancer risks elsewhere, therapies that fine‑tune redox and iron balance in NK cells could one day help bolster the body’s own early warning system against tumors.
Citation: Meng, W., Luo, L., Xiao, Z. et al. Liver kinase B1 maintains natural killer cell survival by regulating redox homeostasis. Cell Death Dis 17, 413 (2026). https://doi.org/10.1038/s41419-026-08629-w
Keywords: natural killer cells, LKB1, oxidative stress, iron homeostasis, cancer immunosurveillance