Clear Sky Science · en
Reduced YTHDF2 inhibits PD-L1 expression by stabilizing m6A-containing SPOP mRNA in colorectal cancer
Why this research matters
Colorectal cancer is one of the most common and deadly cancers worldwide. Many modern treatments try to unleash the body’s own immune system to attack tumors by blocking a brake called PD-1/PD-L1. Yet, most colorectal cancer patients still do not benefit from these drugs. This study uncovers a hidden molecular chain reaction inside cancer cells that helps them keep that immune “brake” pressed down. Understanding this chain offers a new way to make immunotherapy work for many more people.
A hidden message tag on tumor RNA
Our cells constantly read and destroy strands of RNA, the short-lived messages that tell them which proteins to make. In recent years, scientists have discovered that many RNA messages carry tiny chemical tags, including one called m6A, that act like editing marks in a manuscript: they decide how long a message lasts and how strongly it is read. A protein named YTHDF2 specializes in recognizing these m6A tags and usually helps break down tagged RNA, fine-tuning which proteins a cell produces. In colorectal cancer samples from patients and large public databases, the authors found that YTHDF2 levels were noticeably higher in tumors than in normal colon tissue, and that patients with more YTHDF2 tended to have worse survival, pointing to a pro‑cancer role for this RNA “reader.”
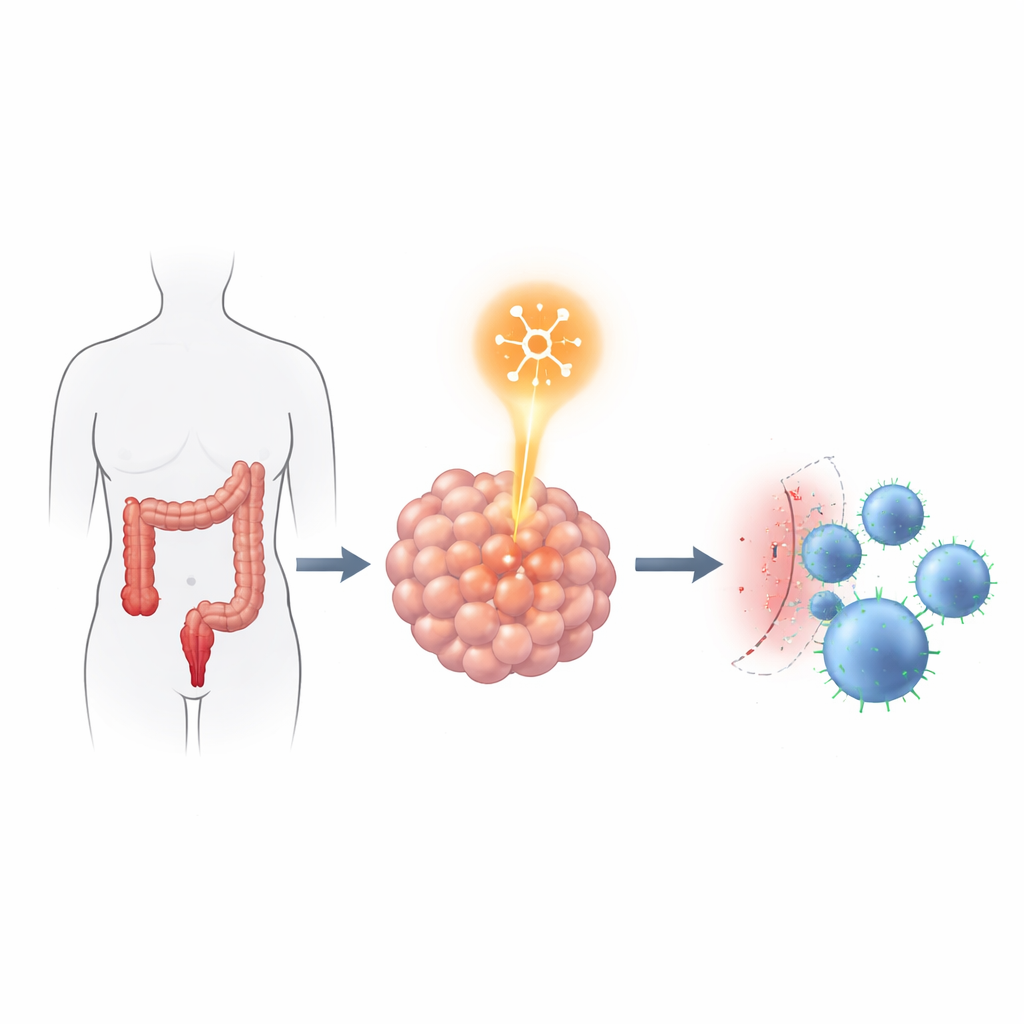
Linking the RNA reader to the immune brake
The team next asked how YTHDF2 might help tumors withstand attacks from immune cells. They implanted human colorectal cancer cells with or without YTHDF2 into special mice that also received human immune cells. Tumors with normal YTHDF2 grew faster and showed fewer cancer‑killing T cells inside. When YTHDF2 was reduced, tumors shrank, and more CD4 and CD8 T cells infiltrated the cancer. Strikingly, lowering YTHDF2 did not change how quickly the cancer cells grew, moved, or invaded in lab dishes, suggesting its main impact was on how the tumor interacted with the immune system rather than on raw tumor aggressiveness.
How cancer cells keep PD-L1 high
PD-L1 is a protein on the surface of tumor cells that binds PD-1 on T cells and tells them to stand down. The researchers observed that when YTHDF2 was knocked down, the amount of PD-L1 protein on cancer cells and in their secreted vesicles (exosomes) fell sharply, even though PD-L1 RNA levels stayed about the same. This hinted that YTHDF2 affects PD-L1 indirectly, after the RNA stage. Detailed tests showed that YTHDF2 loss made PD-L1 protein less stable and easier to destroy by the cell’s protein‑shredding machinery, and it weakened the binding between PD-1 and PD-L1. As a result, T cells in co‑culture experiments killed more cancer cells when YTHDF2 was low, showing a functional boost in immune attack.
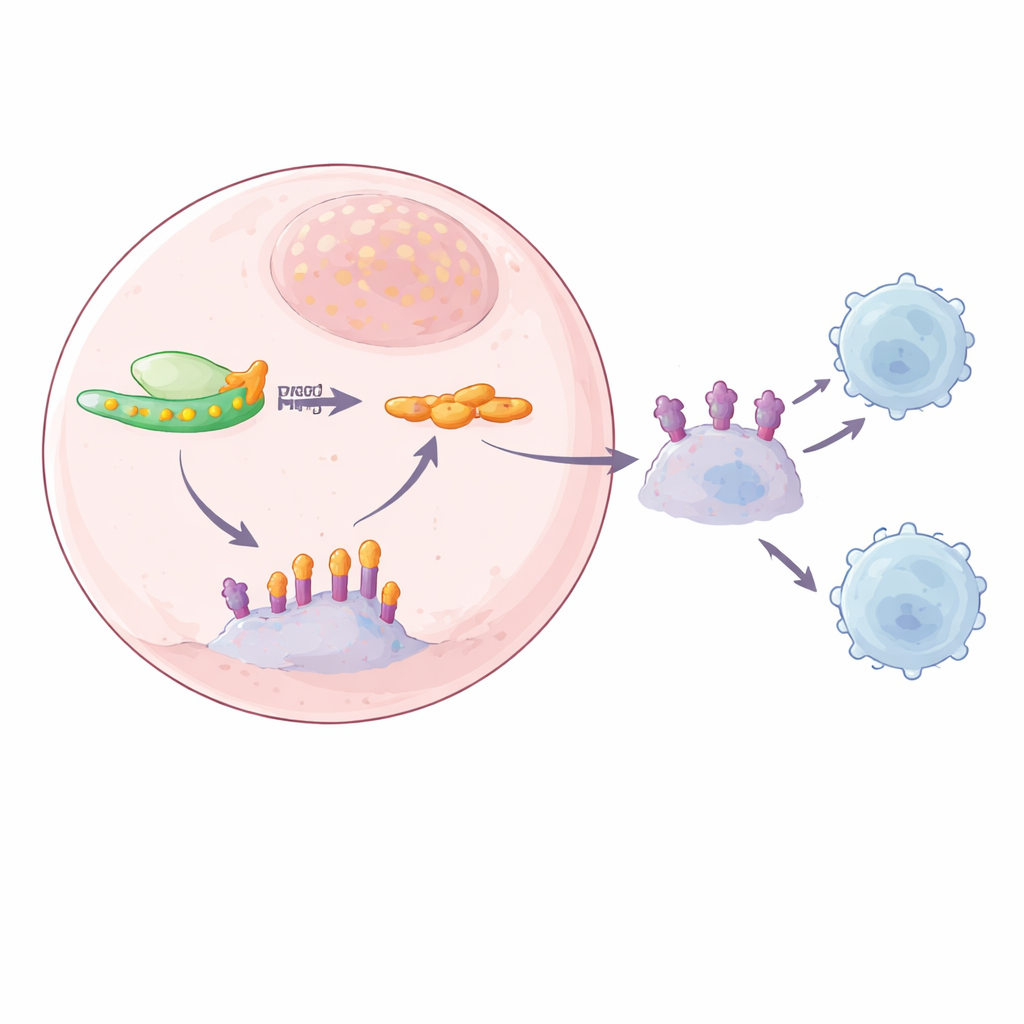
The SPOP connection: a broken clean-up crew
To trace the missing link between YTHDF2 and PD-L1, the team combined several sequencing approaches to hunt for RNA messages that were both tagged with m6A and bound by YTHDF2. One strong candidate was SPOP, part of a protein complex that normally tags PD-L1 for destruction, acting as a clean‑up crew that prevents PD-L1 from piling up. The authors found m6A marks in the tail region of SPOP RNA and showed that YTHDF2 binds exactly there. When YTHDF2 was present at high levels, SPOP RNA was broken down more quickly, leading to lower SPOP protein and less PD-L1 removal. When YTHDF2 was reduced, SPOP RNA became more stable, SPOP protein rose, and PD-L1 protein levels fell, both on the cell surface and in exosomes. Mutating the m6A sites on SPOP RNA or removing SPOP itself broke this chain, confirming that an m6A‑dependent YTHDF2–SPOP pathway controls PD-L1 abundance.
What this means for future treatment
In simple terms, this work reveals a three‑step relay inside colorectal cancer cells: the RNA tag m6A lets YTHDF2 cut down SPOP levels; weakened SPOP cannot properly clear away PD-L1; and the resulting excess PD-L1 shields the tumor from T cells. By interrupting this relay—either by blocking YTHDF2 or strengthening SPOP—it may be possible to lower PD-L1 on tumors and make existing PD-1/PD-L1 drugs more effective, especially for patients who currently see little benefit. The YTHDF2–SPOP–PD-L1 axis thus emerges as a promising new target to help the immune system better recognize and destroy colorectal cancer.
Citation: Xu, X., Chen, H., Zhao, R. et al. Reduced YTHDF2 inhibits PD-L1 expression by stabilizing m6A-containing SPOP mRNA in colorectal cancer. Cell Death Dis 17, 351 (2026). https://doi.org/10.1038/s41419-026-08615-2
Keywords: colorectal cancer, immunotherapy, PD-L1, RNA modification, tumor immune escape