Clear Sky Science · en
Impact of C4BPA on Muscle progenitor cell differentiation: insights for Duchenne muscular dystrophy treatment
Why this muscle story matters
Duchenne muscular dystrophy (DMD) is a severe childhood disease that slowly robs boys of their ability to walk, breathe, and live independently. Most research has focused on the muscle fibers themselves, which are fragile because they lack a key protein called dystrophin. This article looks instead at the “neighbors” of muscle cells—support cells that normally help repair damage—and shows how, in DMD, they begin to sabotage muscle rebuilding through a single secreted protein. Understanding this hidden culprit could open new ways to slow the disease and boost future gene therapies.
Helpers that turn into hindrances
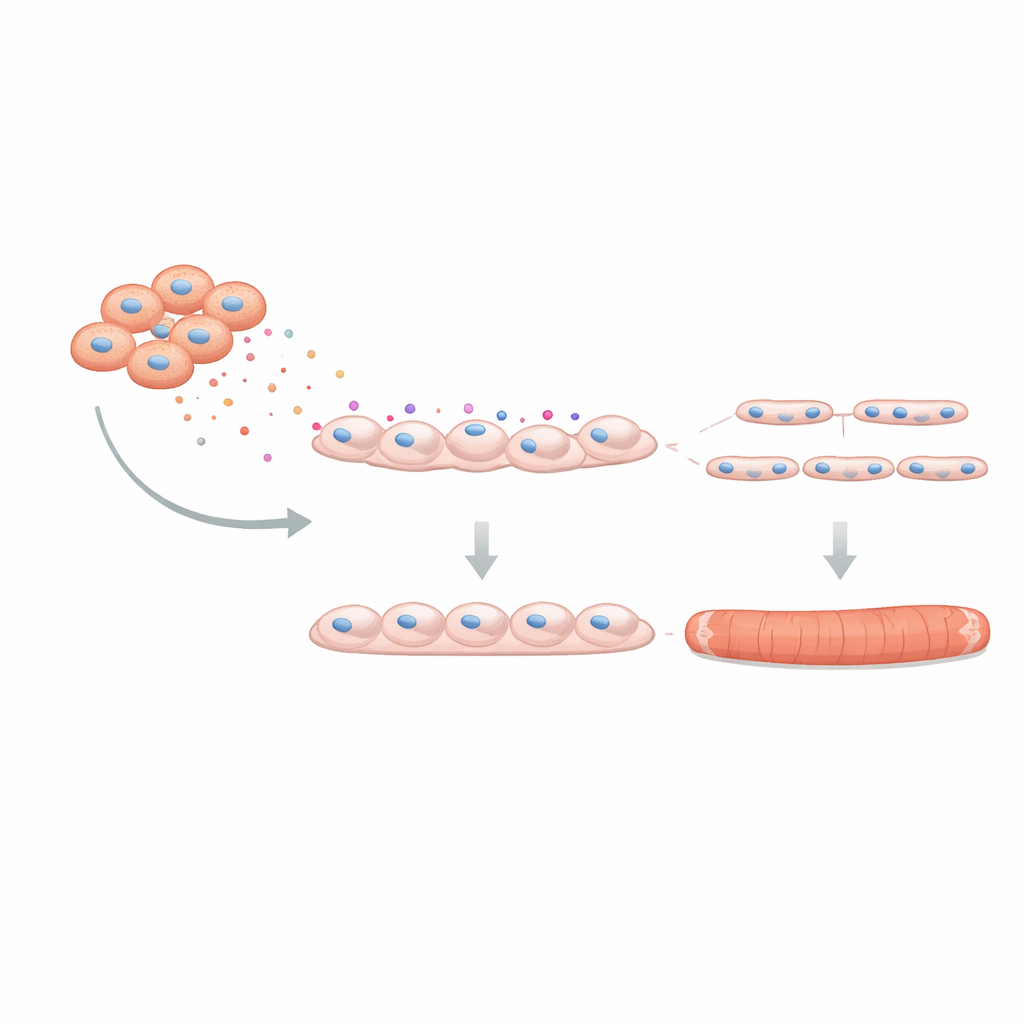
Our muscles do not repair themselves alone. When damage occurs, dormant stem-like cells called myoblasts wake up, divide, and fuse into new muscle fibers. Around them live fibroadipogenic progenitor cells, or FAPs, which normally lay down a temporary scaffold and help coordinate repair. In healthy muscle, these FAPs expand for a short time and then disappear once healing is done. In DMD, however, they stay activated, and the muscle gradually fills with scar and fat. The authors wanted to know: do these altered FAPs directly interfere with the ability of myoblasts to form new fibers?
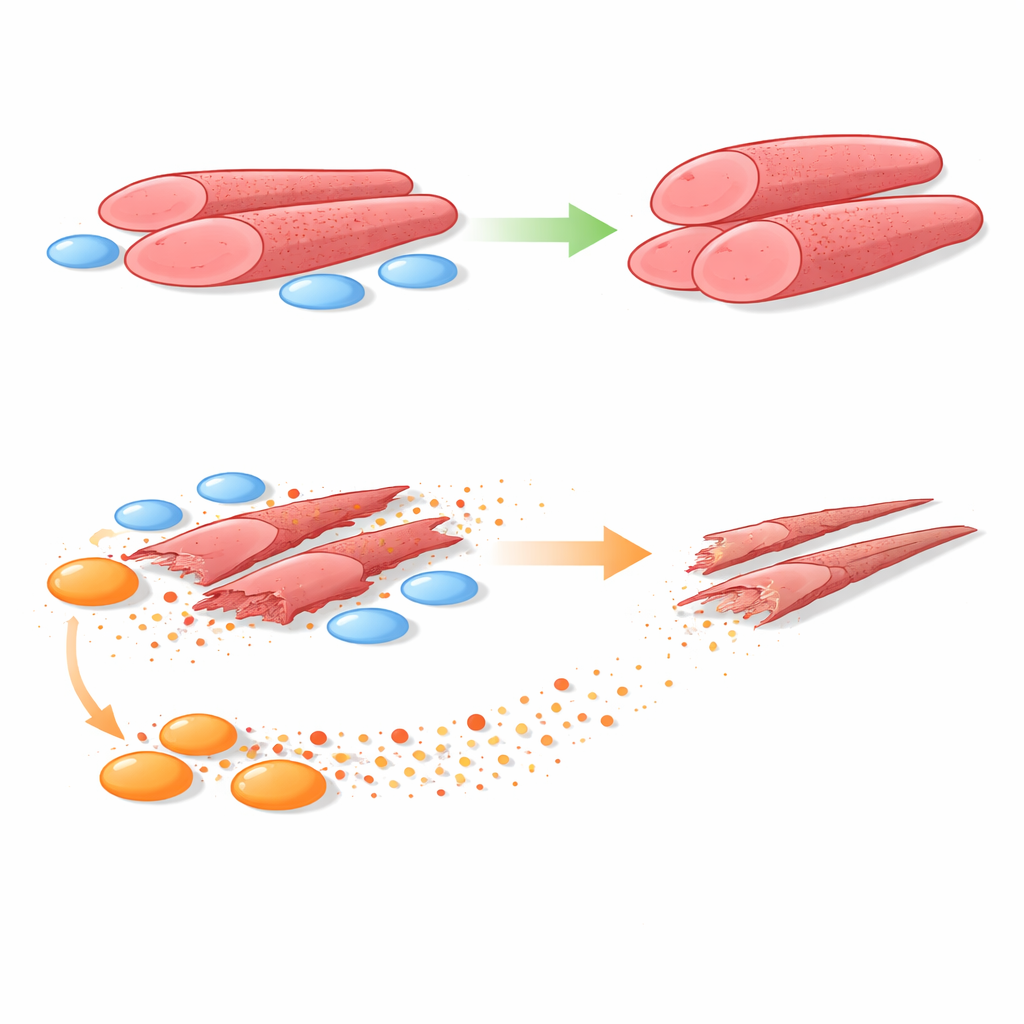
Side-by-side tests of healthy and diseased cells
To answer this, the team grew human myoblasts together with FAPs taken from muscle biopsies of healthy boys and boys with DMD. They used systems that allowed either direct contact between the two cell types or separation by a porous barrier that lets only secreted molecules pass through. In both setups, healthy FAPs encouraged myoblasts to form larger, more robust muscle tubes. In sharp contrast, DMD FAPs consistently reduced the size of these tubes and the number of cell nuclei they contained—clear signs of poorer muscle formation. When physical contact was removed, DMD FAPs still did harm, pointing to soluble factors as the main offenders.
Hunting down a toxic signal
The researchers next catalogued the proteins released by healthy and DMD FAPs using mass spectrometry, a technique that can identify hundreds of molecules at once. Out of 760 detected proteins, nearly half were more abundant in DMD FAPs. By cross-referencing this list with receptors present on human muscle cells, they narrowed the field to 29 candidates that could plausibly act on myoblasts. Further testing singled out one key protein, called C4BPA, which was markedly higher in DMD FAP cultures and also in the blood and muscle tissue of DMD patients. When healthy myoblasts were exposed to purified C4BPA in the lab, their ability to fuse into long, thick tubes dropped sharply, and genes that drive muscle growth were turned down while genes linked to muscle wasting were turned up.
From petri dish to engineered muscle
To see whether this effect mattered for actual muscle function, the team used a three-dimensional “mini-muscle” grown from human cells in a soft scaffold. These engineered tissues can contract when electrically stimulated, mimicking real muscle. When C4BPA was present during their formation, the resulting fibers were thinner and produced weaker twitch and tetanic forces, with signs of earlier fatigue. Finally, the researchers asked whether blocking C4BPA could undo some of the damage. Using a genetic silencing approach to reduce C4BPA production in DMD FAPs, they co-cultured these modified cells with healthy myoblasts. Muscle tube formation improved: more myoblasts fused, and each tube contained more nuclei, indicating that dampening C4BPA partially restores the supportive role FAPs should play.
What this means for future treatments
This study reveals that in Duchenne muscular dystrophy, support cells in the muscle become “bad neighbors” by overproducing C4BPA, a protein that disrupts the growth and function of new muscle fibers. While it does not fix the underlying genetic defect in dystrophin, targeting C4BPA or similar secreted factors could help preserve remaining muscle and keep the tissue more responsive to emerging gene and cell therapies. In simple terms, the work suggests that treating DMD may require not only repairing the muscle’s bricks but also calming the surrounding “construction crew” so that regeneration can proceed instead of being blocked.
Citation: Fernández-Simón, E., Tejedera-Villafranca, A., Fernández-Garabay, X. et al. Impact of C4BPA on Muscle progenitor cell differentiation: insights for Duchenne muscular dystrophy treatment. Cell Death Dis 17, 313 (2026). https://doi.org/10.1038/s41419-026-08588-2
Keywords: Duchenne muscular dystrophy, muscle regeneration, fibroadipogenic progenitors, C4BPA, muscle stem cells