Clear Sky Science · en
NUP93 facilitates the nuclear import of SOX2 to activate G3BP1 transcription and impairs gemcitabine response in pancreatic cancer
Why this research matters
Pancreatic cancer is one of the deadliest cancers, in part because it often shrugs off chemotherapy drugs like gemcitabine. This study uncovers a hidden chain of molecular events inside pancreatic cancer cells that helps them repair the very DNA damage gemcitabine is meant to cause. By tracing this chain from the cell’s outer border to its genetic core, the authors reveal a new weak spot that future treatments might exploit to make chemotherapy work better.
A gatekeeper at the cell’s nucleus
Every cell keeps its DNA inside a protected compartment called the nucleus, which is studded with tiny gateways known as nuclear pores. The protein NUP93 is a key structural part of these gates. By mining large cancer datasets and examining patient samples, the researchers found that NUP93 levels are significantly higher in pancreatic ductal adenocarcinoma than in normal pancreatic tissue. Patients whose tumors carried more NUP93 tended to have worse survival. Lab-grown pancreatic cancer cells were especially dependent on NUP93: when its levels were reduced, these cells grew more slowly and formed fewer colonies, indicating that NUP93 is tied to aggressive tumor behavior.
How cancer cells dodge chemotherapy
Gemcitabine works by inserting itself into DNA as it is being copied, triggering errors and breaks that should stop tumor cells from dividing. Cancer cells, however, can fight back by activating DNA repair systems. By comparing tumors with high and low NUP93 levels, the team saw that genes involved in DNA repair were more active when NUP93 was abundant. In cell experiments, knocking down NUP93 made pancreatic cancer cells far more sensitive to gemcitabine, while extra NUP93 made them harder to kill. Markers of DNA damage increased when NUP93 was lost and decreased when it was overproduced, and a "comet" assay that visualizes broken DNA confirmed that cells without NUP93 accumulated more damage after drug treatment. In mice, tumors lacking NUP93 grew more slowly and responded much better to gemcitabine.
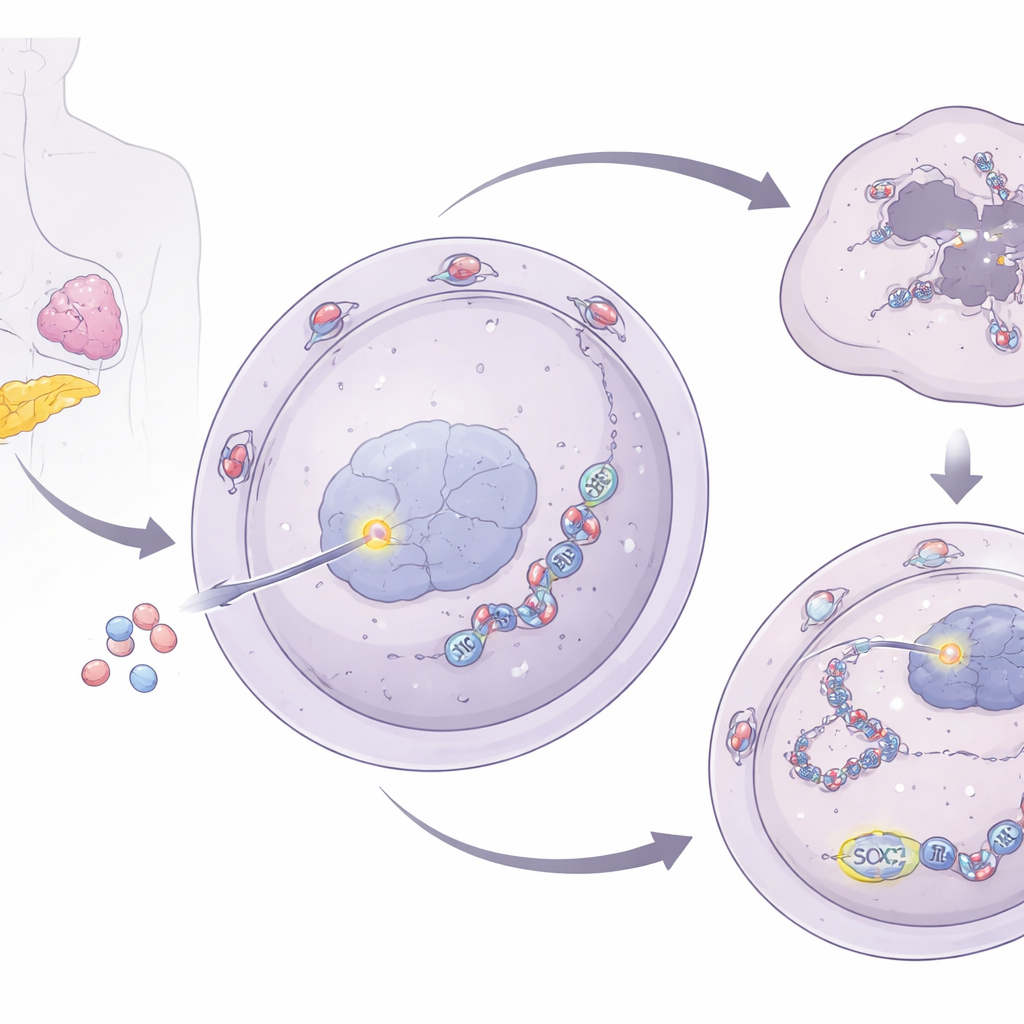
A relay from the nuclear gate to stress granules
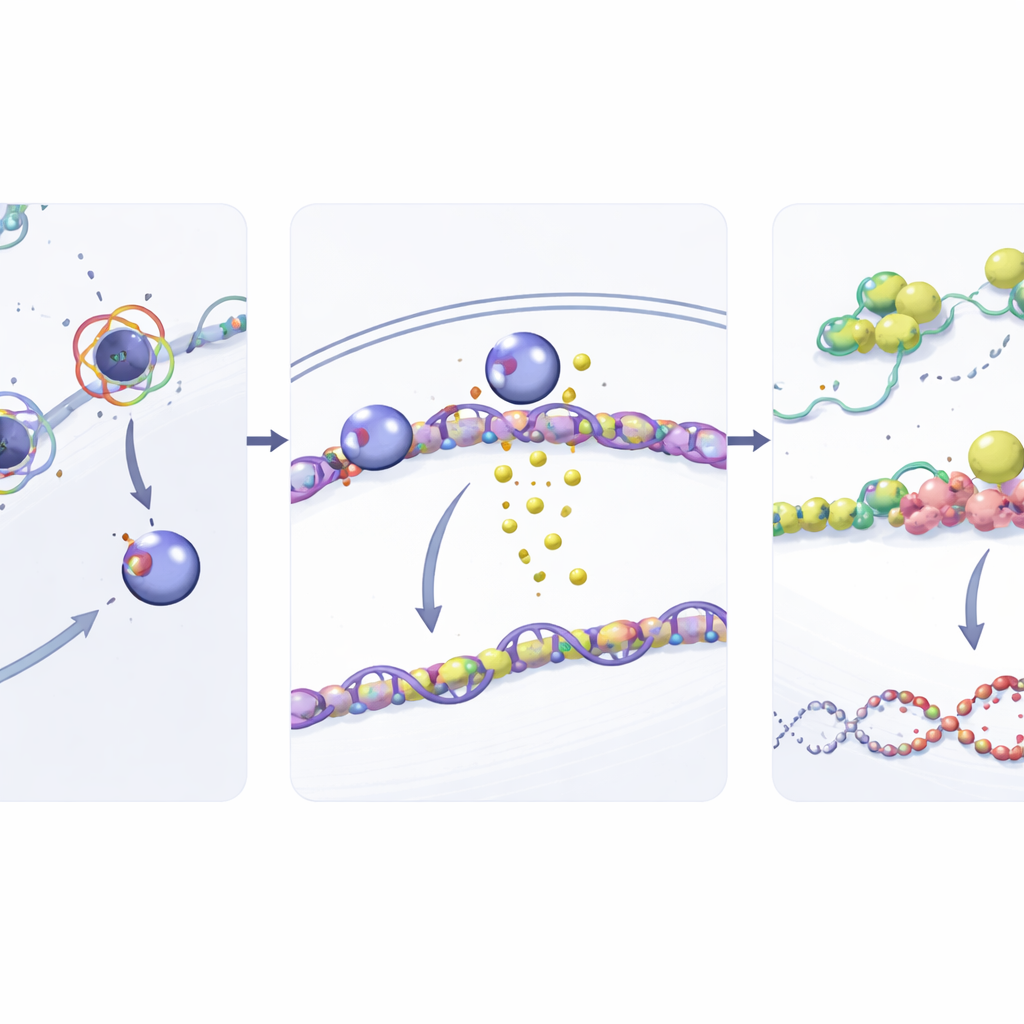
To understand how NUP93 strengthens DNA repair, the researchers looked at its links to cellular stress granules—temporary clusters of RNA and proteins that help cells survive harsh conditions. They discovered that NUP93 boosts levels of G3BP1, a core stress granule protein. When NUP93 was increased, G3BP1 levels rose; when NUP93 was reduced, G3BP1 fell. Importantly, removing G3BP1 canceled out NUP93’s ability to promote cell growth and gemcitabine resistance and restored DNA damage after treatment. The team then traced G3BP1 control back to SOX2, a well-known transcription factor that turns genes on and off. NUP93 physically interacts with SOX2 at the nuclear pore and helps ferry it into the nucleus. Once inside, SOX2 binds directly to the G3BP1 gene’s control region, turning up its production.
From RNA protection to DNA repair
The story does not end with G3BP1. The researchers showed that G3BP1, in turn, helps stabilize the messenger RNA for RAD51, a central player in a high-accuracy DNA repair pathway called homologous recombination. G3BP1 binds directly to RAD51 RNA and slows its breakdown, leading to higher RAD51 protein levels. With more RAD51 available, cancer cells are better equipped to mend gemcitabine-induced DNA breaks. When G3BP1 was depleted, RAD51 levels dropped, RAD51 RNA degraded more quickly, and DNA damage accumulated. In mouse models, blocking SOX2 or G3BP1 reduced tumor growth and made gemcitabine much more effective, confirming the importance of this relay in living systems.
A new target to help chemotherapy work
Putting the pieces together, the authors propose a clear pathway: NUP93 at the nuclear pore ushers SOX2 into the nucleus, SOX2 activates G3BP1, and G3BP1 preserves RAD51 RNA, ultimately boosting DNA repair and helping pancreatic cancer cells resist gemcitabine. This NUP93–SOX2–G3BP1–RAD51 axis acts like an internal rescue team that repairs the damage chemotherapy is supposed to inflict. By disrupting one or more steps in this chain, future therapies might weaken cancer cells’ defenses and make existing drugs more potent, offering new hope for improving outcomes in this notoriously hard-to-treat disease.
Citation: Sun, H., Yang, C., Du, J. et al. NUP93 facilitates the nuclear import of SOX2 to activate G3BP1 transcription and impairs gemcitabine response in pancreatic cancer. Cell Death Dis 17, 423 (2026). https://doi.org/10.1038/s41419-026-08586-4
Keywords: pancreatic cancer, gemcitabine resistance, DNA repair, nuclear pore, stress granules