Clear Sky Science · en
ZNRF1 deficiency disrupts Fas ligand trafficking and immune balance
Why this matters for our immune health
Our immune system must walk a fine line: it has to attack threats such as microbes and foreign blood cells, yet shut itself down before the response spirals out of control. This study uncovers a hidden control switch inside a common immune cell called the macrophage that helps decide whether activated immune cells are quietly removed or allowed to keep multiplying. Understanding this switch may eventually help us better manage conditions involving chronic inflammation, autoimmunity, or complications from transfusion.
A traffic light for immune self‑destruction
When immune cells have done their job, many of them are eliminated through a built‑in self‑destruct signal involving a surface molecule called Fas ligand. Fas ligand on one cell triggers death in another cell that carries its matching receptor, helping to trim back overactive responses. Most research has focused on how much Fas ligand a cell makes. Here, the authors asked a different question: how is Fas ligand moved from internal storage compartments to the outer surface of macrophages at just the right time, and what happens if that delivery step goes wrong?
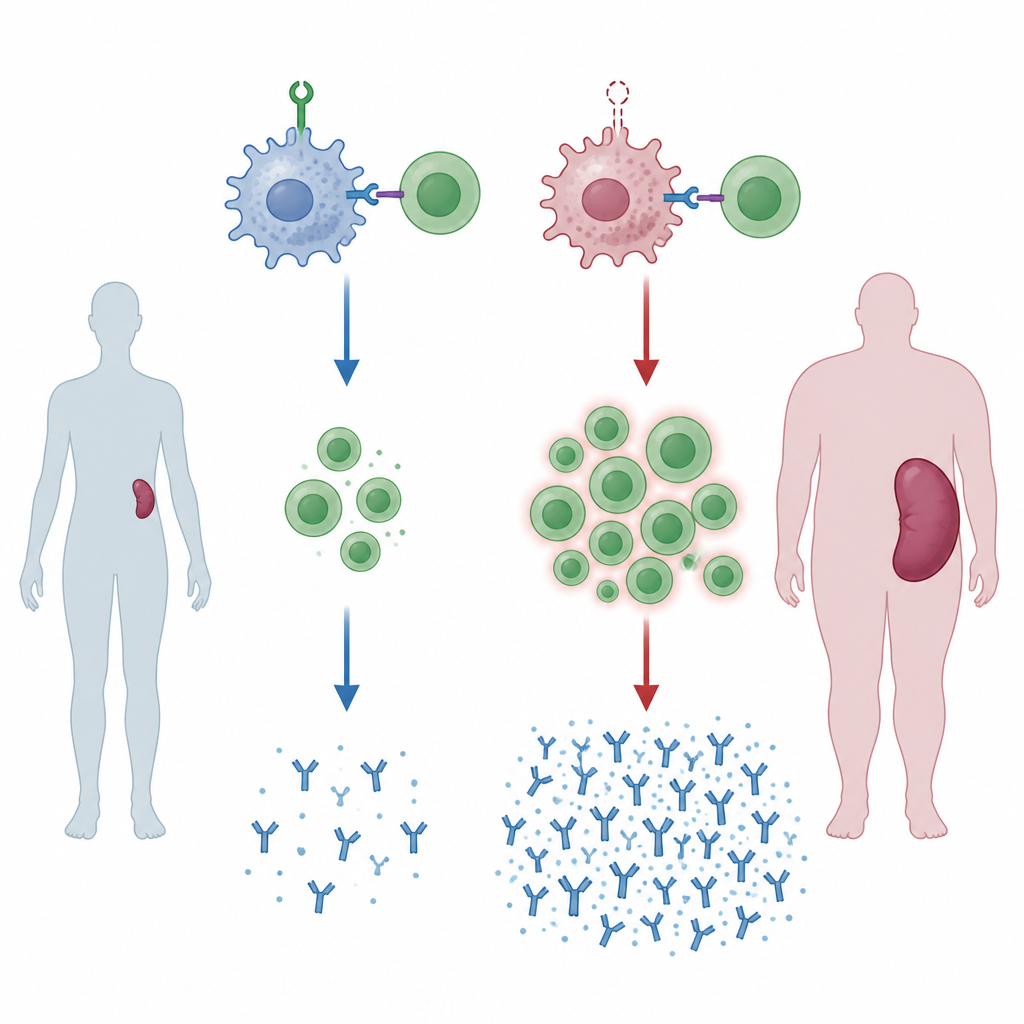
A missing guardian leads to swollen spleens
The team engineered mice whose myeloid cells, including macrophages, lack a protein called ZNRF1. As these mice aged, their spleens became enlarged, a sign of chronic immune activation. When the animals were exposed to foreign blood, mimicking a transfusion from a genetically different donor, the changes became more striking. Their spleens showed expanded regions where B cells and T cells interact and multiply, and the balance of helper T cells to killer T cells shifted upward. These mice also produced higher amounts of certain classes of antibodies against the donor blood, indicating that, without ZNRF1 in myeloid cells, the immune system is primed to respond too strongly.
Surface signal blocked while cargo piles up inside
To understand what ZNRF1 does inside macrophages, the researchers turned to cell culture. They stimulated normal and ZNRF1‑deficient macrophages with bacterial components or live bacteria and measured Fas ligand. Surprisingly, cells lacking ZNRF1 made plenty of Fas ligand overall, often even more than normal cells, but they failed to display it on their surface. Under the microscope, internal storage compartments marked by the protein LAMP1 still moved toward the cell edge in both cell types, but in ZNRF1‑deficient cells the Fas ligand cargo did not travel with them to the cortex and remained stuck near the center. This pointed to a late breakdown in the docking or fusion step rather than a problem in making or initially storing Fas ligand.
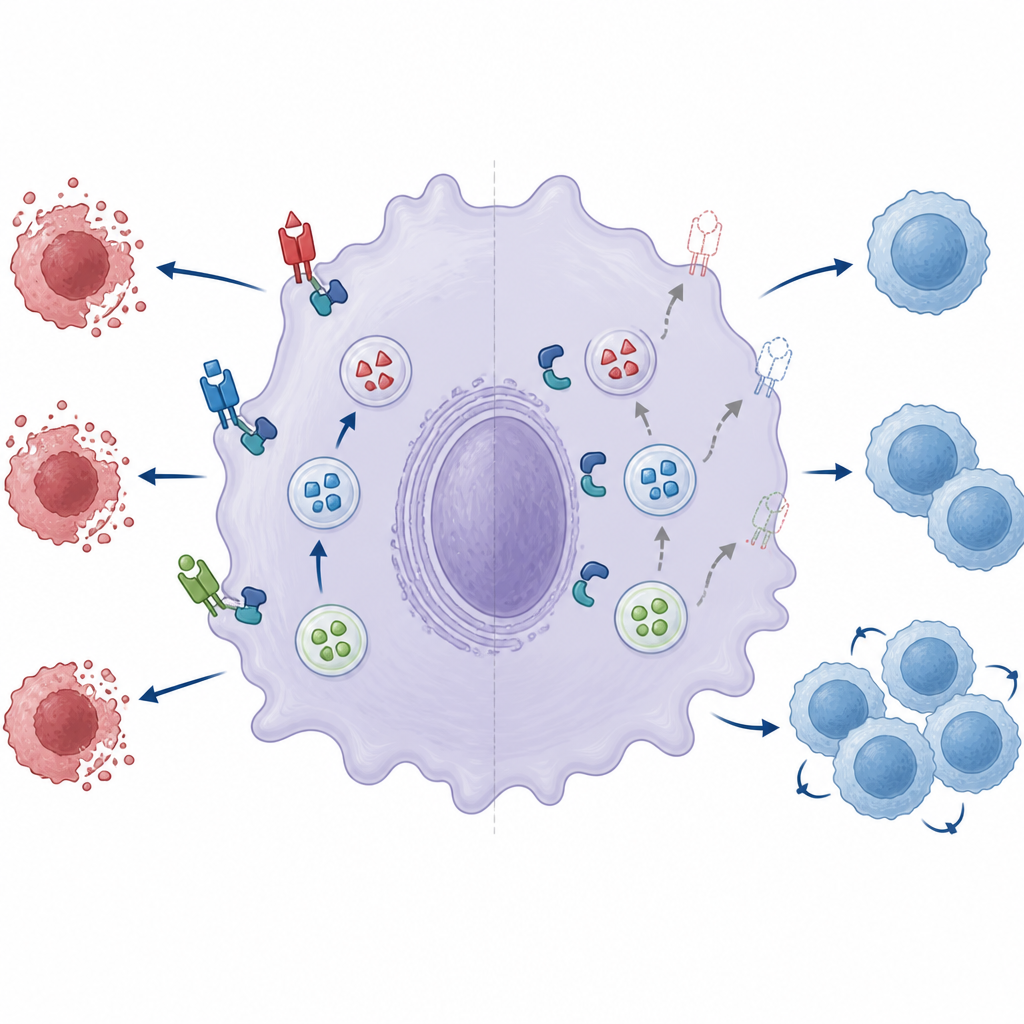
A broken fusion machine weakens immune braking
The authors then examined two key components of the tiny fusion machine that lets storage vesicles merge with the cell surface: a pair of proteins called Munc18‑2 and Syntaxin‑3. In normal macrophages, these two partners form a complex that helps vesicles open and release their cargo. When ZNRF1 was missing, the partnership between Munc18‑2 and Syntaxin‑3 was markedly weaker. Reducing Munc18‑2 levels further cut surface Fas ligand, underscoring its importance. Re‑introducing normal ZNRF1 into deficient cells improved Fas ligand display, but a catalytically inactive version of ZNRF1 did not, suggesting that ZNRF1’s enzymatic activity is needed to make the fusion machinery fully competent even if the complex itself can still assemble.
Consequences for killing off spent immune cells
Because Fas ligand’s job is to trigger death in activated cells, the team tested how well macrophages could kill target cells. When normal macrophages were mixed with activated helper T cells or with a cell line highly sensitive to Fas signals, many targets died over time. In contrast, ZNRF1‑deficient macrophages spared far more of these cells, even when they were strongly stimulated. This reduced killing ability fits with the mouse data: if macrophages cannot efficiently expose Fas ligand on their surface, activated T cells survive longer, providing more help to B cells and fueling enlarged spleens and boosted antibody production.
What this means for controlling runaway immunity
Together, the findings reveal ZNRF1 as a built‑in safety device that links the internal movement of Fas ligand–filled packets to their final fusion with the macrophage surface. When ZNRF1 functions normally, Fas ligand reaches the outside of the cell, allowing macrophages to cull activated T cells and keep immune reactions in check. When ZNRF1 is absent, this delivery step fails, surface Fas ligand stays low, and immune cells are left to proliferate more freely. While much work remains before these insights can be translated into therapies, the study highlights a specific molecular handle by which the body fine‑tunes the shut‑down phase of immune responses.
Citation: Lai, TY., Chang, YC., Lin, YS. et al. ZNRF1 deficiency disrupts Fas ligand trafficking and immune balance. Cell Death Dis 17, 422 (2026). https://doi.org/10.1038/s41419-026-08566-8
Keywords: Fas ligand, macrophages, immune homeostasis, ZNRF1, T cell apoptosis