Clear Sky Science · en
HIF-1α suppresses SNPH expression to facilitate liver metastasis of colorectal cancer through regulating mitochondrial dynamics and filopodia formation
Why this research matters
Colorectal cancer is common, and about half of patients eventually develop tumors in the liver, which are difficult to treat and often deadly. This study digs into the tiny power plants inside cancer cells—the mitochondria—to understand how changes in their behavior help cancer cells break away from the intestine, travel through the body, and settle in the liver. By uncovering a hidden control system that links low oxygen, energy machinery, and cell movement, the work points to new ways to slow or block the spread of colorectal cancer.
A cellular traffic cop goes missing
The researchers focused on a protein called syntaphilin (SNPH), which acts like a traffic cop for mitochondria, helping keep them anchored and fused together. By mining large public gene databases and analyzing patient samples, they found that SNPH levels are higher in nearby healthy tissue, lower in primary colorectal tumors, and lowest in liver metastases. Patients whose tumors had little SNPH tended to have worse survival. In both cell lines and mouse models, boosting SNPH reduced the number and size of liver tumors, while low SNPH was tied to more aggressive spread. These patterns suggest that losing this mitochondrial "anchor" is a key step in making cancer cells more mobile and dangerous.
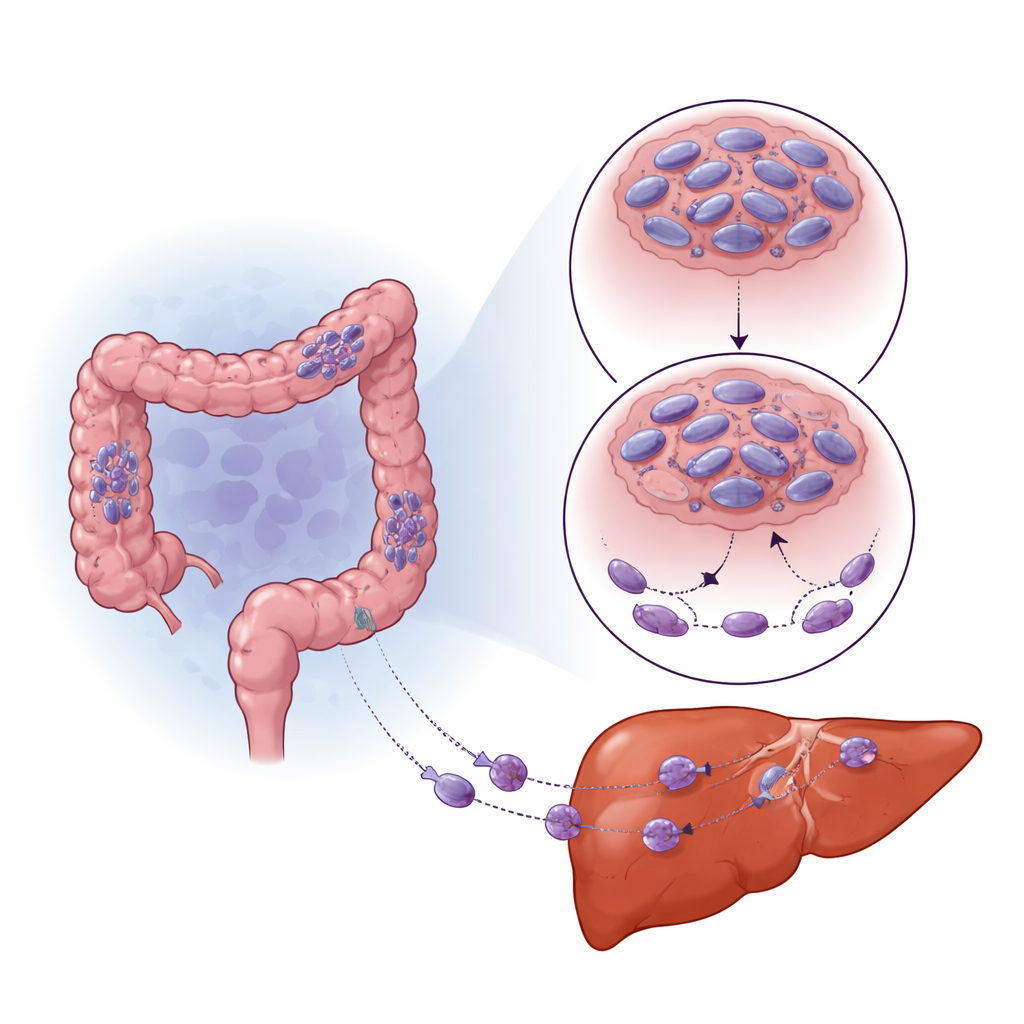
How low oxygen rewires tumor behavior
Solid tumors often outgrow their blood supply, creating pockets of low oxygen (hypoxia). Cells sense this through a master switch protein called HIF-1α, which helps them adapt and survive. The team noticed that areas of colorectal tumors and liver metastases with high HIF-1α had especially low SNPH. When they grew cancer cells under low-oxygen conditions in the lab, SNPH levels dropped sharply over time, and the cells became more mobile and invasive. Blocking HIF-1α partly rescued SNPH levels and reduced this increase in movement. Rather than turning SNPH off directly, HIF-1α boosted a small regulatory RNA, miR-130a-3p, which then bound to SNPH’s message and prevented it from being made. Inhibiting miR-130a-3p protected SNPH, curbed the growth of thin cell “fingers” and reduced invasion, even in low oxygen.
From power plants to “grappling hooks”
SNPH loss did more than just free mitochondria to move. Under normal conditions, mitochondria tend to be elongated and clustered, but when SNPH was reduced, they became fragmented and drifted to the cell’s outer edge. This rearrangement boosted the production of reactive oxygen species (ROS), chemically reactive byproducts of metabolism. These ROS switched on a signaling chain centered on the AKT and cdc42 proteins, which in turn activated downstream partners PAK1 and Cofilin. The combined effect was a remodeling of the actin skeleton—the internal scaffolding of the cell—promoting long, thin protrusions called filopodia. These structures act like grappling hooks, helping cells pull themselves forward and squeeze through surrounding tissue. Treating cells with ROS scavengers dampened this signaling and reduced filopodia, while adding ROS mimics made filopodia more abundant unless SNPH was restored.
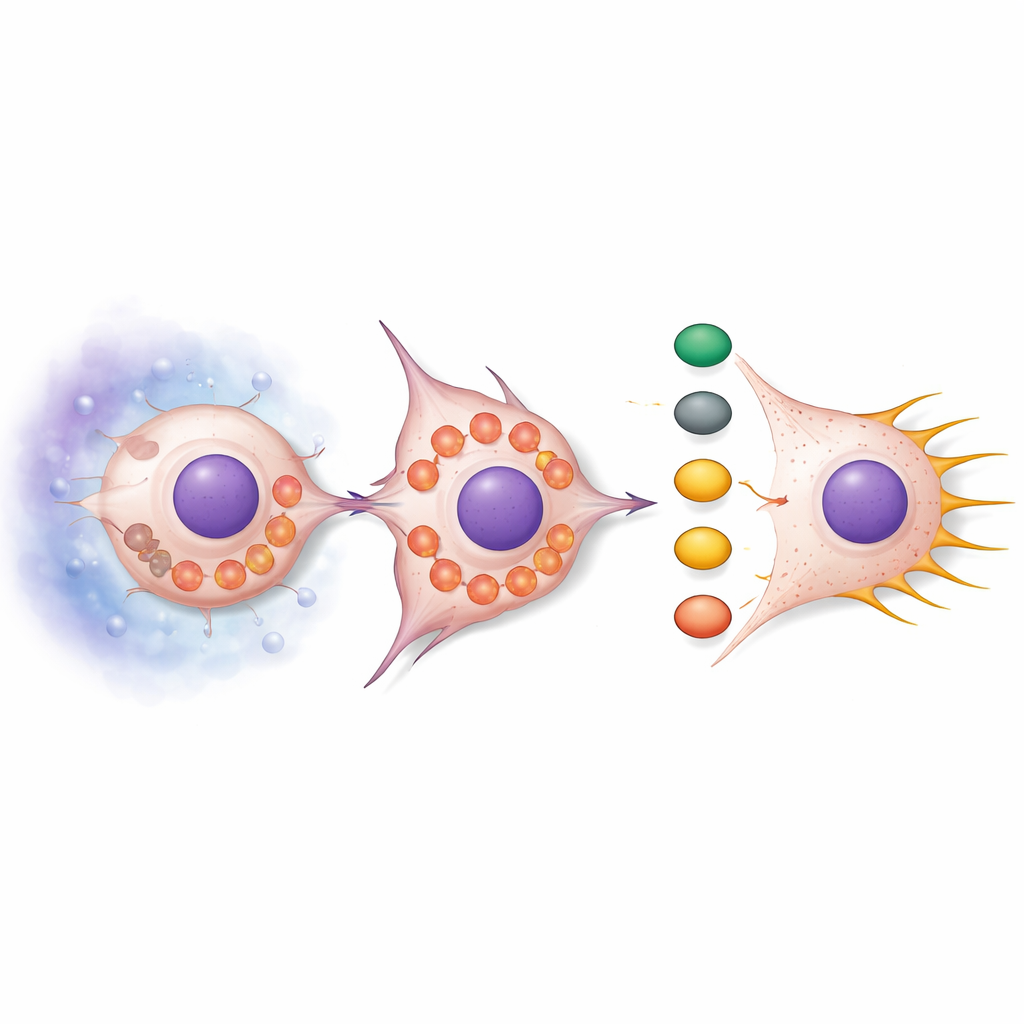
Testing the mechanism in living animals
To see whether this pathway truly affects spread to the liver, the researchers injected colorectal cancer cells with or without extra SNPH into mice. Animals receiving SNPH-rich cells developed far fewer and smaller liver tumors, even though the overall growth rate of the cancer cells was similar. Microscopy of liver metastases showed that SNPH-overexpressing tumors had longer, more fused mitochondria, lower HIF-1α levels, and weaker activation of the PAK1/Cofilin arm of the signaling cascade. In other words, restoring the mitochondrial “anchor” shifted both mitochondrial shape and the chemical messaging network back toward a less mobile state, making it harder for cancer cells to colonize the liver.
What it means for patients
Put simply, this study reveals a chain reaction: low oxygen in tumors activates HIF-1α, which turns on miR-130a-3p, which then silences SNPH. Without SNPH, mitochondria fragment and move to the cell edge, generating more ROS that switch on a movement-promoting signaling pathway. That, in turn, reshapes the cell surface into filopodia that help colorectal cancer cells migrate and seed the liver. Targeting any part of this chain—HIF-1α, miR-130a-3p, ROS production, or the downstream signaling steps—could offer new strategies to prevent or limit liver metastasis, and SNPH itself may serve as a useful marker of how likely a patient’s cancer is to spread.
Citation: Zhan, L., Li, X., Li, X. et al. HIF-1α suppresses SNPH expression to facilitate liver metastasis of colorectal cancer through regulating mitochondrial dynamics and filopodia formation. Cell Death Dis 17, 380 (2026). https://doi.org/10.1038/s41419-026-08551-1
Keywords: colorectal cancer metastasis, mitochondrial dynamics, hypoxia HIF-1α, microRNA-130a-3p, reactive oxygen species