Clear Sky Science · en
APOE4 exacerbates glucocorticoid stress hormone-induced tau pathology via mitochondrial dysfunction
Why stress and memory genes matter to all of us
Many people worry that long periods of stress might raise their chances of developing memory problems later in life. At the same time, a common gene variant called APOE4 is known to strongly increase the risk of Alzheimer’s disease. This study brings these two threads together, asking how stress hormones and APOE4 interact inside brain cells, and whether that interaction might point to new ways to protect the aging brain.
How a common risk gene meets everyday stress
The researchers focused on APOE4, a version of a cholesterol-handling protein carried by millions of people worldwide. They paired this genetic background with exposure to glucocorticoids, the main stress hormones released during chronic psychological pressure. Using mice engineered to carry human APOE3 (the more neutral version) or APOE4, and nerve cells grown from their brains, they zoomed in on the hippocampus, a memory hub that is especially sensitive to both stress and Alzheimer’s disease. Their goal was to see whether APOE4 makes these neurons more vulnerable to the harmful effects of stress hormones.
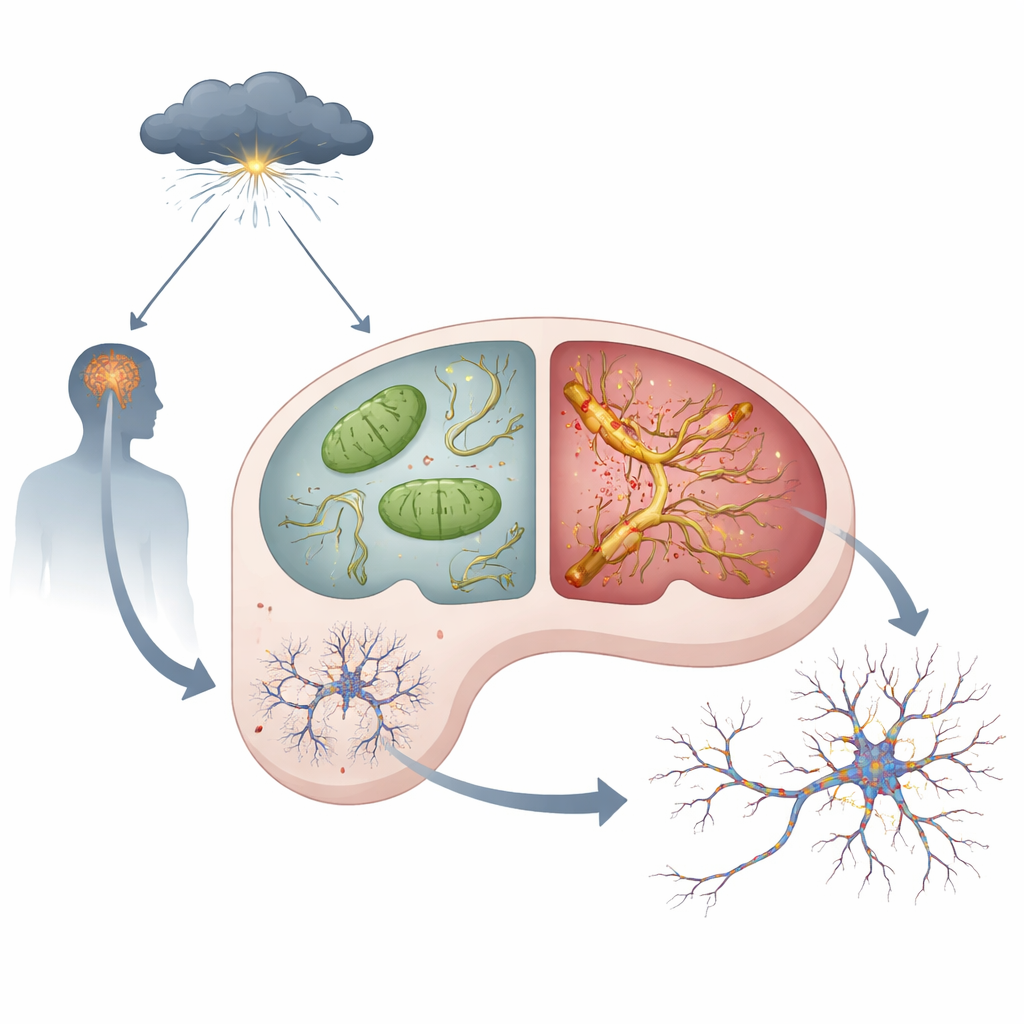
Stress hormones, faulty power plants, and toxic tau
Inside each neuron sit mitochondria, tiny power plants that generate the energy needed for thinking and remembering. The team found that even under calm conditions, APOE4 mice showed signs of mitochondrial trouble in the hippocampus: more reactive oxygen byproducts, weaker activity of a key energy-making complex, and lower levels of ATP, the cell’s energy currency. When the animals were treated with high doses of a synthetic stress hormone that mimics chronic stress, these problems became worse in APOE4 mice than in APOE3 mice. At the same time, a protein called tau — which normally helps stabilize internal cell “tracks” — began to build up, become excessively modified, clump, and spread from neuron to neuron. These tau changes, often called tau pathology, were much stronger and spread farther in APOE4 brains exposed to stress hormones.
Overactive stress signaling in vulnerable brains
The study also examined how strongly brain cells responded to stress signals. Glucocorticoids act through receptors that normally sit in the cell’s fluid and move into the nucleus and mitochondria when activated. In APOE4 mice, these receptors were already more active at baseline and shifted more readily into the nucleus and mitochondria, even though overall hormone levels in the blood were similar to APOE3 animals. Supporting this, APOE4 animals and neurons showed higher levels of helper proteins that promote receptor activation, and gene-expression patterns consistent with stronger stress-hormone signaling. In cell culture, very low doses of stress hormone that had little effect on APOE3 neurons were enough to damage mitochondria and trigger tau buildup and release in APOE4 neurons, showing that this genetic background lowers the threshold for stress-related harm.
A critical pore in mitochondria and a possible way to intervene
Digging deeper, the researchers homed in on the mitochondrial permeability transition pore (mPTP), a channel that can open in the inner mitochondrial membrane. When this pore opens, mitochondria lose their electrical charge, pump out harmful reactive molecules, and can push cells toward dysfunction. In APOE4 neurons, even modest stress-hormone exposure was sufficient to increase levels of cyclophilin D, a key pore regulator, and to trigger pore opening. Blocking this pore with two different drugs — one that directly interferes with cyclophilin D and another that dampens mitochondrial oxidant production (mito-apocynin) — protected APOE4 neurons from energy loss, excess reactive molecules, and tau changes.
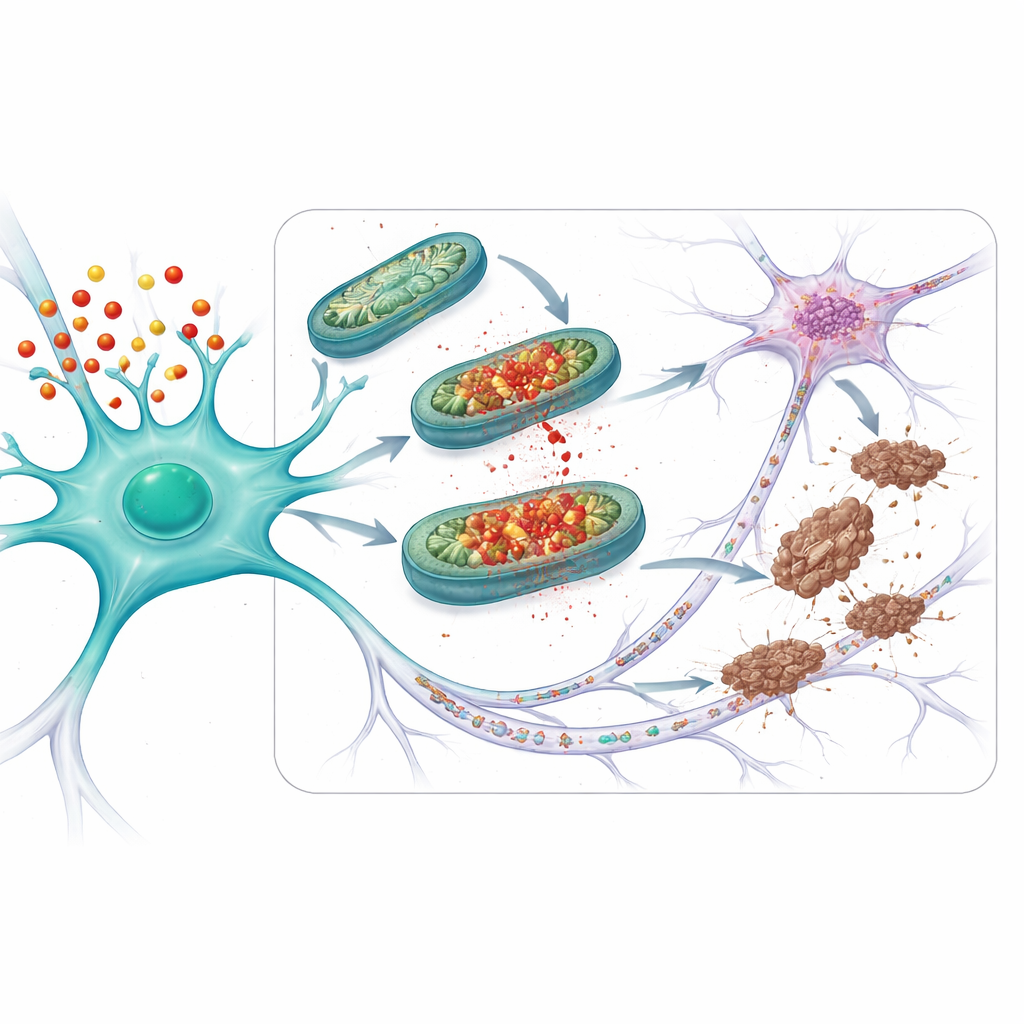
Protecting connections and aging brains
The team then asked whether this protective strategy would help in more disease-like settings. They studied mice that not only carried APOE4 but also produced a mutant form of tau that readily forms tangles. In these animals, mitochondrial problems, abnormal tau, and loss of synapses — the connection points between neurons — were all worse than in mice with the safer APOE3 version. Treating APOE4 mice with mito-apocynin, starting before obvious symptoms appeared, reduced mitochondrial damage, tau changes, and synapse loss. In older APOE4 mice with naturally emerging tau problems (even without added mutant tau), short-term mito-apocynin treatment similarly restored mitochondrial function, reduced abnormal tau, and limited how far tau spread between neurons.
What this means for people at risk
In simple terms, this work suggests that APOE4 makes brain cells’ power plants more fragile and their stress-response switches more easily flipped. When chronic stress hormones are added on top, this combination accelerates the cascade leading from mitochondrial damage to toxic tau buildup, spread, and loss of brain connections. Crucially, the study shows that blocking a specific mitochondrial pore can interrupt this cascade in APOE4 models. While much more work is needed before any treatment reaches patients, targeting this pore — and managing chronic stress, especially in APOE4 carriers — may become part of future strategies to slow or prevent Alzheimer’s-related damage.
Citation: Yu, Q., Du, F., Puerta-Alvarado, V. et al. APOE4 exacerbates glucocorticoid stress hormone-induced tau pathology via mitochondrial dysfunction. Cell Death Dis 17, 419 (2026). https://doi.org/10.1038/s41419-026-08543-1
Keywords: APOE4, chronic stress, mitochondria, tau pathology, Alzheimer’s disease