Clear Sky Science · en
ADAR2 induces the differentiation of osteosarcoma cells by editing activity on IGFBP7: new implications for therapy
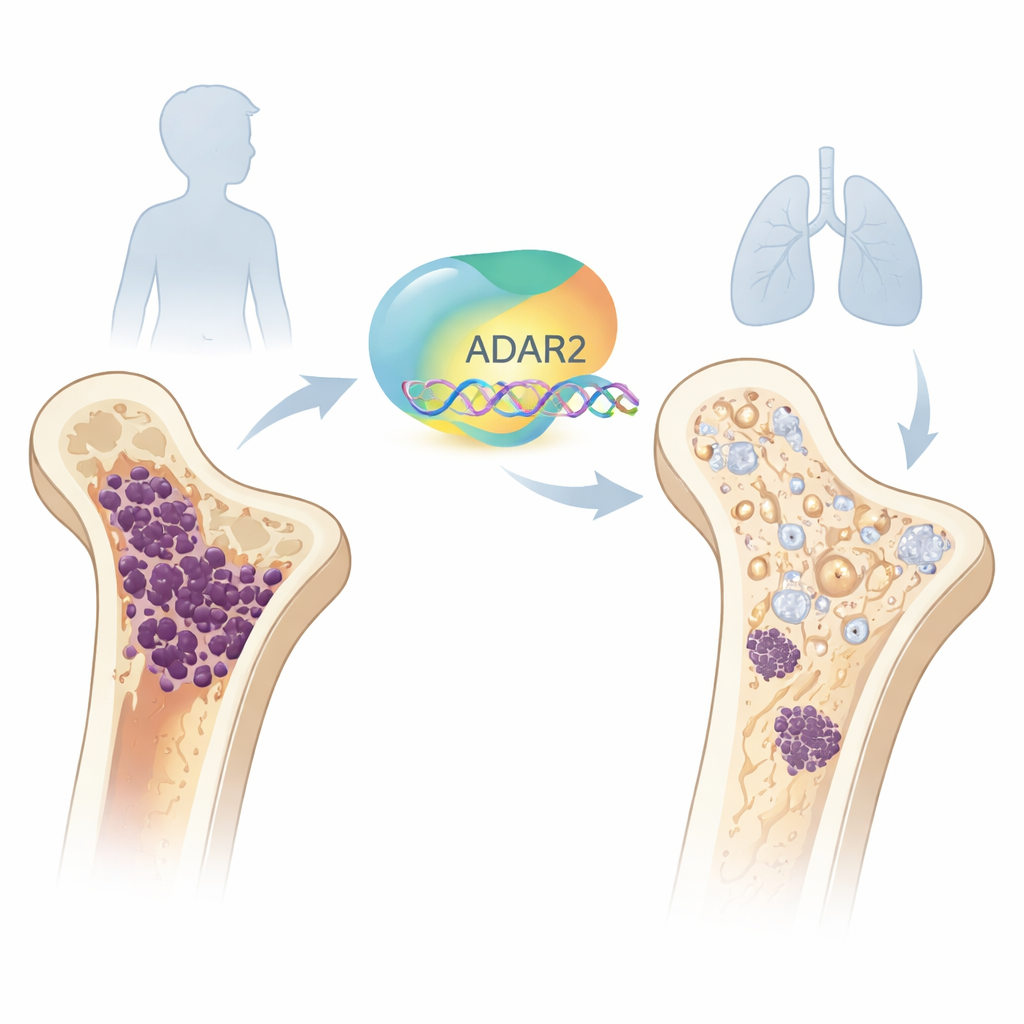
Turning a Dangerous Bone Cancer Toward Healing
Osteosarcoma is a rare but aggressive bone cancer that mainly strikes children and teenagers. Despite surgery and powerful chemotherapy, too many young patients still relapse or develop lung metastases. This study explores an emerging idea: instead of only trying to kill cancer cells, can we coax them to grow up and behave more like normal bone-forming cells? The researchers focus on a natural RNA-editing enzyme called ADAR2 and uncover how boosting its activity can push osteosarcoma cells toward a more mature, less dangerous state.
A Molecular Editor With a Protective Side
Inside every cell, messages that carry genetic information are written in RNA. ADAR2 is one of the body’s "editors" that can subtly change these messages after they are written. The team first looked at normal bone development and found that ADAR2 levels are low in early stem cells from the bone marrow but rise as these cells mature into osteoblasts, the cells that build bone. When they artificially increased ADAR2 in healthy stem cells, these cells switched on bone-related genes and boosted their ability to form mineralized bone matrix. Conversely, dialing down ADAR2 in healthy osteoblasts weakened key bone markers and reduced their bone-forming activity, showing that ADAR2 is important for normal bone building.
Low ADAR2 Marks More Aggressive Tumors
The researchers then turned to osteosarcoma samples and cell lines. Across patient datasets, tumors had far less ADAR2 than healthy bone marrow stem cells, and patients whose tumors showed particularly low ADAR2 had poorer survival and more metastases. In a mouse model in which osteosarcoma cells spread from bone to lung, the migrating cells also had markedly lower ADAR2 levels than those remaining in the bone. Together, these findings suggest that losing this molecular editor is linked with more aggressive disease and worse clinical outcomes.
Forcing Cancer Cells to Grow Up
To test whether restoring ADAR2 could tame osteosarcoma, the team engineered two human osteosarcoma cell lines to overproduce either active ADAR2, an inactive mutant version, or a control. Cells with active ADAR2 divided more slowly, paused longer in a resting phase of the cell cycle, and invaded surrounding tissue less readily. In one cell line capable of partial bone-like behavior, active ADAR2 strongly enhanced the production of mineralized nodules and switched on genes associated with fully mature bone cells, while reducing markers linked to stemness and malignancy. Importantly, when these engineered cells were implanted into the leg bones of immunodeficient mice, tumors formed by ADAR2-overexpressing cells were smaller and less invasive, and the mice developed fewer liver and lung metastases. The inactive mutant did not provide this protection, indicating that ADAR2’s editing activity is crucial.
Editing a Growth Switch in Cancer Cells
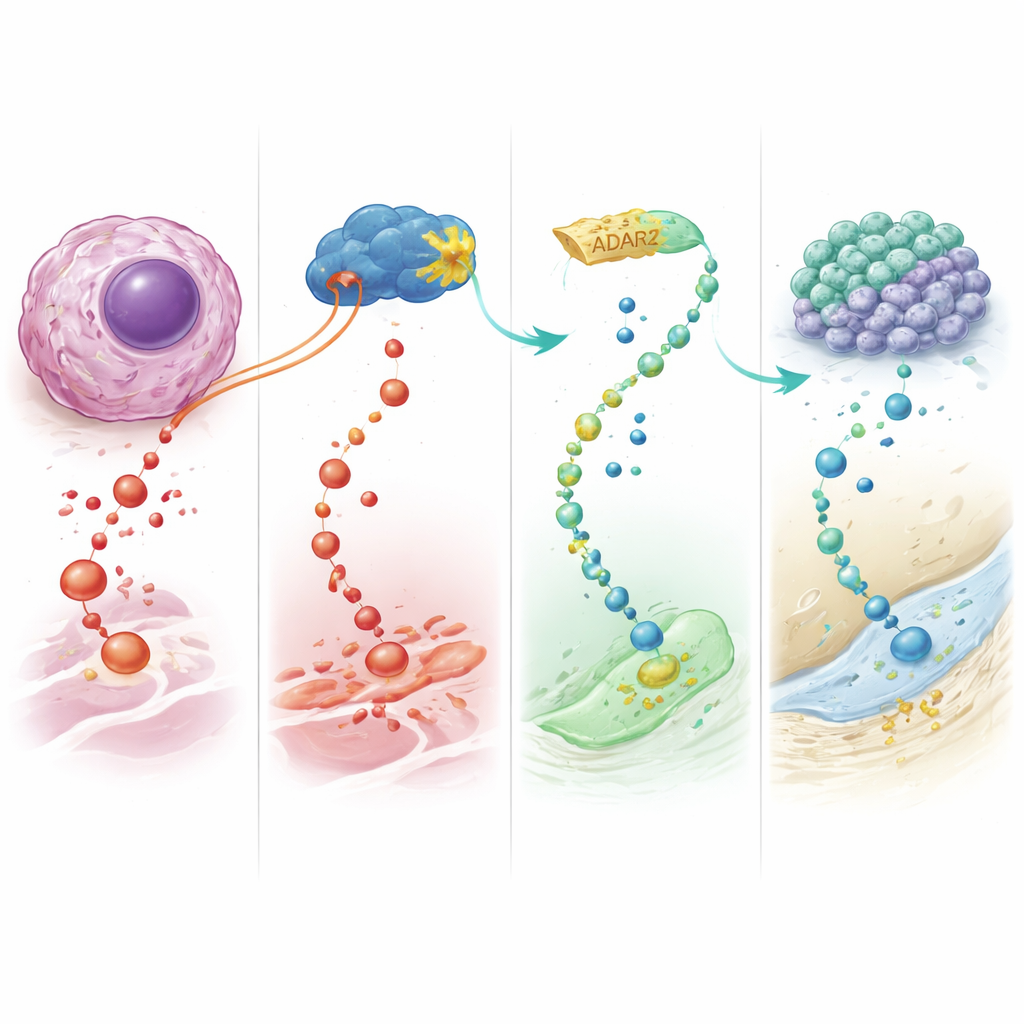
To uncover how ADAR2 exerts these effects, the team used RNA sequencing to map both gene expression changes and editing events. They found hundreds of genes altered in ways that fit with reduced malignancy and better bone specialization. Most strikingly, ADAR2 heavily edited a gene called IGFBP7, which encodes a protein that can boost growth signals through the IGF1 receptor, a pathway known to promote tumor cell survival and proliferation. ADAR2 converted a single amino acid within IGFBP7, producing a slightly different protein variant. In osteosarcoma cells, this edited form was linked to weaker activation of the IGF1 signaling chain and to lower activity of downstream growth drivers. When cells were treated with the unedited IGFBP7 protein, growth pathways lit up, cells proliferated faster, and a key bone transcription factor decreased. By contrast, the edited form failed to stimulate growth, instead enhancing the bone-related factor and favoring differentiation over division.
Reframing Therapy as Reprogramming
In essence, this work reveals ADAR2 as a natural tumor suppressor in osteosarcoma that works by rewriting specific RNA messages, especially those for IGFBP7. Boosting ADAR2 steers malignant bone cells away from uncontrolled growth and toward terminal bone-like differentiation, while also making them more sensitive to existing chemotherapy drugs. For patients, the long-term hope is a new class of "differentiation therapies" that reprogram, rather than simply destroy, cancer cells—potentially improving outcomes in a childhood cancer where better options are urgently needed.
Citation: Rossi, M., Scotto di Carlo, F., Di Gregorio, J. et al. ADAR2 induces the differentiation of osteosarcoma cells by editing activity on IGFBP7: new implications for therapy. Bone Res 14, 38 (2026). https://doi.org/10.1038/s41413-026-00516-6
Keywords: osteosarcoma, RNA editing, ADAR2, bone differentiation, IGFBP7