Clear Sky Science · en
Osteoblastic sclerostin loop3-LRP4 interaction required by sclerostin to inhibit bone formation
Why Bone-Building Needs a Smarter Switch
Fragile bones and fractures are a growing concern as people live longer, and powerful new drugs now exist to stimulate bone growth. Yet one such medicine, an antibody that blocks a bone-inhibiting protein called sclerostin, has been tied to serious heart problems in some patients. This study unpacks the fine details of how sclerostin slows bone formation and reveals a way to switch off its brake in bone cells while preserving its protective roles in the cardiovascular system.
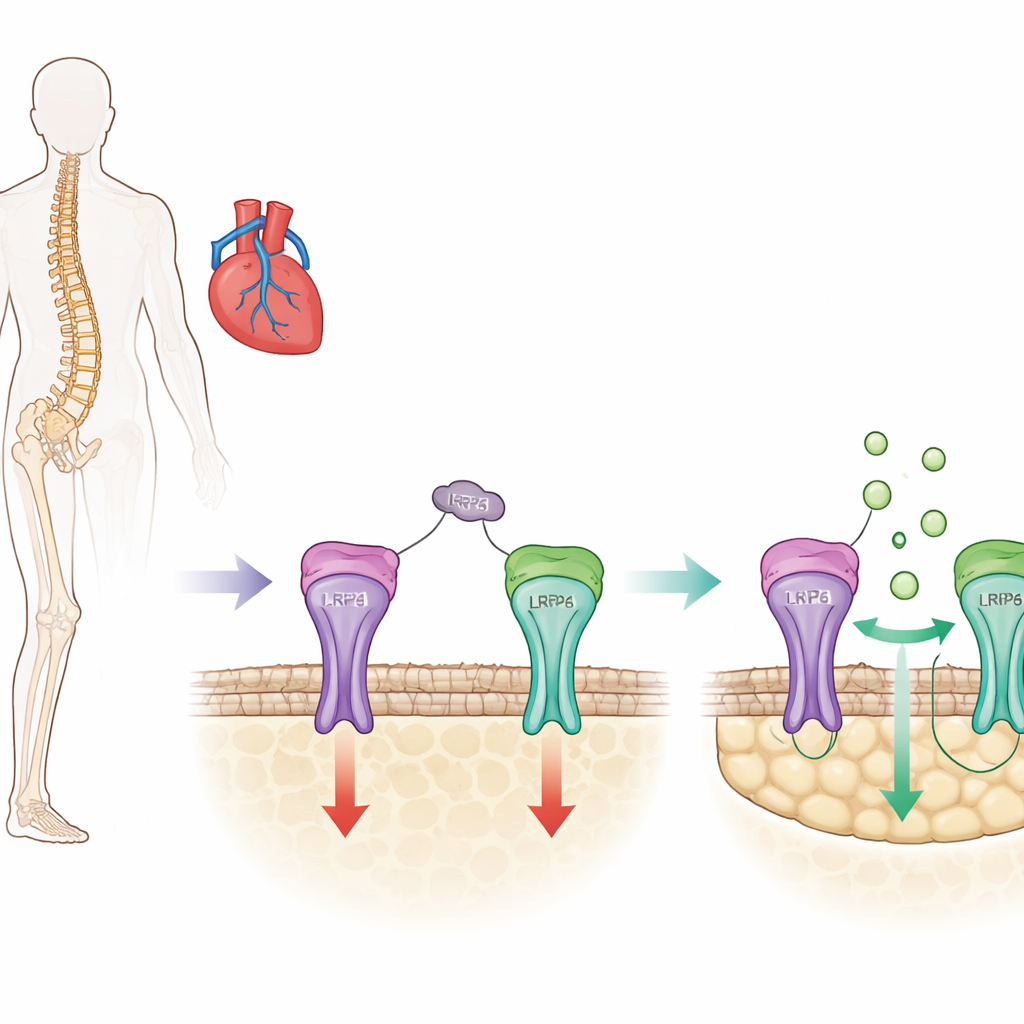
A Key Bone Brake with a Hidden Side
Bone is constantly renewed as specialized cells build new tissue while others remove old material. Sclerostin is a protein made mainly by bone cells that acts as a brake on this building process by dampening a growth-promoting pathway known as Wnt/β-catenin signaling in bone-forming cells (osteoblasts). Current osteoporosis drugs such as romosozumab work by blocking sclerostin, lifting this brake and rapidly increasing bone mass. However, clinical trials have suggested that broad sclerostin blockade may also disturb protective functions in blood vessels and the heart, raising the risk of heart attack and stroke in some individuals.
Zooming In on a Tiny Loop
Sclerostin has three small loop-shaped regions in its structure, and previous work from this group showed that one of them, called loop3, helps sclerostin suppress bone formation but is not needed for its protective effects in blood vessels. In the present study, the researchers asked exactly how loop3 contributes to bone inhibition. They discovered that loop3 binds to a receptor on the bone cell surface called LRP4. This binding acts like an anchor that positions sclerostin so it can reach another nearby receptor, LRP6, which directly controls the Wnt/β-catenin pathway. Using biochemical binding tests and computer modeling, they pinpointed specific amino acids on the LRP4 protein that are essential contact points for loop3.
Breaking the Anchor to Free Bone Growth
Armed with this structural insight, the team engineered two types of tools to disrupt the loop3–LRP4 anchor: a genetic version that subtly altered the key amino acids in LRP4 (called Lrp4m), and a short synthetic peptide (LRP4-Pep) that mimics the loop3-binding region of LRP4 and competes for sclerostin. In cultured osteoblasts, both strategies weakened the ability of sclerostin to bind LRP6, reduced its suppression of Wnt/β-catenin signaling, and preserved the cells’ ability to mature and deposit mineral. Computer simulations supported this picture, showing that normal LRP4 holds sclerostin close to LRP6, while the mutated form fails to stabilize this contact.
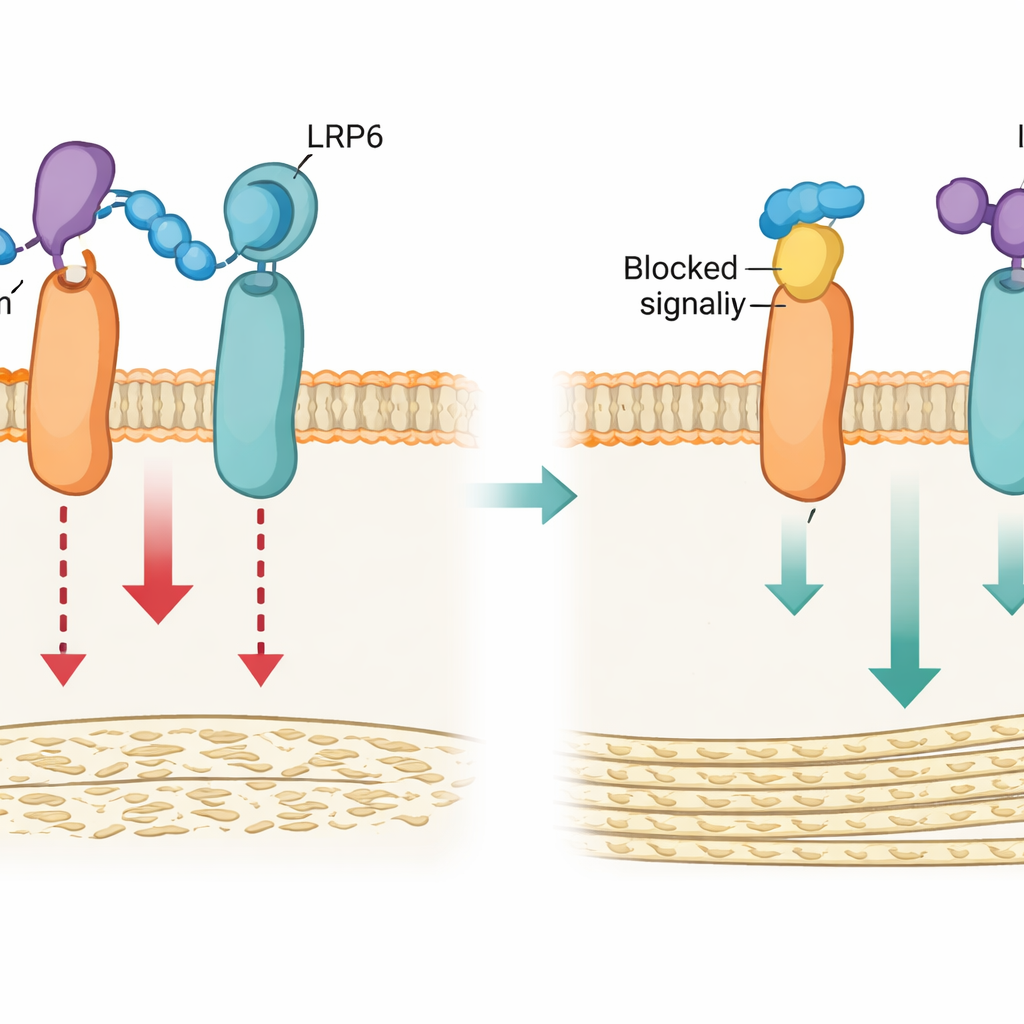
Stronger Bones in Mice Without Extra Risks
The researchers then moved into mouse models to confirm that disrupting the loop3–LRP4 interaction improves bone health in living animals. Mice carrying the Lrp4m mutation throughout the body developed denser, stronger bones, with improved internal architecture and higher blood levels of bone-formation markers, and without measurable harm to muscle. When the same mutation was present in mice that genetically lack sclerostin, there was no added effect, but reintroducing sclerostin showed that its bone-suppressing power was much weaker in Lrp4m animals. Likewise, in mice engineered to overproduce sclerostin or in mice with established estrogen-deficiency–related bone loss, repeated treatment with the LRP4-Pep peptide dose-dependently increased bone mass, improved bone quality, and enhanced mechanical strength, yet had no detectable impact when sclerostin was absent.
Toward Safer Bone-Anabolic Therapies
Taken together, these findings show that an interaction between sclerostin loop3 and LRP4 in osteoblasts is the anchor that sclerostin needs in order to latch onto LRP6 and shut down bone formation. By selectively disrupting this anchor—either through precise mutations or a tailored peptide—the researchers could boost bone building and strength in mice without directly blocking the parts of sclerostin that seem important for cardiovascular protection. For patients, this work points to a new generation of bone-strengthening treatments that aim not to silence sclerostin entirely, but to uncouple its effects in bone from its beneficial roles in the heart and blood vessels.
Citation: Wang, L., Tao, X., Jiang, H. et al. Osteoblastic sclerostin loop3-LRP4 interaction required by sclerostin to inhibit bone formation. Bone Res 14, 45 (2026). https://doi.org/10.1038/s41413-026-00511-x
Keywords: osteoporosis, sclerostin, bone formation, LRP4, Wnt signaling