Clear Sky Science · en
Tumor-derived DNA drives cancer-associated anemia by promoting reticulocyte clearance
Why Cancer-Related Anemia Matters
Anemia is a common and exhausting problem for people with cancer. When red blood cells are too few or too weak, patients feel tired, short of breath, and less able to tolerate treatment. Doctors have long known that tumors somehow disturb the body’s red blood cell balance, but the exact reasons have been murky. This study uncovers a surprising culprit: loose pieces of DNA that escape from tumors into the bloodstream and directly damage young red blood cells, offering a fresh way to understand and possibly treat cancer-associated anemia.
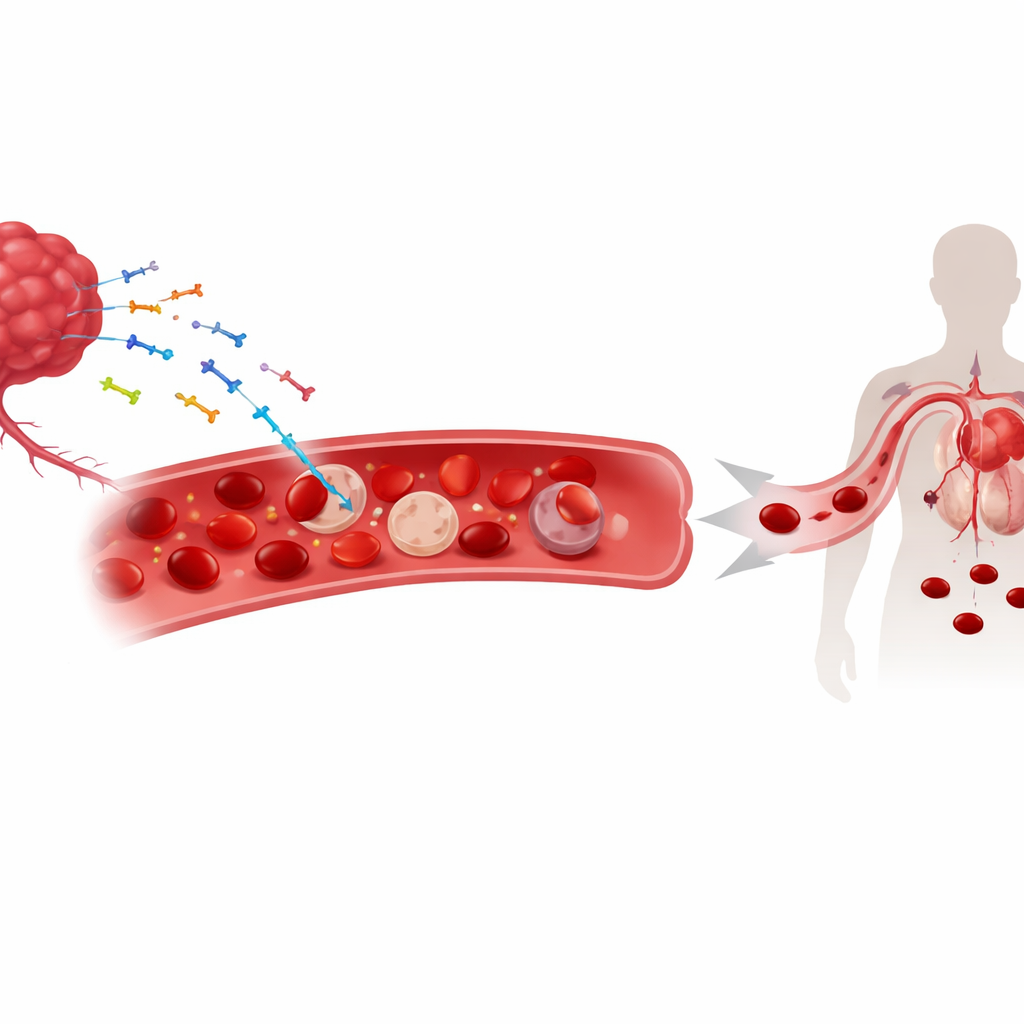
Stray Tumor DNA in the Bloodstream
As tumors grow, they shed fragments of their genetic material—both nuclear and mitochondrial DNA—into the circulation. These bits of “cell-free” DNA are already used as biomarkers to track cancer. The researchers found that a substantial portion of this tumor-derived DNA does not just float in plasma; instead, it sticks to the surface of red blood cells. In mouse models of lung, skin, and colon cancers and in patients with several solid tumors, higher amounts of red blood cell–bound DNA closely tracked with lower hemoglobin levels and more severe anemia. This link did not appear in non-cancer forms of anemia, suggesting that DNA binding is a distinctive feature of the cancer setting rather than a general consequence of low red blood cell counts.
Young Red Cells Take the Hit
Red blood cells are born as immature forms called reticulocytes, which circulate briefly while they finish maturing. The team discovered that tumor-derived DNA selectively targets these young cells. Using fluorescent dyes and microscopy in mice and cancer patients, they showed that over 95 percent of DNA-positive red cells carried markers of immaturity. These DNA-coated reticulocytes had abnormal shape, roughened membranes, and reduced hemoglobin and heme content, all signs that their development had been derailed. When the bound DNA was removed with enzymes or washed off with special solutions, the cells regained more normal features and advanced further along their maturation pathway.
From Surface Damage to Premature Clearance
Why does DNA on the cell surface matter so much? The study shows that it turns young red cells into prime targets for destruction. Reticulocytes bound to DNA displayed hallmarks of “eryptosis,” a programmed death process for red cells, including calcium surges inside the cell and exposure of “eat me” signals on the membrane. In laboratory and animal experiments, spleen and liver macrophages—immune cells that clear aged or damaged red cells—preferentially engulfed DNA-coated reticulocytes over normal ones. Treating red cells or tumor-bearing mice with the DNA-cutting enzyme DNase I stripped away the bound DNA and sharply reduced this accelerated clearance, allowing red cells to persist longer in circulation.
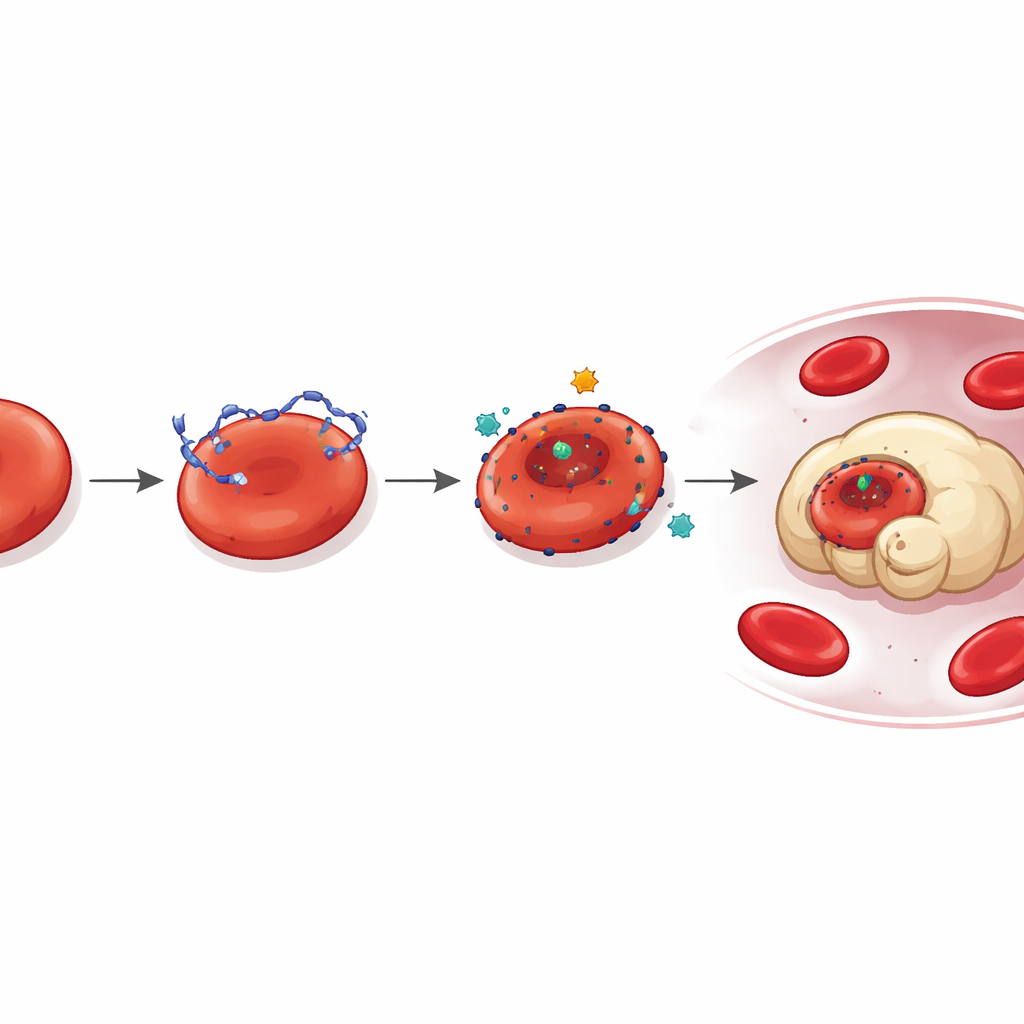
A Hidden Docking Site for DNA
Digging deeper, the researchers asked how DNA latches onto reticulocytes in the first place. Proteomic analysis pointed to LONP1, a protein normally found in mitochondria, as a key player. In tumor-bearing mice and cancer patients, LONP1 became mislocalized to the outer surface of circulating reticulocytes. There it acted as a high-affinity docking site for tumor-derived DNA. Cells with surface LONP1 bound more DNA, showed greater deformity, and were swallowed more rapidly by macrophages. Blocking LONP1’s activity or reducing its levels cut down DNA binding and the resulting damage, revealing a tumor-induced pathway in which mispositioned LONP1 and stray DNA together sabotage young red cells.
Turning a Harmful Signal into a Treatment Target
Because many current treatments for cancer-related anemia focus only on making more red blood cells, they can be blunt tools that carry risks such as blood clots or unwanted effects on the immune system. This work suggests a more precise approach: prevent tumor-derived DNA from wrecking reticulocytes in the first place. In mice, DNase I therapy alone raised hemoglobin levels without speeding tumor growth by clearing away harmful DNA from red cell surfaces. When combined with erythropoietin, a hormone that stimulates red blood cell production, DNase I produced even stronger, synergistic improvements: more red cells were made, and fewer were destroyed prematurely. For patients, this DNA-centered view of cancer-associated anemia opens the door to therapies that simultaneously restore red blood cell balance and avoid undermining anti-tumor immunity.
Citation: Yuan, T., Liu, X., Wu, Y. et al. Tumor-derived DNA drives cancer-associated anemia by promoting reticulocyte clearance. Sig Transduct Target Ther 11, 152 (2026). https://doi.org/10.1038/s41392-026-02674-9
Keywords: cancer-associated anemia, tumor-derived DNA, reticulocytes, red blood cell clearance, DNase I therapy