Clear Sky Science · en
Bioengineered iPSC-derived human macrophages with increased angiotensin-converting enzyme (ACE) expression suppress solid tumor growth
Turning Our Own Defenders Against Cancer
Cancer immunotherapy has changed the outlook for some patients, but many tumors still find ways to hide from or wear down the immune system. This study explores a new way to supercharge one of the body’s natural defenders—macrophages—by reprogramming them in the lab so they become far more aggressive toward solid tumors. The work suggests a future in which doctors might infuse patients with tailor‑made immune cells that attack a broad range of cancers without needing to recognize a single specific tumor marker.
Why Macrophages Matter in Cancer
Macrophages are white blood cells that act as both garbage collectors and frontline soldiers. Inside many solid tumors, they are plentiful—but often in the wrong mode. Instead of attacking, they slip into a “healer” state that actually helps tumors grow, spread, and evade other immune cells. The authors reasoned that if they could reliably push macrophages into a hard‑hitting, inflammatory state and keep them there inside the tumor, these cells might both kill cancer cells directly and rally other immune fighters such as T cells and natural killer (NK) cells.
Borrowing a Blood Pressure Enzyme for Immune Power
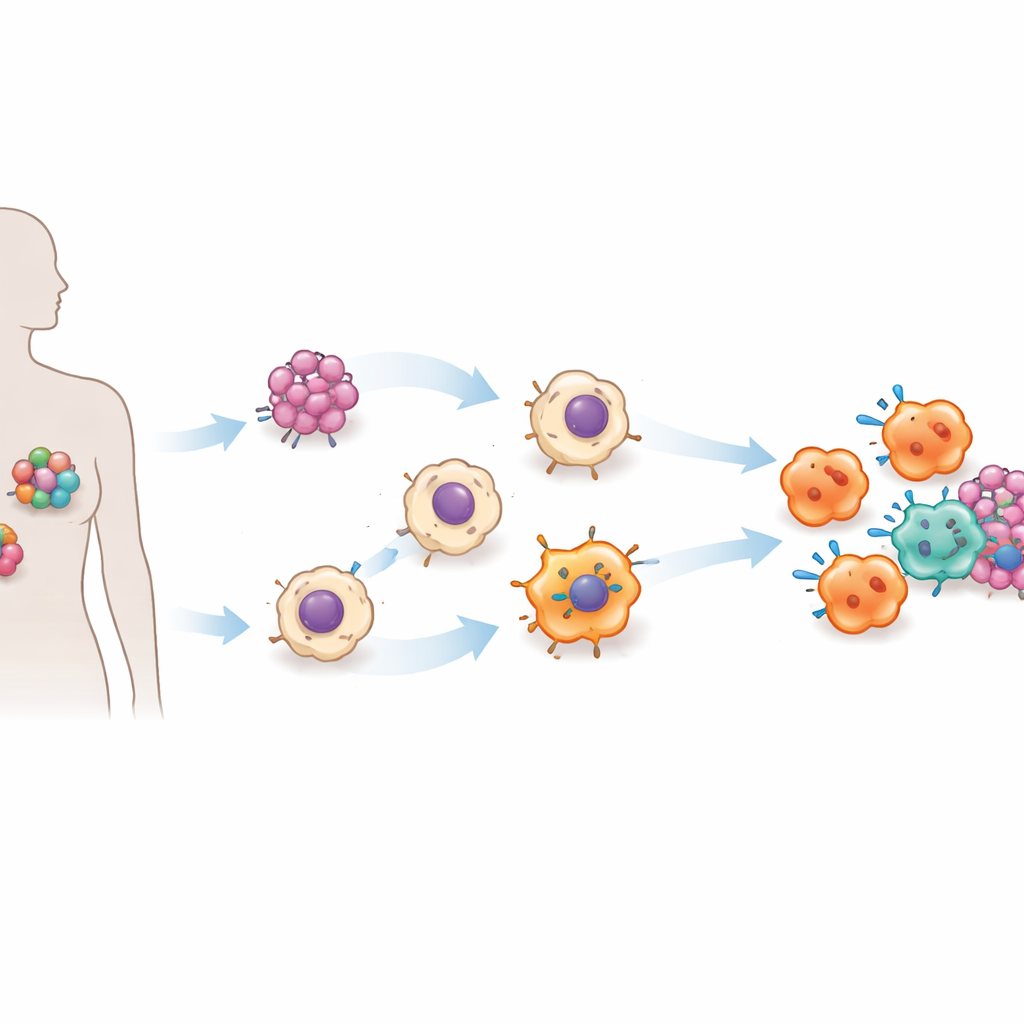
The team focused on angiotensin‑converting enzyme (ACE), best known for its role in controlling blood pressure and as the target of common heart medications. Earlier work in mice hinted that higher ACE levels in myeloid cells, including macrophages, make these cells better at fighting infections and tumors. In this study, the researchers brought that idea into a human system. They used induced pluripotent stem cells—adult cells reprogrammed back into a stem‑like state—and inserted a genetic switch that lets them dial ACE levels up or down with the common antibiotic doxycycline. From these engineered stem cells, they produced large numbers of macrophages on demand, a scalable platform they call ACE‑iMac.
Building Supercharged Macrophages in the Lab
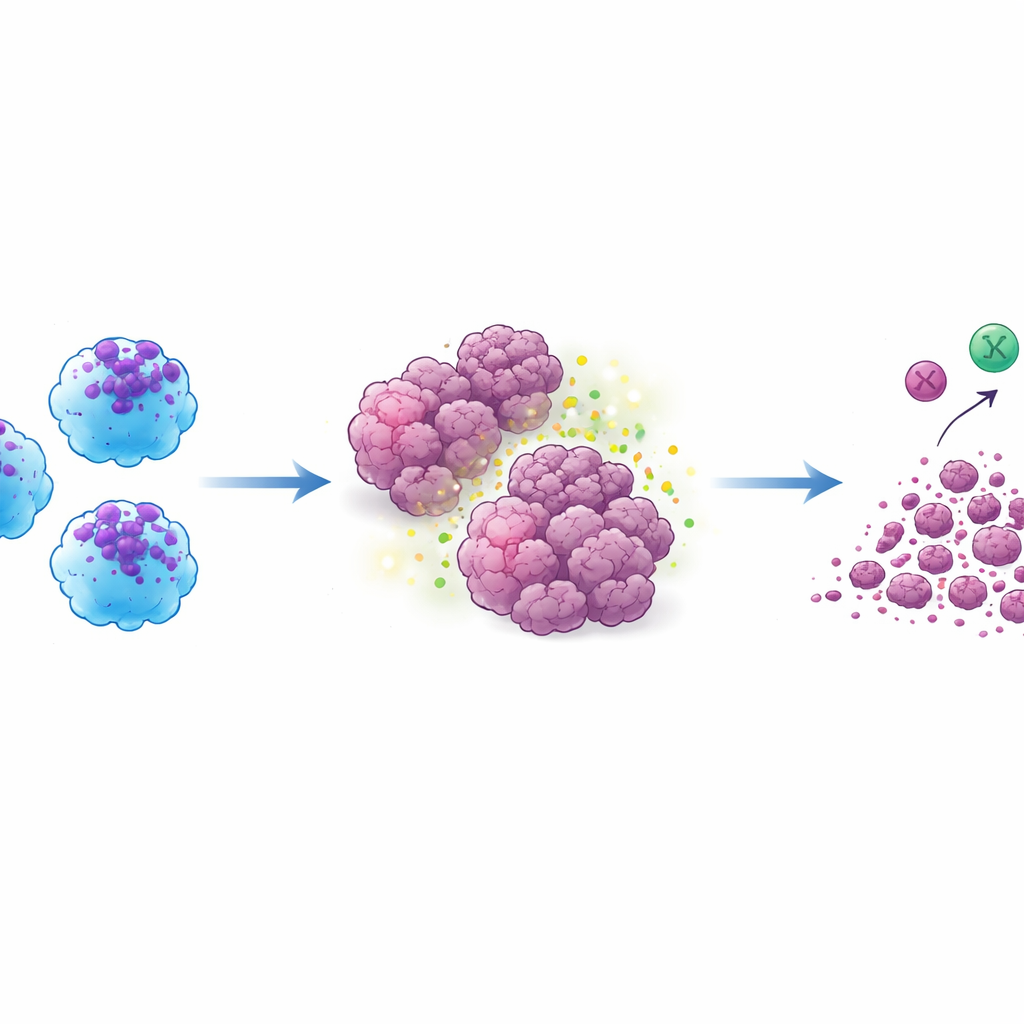
When the ACE switch was turned on, the resulting ACE‑iMac cells looked and behaved very differently from regular lab‑grown macrophages. They released much higher amounts of inflammatory signals, generated large bursts of reactive oxygen and nitric oxide—chemical weapons that damage tumor cells—and showed gene activity patterns associated with a highly activated, “M1‑like” state. At the same time, they reduced production of molecules linked to a more suppressive, tumor‑aiding mode. In culture dishes, ACE‑iMac cells slowed the growth of melanoma, triple‑negative breast cancer, and drug‑resistant head and neck cancer cells far more effectively than control macrophages.
Stopping Tumors and Waking Up Other Immune Cells
The crucial test came in mouse models bearing human tumors. When ACE‑iMac cells were injected directly into solid tumors, those tumors shrank dramatically—by roughly three‑quarters or more compared with tumors receiving ordinary macrophages. This held true across melanoma, breast, and head and neck cancer models. Inside the tumors, ACE‑iMac cells showed strong inflammatory signatures and were accompanied by more active NK cells and, in specially humanized mice with a functional human immune system, more potent human killer T cells. Interestingly, the overall number of these other immune cells did not change much; instead, they became more activated, suggesting that ACE‑iMac cells act as on‑site coaches that energize the broader immune response rather than simply adding more fighters.
Decoding the Inner Workings
To understand what drives this behavior, the researchers examined global gene activity in ACE‑iMac cells. Turning on ACE boosted networks linked to classical macrophage activation, cytokine production, antigen processing, and the assembly of cellular “stomachs” called phagosomes that digest targets. It also tuned pathways that help macrophages present fragments of tumor proteins to T cells, which may explain the stronger T‑cell response in humanized mice. Under tumor‑like stimulation, ACE‑iMac cells showed further gains in genes involved in metabolism and differentiation, changes that likely help them stay active in the harsh tumor environment instead of becoming exhausted or switching sides.
What This Could Mean for Future Cancer Care
To a non‑specialist, the bottom line is that the authors have built a controllable, renewable source of human macrophages that behave like highly focused tumor hunters and immune boosters. By raising ACE levels only when needed using an external drug, they can, in principle, tune the strength and timing of this response, which may improve safety. While many steps remain before patient use—including testing intravenous delivery, long‑term safety, and combinations with existing therapies—this work points to a new class of cell therapy. Rather than targeting one tumor marker at a time, ACE‑iMac cells aim to broadly reset the immune landscape inside solid tumors, potentially making cancer both more visible and more vulnerable to the body’s own defenses.
Citation: Shibata, T., Bhat, S., Cao, D. et al. Bioengineered iPSC-derived human macrophages with increased angiotensin-converting enzyme (ACE) expression suppress solid tumor growth. Sig Transduct Target Ther 11, 132 (2026). https://doi.org/10.1038/s41392-026-02650-3
Keywords: cancer immunotherapy, macrophages, induced pluripotent stem cells, angiotensin-converting enzyme, solid tumors