Clear Sky Science · en
The impact of K-Ras Gly12 mutants on homeostasis and tumorigenesis in the colonic epithelium
Why tiny changes in a cancer gene matter
Colon cancer is one of the most common cancers, and many cases are driven by damage to a gene called KRAS. Doctors have long known that patients whose tumors carry KRAS mutations often respond poorly to treatment, but not all KRAS changes are alike. This study asks a deceptively simple question with big implications for precision medicine: do three very common versions of mutant KRAS behave differently in the colon, and can those differences be turned into more effective, tailored treatments?
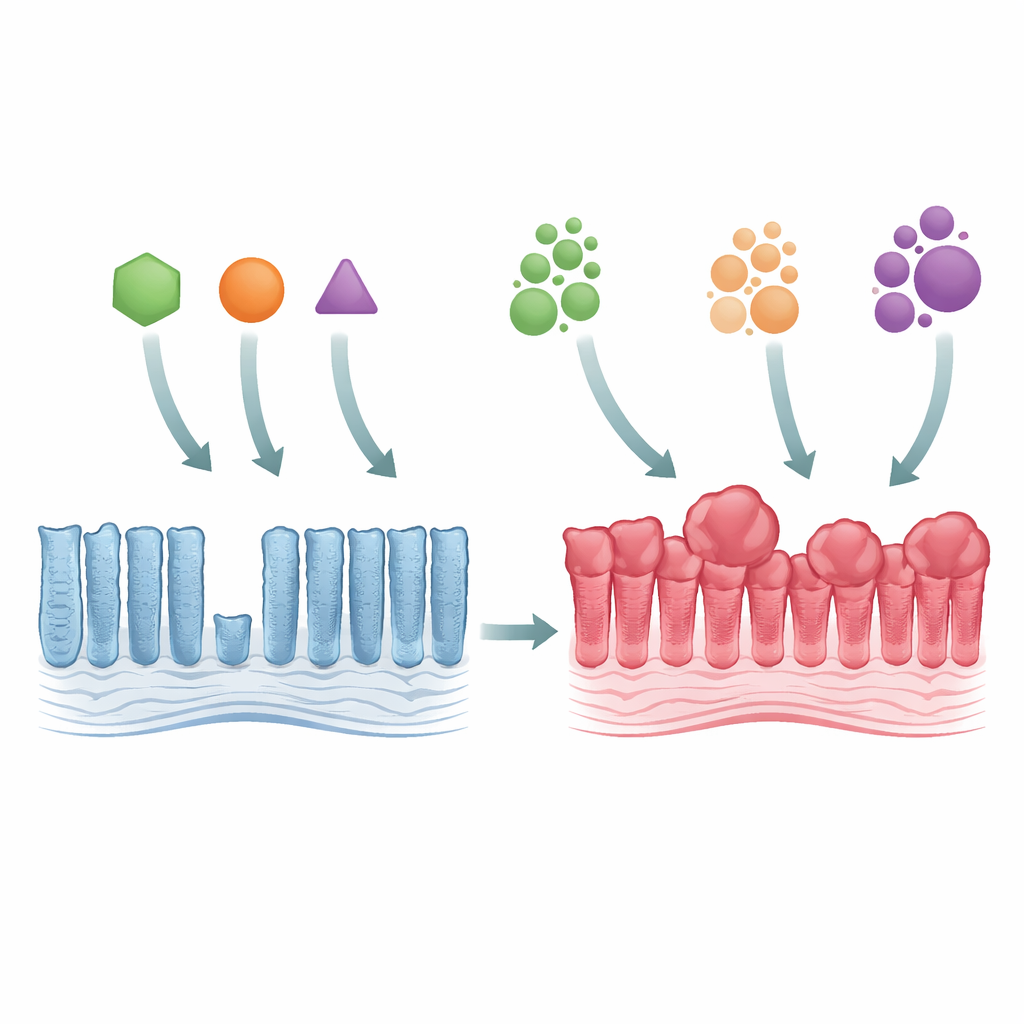
From normal gut lining to early tumor growth
The researchers used genetically engineered mice to mimic three major KRAS mutations, all affecting the same building block in the protein (called Gly12) but changing it in different ways (G12C, G12D, and G12V). When any of these mutants were switched on in the colon lining, the tissue became thicker and more crowded, with elongated glands and an overload of mature cells. Despite this overgrowth, the actual zone where cells were dividing did not expand much, suggesting that the problem was not faster cell birth but slower cell turnover. All three mutants strongly activated a growth-control pathway known as MAPK, and they wiped out certain specialized gut cells, showing that even subtle genetic changes can broadly disturb the intestine’s internal balance.
Same-looking tumors, very different outcomes
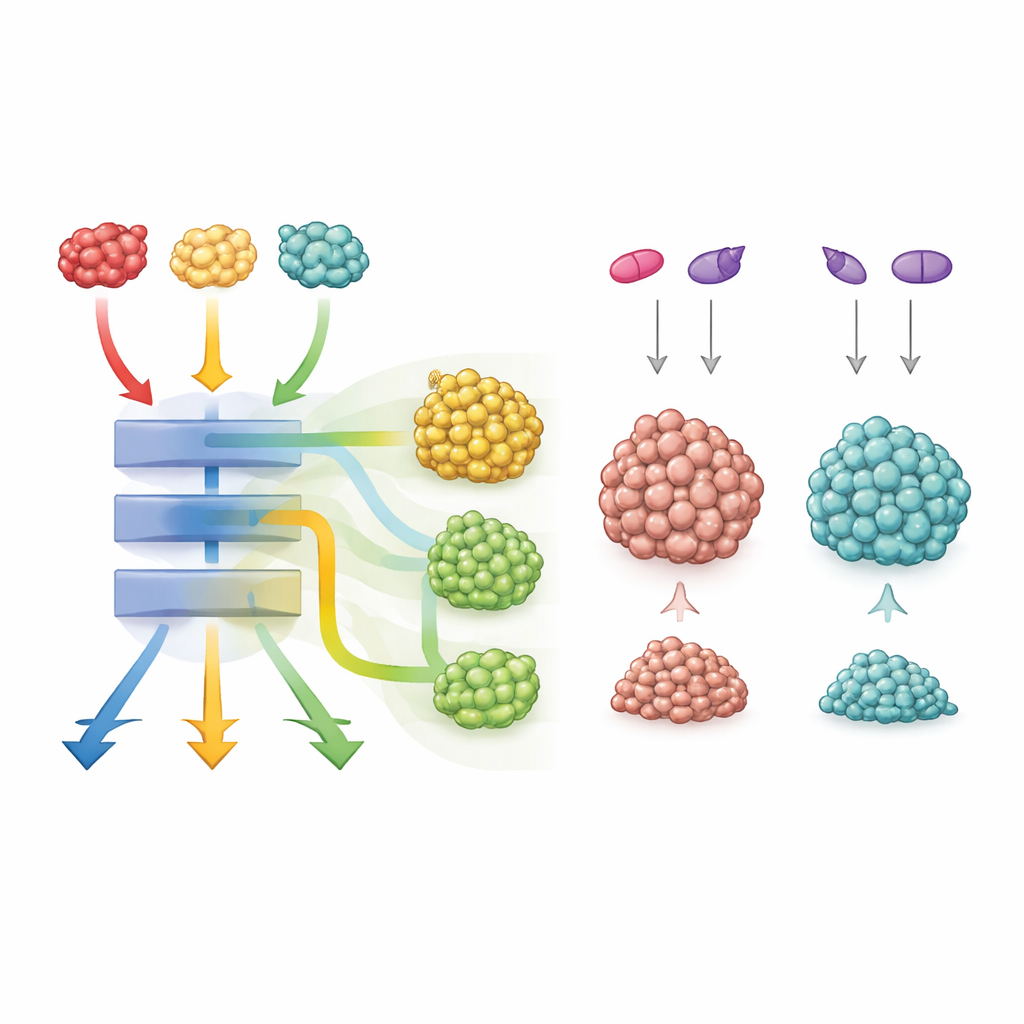
To study cancer formation, the team combined each KRAS mutant with loss of the tumor suppressor gene APC, a common early event in human colon cancer. The resulting tumors all looked similar under the microscope and showed comparable activation of the core growth pathway driven by KRAS. Yet the mice did not fare the same. Animals with the G12D mutation developed the most tumors and had the shortest survival, those with G12V were intermediate, and those with G12C had fewer tumors and lived longer. Mini-tumors grown in the lab from these mice (organoids) also showed distinct responses to new KRAS-blocking drugs: G12D tumors were hardest to kill, G12V the most sensitive, and G12C fell in between and responded differently depending on the drug’s mode of action.
Shared growth programs across mice and humans
To understand what these mutations were doing inside cells, the researchers measured global gene activity and protein levels in both mouse tumors and human colon cancer datasets. They found a common KRAS mutant “signature” across species: genes that push cell division and support the MAPK growth pathway were switched on, while many energy metabolism genes were dialed down, consistent with cancer cells rewiring how they use fuel. Several genes and proteins that act in feedback loops—either amplifying or trying to restrain KRAS signaling—were consistently elevated. The team also saw stronger activity in pathways linked to inflammation and immune signals, hinting that these mutations may help shape a tumor-friendly microenvironment early in disease.
A special vulnerability in one mutant
Despite this shared core program, one mutant, G12C, stood out. Tumors carrying G12C showed extra activation of developmental pathways such as Wnt/β-catenin and Notch, which influence how cells decide their fates. On their own, drugs that block these pathways had similar effects across all mutants. But when the researchers combined a G12C-specific KRAS inhibitor with blockers of Wnt or Notch, organoids with the G12C mutation shrank dramatically, far more than those with G12D or G12V. This suggests that G12C-driven tumors lean especially heavily on these additional pathways and can be pushed over the edge when both KRAS and its helper routes are targeted together.
What this means for future cancer care
Overall, the study shows that three KRAS mutations at the very same position can produce colon tumors that look alike but behave quite differently in growth, survival, and drug response. All share a powerful core engine that drives cell division and metabolic rewiring, yet each mutant fine-tunes additional signaling networks in its own way. For patients, this means that simply knowing a tumor is “KRAS-mutant” is not enough. Understanding exactly which KRAS alteration is present, and which partner pathways it engages, could guide smarter combinations of targeted drugs—especially for the G12C mutant, where pairing KRAS inhibitors with Wnt or Notch blockers may offer a new path to better treatments.
Citation: Yang, M.H., Sheth, S., Shui, B. et al. The impact of K-Ras Gly12 mutants on homeostasis and tumorigenesis in the colonic epithelium. Oncogene 45, 1828–1839 (2026). https://doi.org/10.1038/s41388-026-03771-3
Keywords: KRAS mutations, colon cancer, allele-specific therapy, tumor signaling, combination targeting