Clear Sky Science · en
LINC-AC092535.5 regulates MICAL2 mRNA level to inhibit p53-mediated ferroptosis in nasopharyngeal carcinoma
Why this study matters
Nasopharyngeal carcinoma is a cancer that arises behind the nose and above the throat, and it remains difficult to cure once it returns or spreads. This study uncovers a hidden molecular "brake" that helps these tumor cells dodge a newly recognized form of cell death called ferroptosis, which relies on iron and oxidative damage. By exposing this brake, the researchers point to fresh strategies for making stubborn nasopharyngeal tumors more vulnerable to treatment.
A different way for cancer cells to die
Most cancer drugs try to kill tumor cells by triggering well-known death programs such as apoptosis. Tumors, however, often learn to evade these routes. Ferroptosis is a distinct type of cell death driven by iron and bursts of reactive oxygen molecules that destroy cell membranes, particularly in mitochondria, the cell’s power stations. Many cancers that resist standard therapies can still be pushed into ferroptosis. Nasopharyngeal carcinoma cells are particularly sensitive to drugs that spark this process, suggesting ferroptosis could be turned into a therapeutic advantage—if scientists can understand what holds it back.
A protein that shields tumors from iron-driven damage
Searching large genetic databases and tumor samples from patients, the team homed in on a protein called MICAL2, previously linked to cancer growth and oxidative chemistry. They found that MICAL2 levels are markedly higher in nasopharyngeal tumors than in normal tissue, and patients with more MICAL2 tend to have more advanced disease, more metastases, and poorer survival. When the researchers reduced MICAL2 levels in nasopharyngeal cancer cell lines, the cells grew and spread less readily, and they became far more sensitive to ferroptosis-inducing drugs such as Erastin and Sorafenib. Measures of iron, oxidative stress, and antioxidant balance all shifted in a way consistent with heightened ferroptosis, both in cultured cells and in mouse tumor models.
A newly discovered RNA that turns the shield up
The investigators then asked why MICAL2 is so abundant in these tumors. They discovered a previously uncharacterized long non-coding RNA, named LINC-AC092535.5, that is strongly elevated in head and neck cancers and parallels MICAL2 levels. Unlike RNAs that encode proteins, this RNA acts as a regulator. It physically binds MICAL2’s message molecule and boosts its production in two ways: it enhances activity of the MICAL2 gene’s promoter, and it slows the breakdown of MICAL2 messenger RNA. When LINC-AC092535.5 was silenced, MICAL2 levels dropped, ferroptosis markers increased, and tumor cells became more vulnerable to iron-driven oxidative injury. Restoring MICAL2 could partially undo these effects, showing that this RNA and MICAL2 function together as a reinforcing circuit that protects cancer cells from ferroptosis.
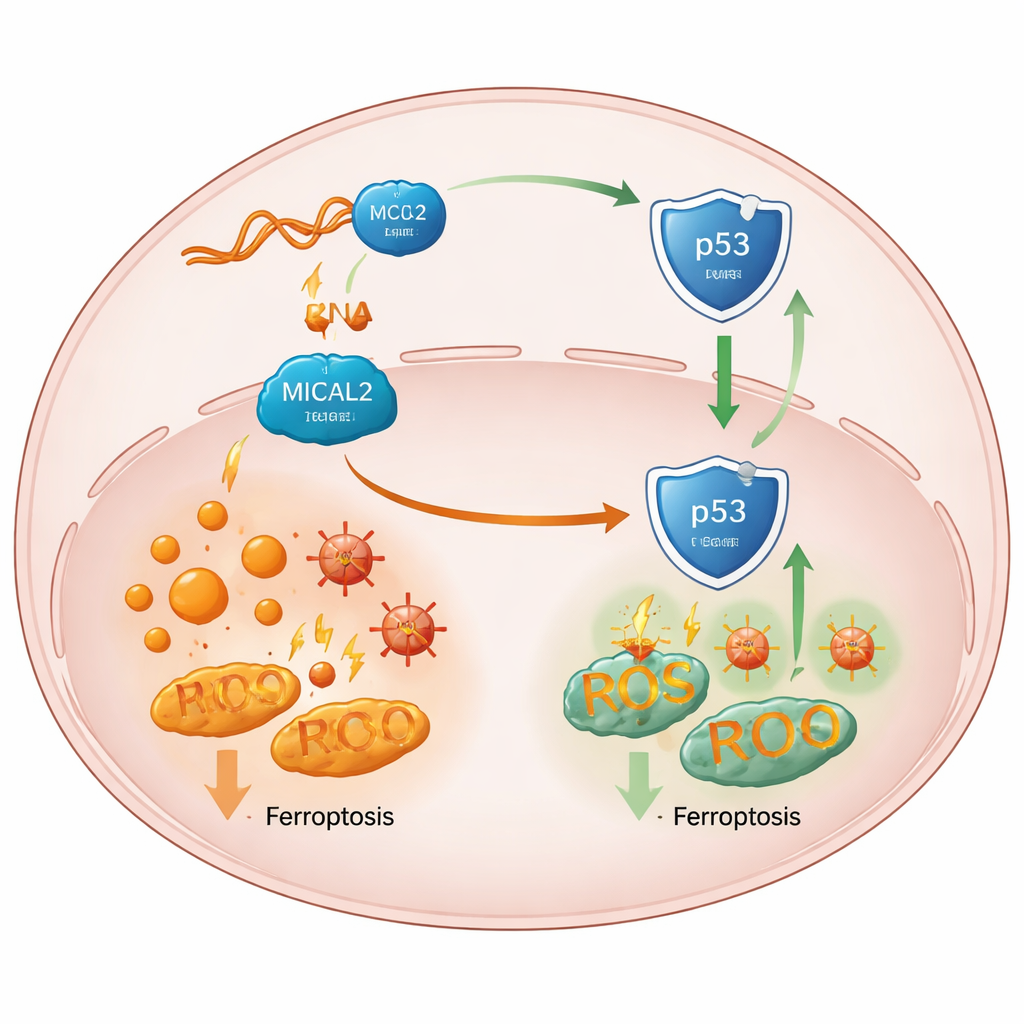
How the shield weakens a key tumor guardian
Digging deeper, the team traced the effect of MICAL2 to p53, a central “guardian” protein that normally helps prevent tumors and can encourage ferroptosis in stressed cells. High MICAL2 activity dampened p53’s pathway, while loss of MICAL2 stabilized and increased p53 protein without changing its gene’s activity. The researchers showed that MICAL2 physically associates with p53 and with MDM2, a protein that tags p53 for destruction. MICAL2 helps MDM2 attach more of these tags, sending p53 to the cellular waste system and shuttling it out of the nucleus, where it would otherwise turn on protective genes. When MICAL2 or LINC-AC092535.5 were knocked down, p53 accumulated in the nucleus, its downstream signals shifted toward ferroptosis, and cancer cells across several tumor types showed more iron-driven death—unless p53 itself was removed.
What this means for future treatments
Taken together, the work outlines a new control axis in nasopharyngeal carcinoma: the RNA LINC-AC092535.5 boosts MICAL2, and MICAL2 in turn helps strip away p53’s protective influence, allowing tumor cells to resist ferroptosis. Interrupting this axis—by inhibiting the RNA, blocking MICAL2, or preventing p53 degradation—could restore iron-driven cell death and make existing ferroptosis-inducing drugs more effective. For patients with relapsed or treatment-resistant nasopharyngeal cancer, such combinations may one day offer a way to turn the cancer’s own iron and oxidative stress against it.
Citation: Zhang, S., Chen, W., Yang, J. et al. LINC-AC092535.5 regulates MICAL2 mRNA level to inhibit p53-mediated ferroptosis in nasopharyngeal carcinoma. Oncogene 45, 1260–1274 (2026). https://doi.org/10.1038/s41388-026-03714-y
Keywords: nasopharyngeal carcinoma, ferroptosis, p53, long noncoding RNA, MICAL2