Clear Sky Science · en
Antimicrobial peptide-induced inner membrane hyperpolarization is associated with antibiotic sensitization and attenuated MIC escalation in multidrug-resistant Gram-negative pathogens
Why this research matters for everyday health
Deadly infections are becoming harder to treat as bacteria learn to resist our strongest antibiotics. This study explores a new way to help existing drugs work again by adding a small, naturally inspired molecule called a peptide. The work suggests we may not need brand‑new antibiotics for every threat; instead, we can "supercharge" the ones we already have while also calming the runaway inflammation that makes sepsis so dangerous.
A helper molecule for struggling antibiotics
The researchers focused on a modified fish‑derived molecule named TP2‑5, a positively charged peptide that by itself can kill some bacteria. They asked whether using very small amounts of this peptide alongside standard antibiotics could make tough, multidrug‑resistant germs easier to treat. Working with problem strains of Gram‑negative bacteria such as drug‑resistant Escherichia coli and Acinetobacter baumannii, they found that adding only one‑quarter of the minimum TP2‑5 dose needed to inhibit growth allowed several different antibiotics, including meropenem, colistin, ciprofloxacin and even usually ineffective vancomycin, to work far better. In lab tests, combinations that used weak doses of both drugs together could completely clear bacteria that either drug alone could not control.
Keeping resistance from climbing so fast
Antibiotic resistance usually worsens over time when bacteria are repeatedly exposed to a drug. To mimic this process, the team grew resistant E. coli for three weeks under daily exposure to either antibiotics alone, TP2‑5 alone, or antibiotics plus low‑dose TP2‑5. When antibiotics were used by themselves, the amount needed to stop bacterial growth rose dramatically—up to 128‑fold for some drugs—signaling strong new resistance. When the same antibiotics were paired with TP2‑5, this escalation was much milder, suggesting that the helper peptide can slow the bacteria’s ability to adapt. Meanwhile, bacteria exposed to TP2‑5 alone showed little change in their sensitivity to the peptide, hinting that it may be harder for them to evolve strong defenses against this kind of membrane‑targeting attack.
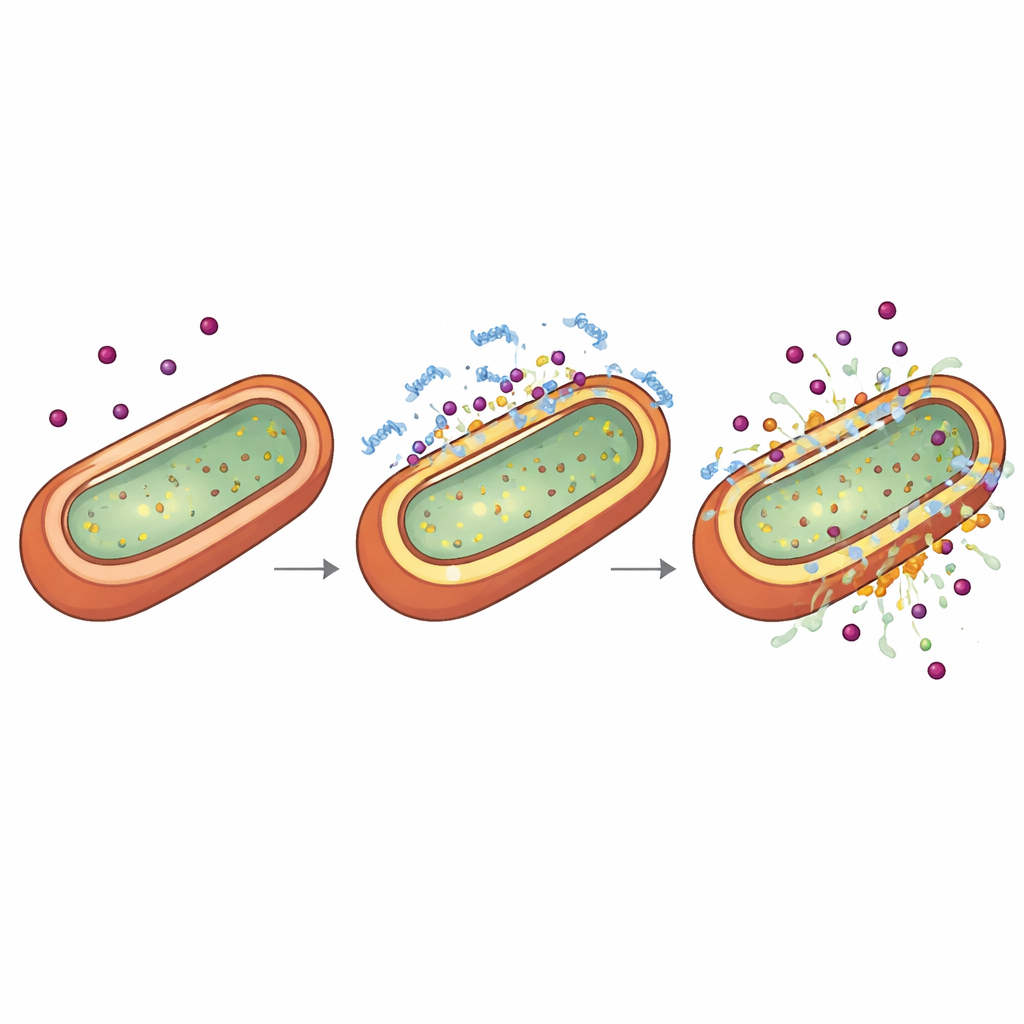
How the peptide reshapes the bacterial shell
To understand why TP2‑5 boosts antibiotic power, the scientists examined the outer shell of Gram‑negative bacteria, which acts as both armor and gatekeeper. Using chemical dyes that light up when membranes leak or change voltage, along with cryo‑electron tomography that images frozen cells in three dimensions, they showed that very low, non‑lethal doses of TP2‑5 subtly thin and puncture the outer layer while putting the inner layer into an unusually energized or “hyperpolarized” state. At higher doses, the same peptide tears the membranes apart. Timing experiments revealed that when bacteria are first put into this hyperpolarized, slightly leaky condition by TP2‑5, later exposure to antibiotics rapidly collapses their inner membrane and kills them. The peptide prefers to bind to negatively charged components called lipopolysaccharides and specific phospholipids, which are abundant in Gram‑negative envelopes, explaining its selectivity for these pathogens.
Quieting the body’s dangerous overreaction
Severe infections do not just depend on the microbe; the body’s own immune response often spirals out of control, leading to sepsis and organ failure. Because TP2‑5 sticks to bacterial surface molecules that trigger inflammation, the team tested whether it could also dampen immune overdrive. In mouse immune cells, TP2‑5 blocked the binding of these bacterial fragments, blunted key signaling pathways, and broadly reduced the activation of genes that drive inflammation. In animal models, including a caterpillar infection system, a drug‑resistant pneumonia model, and a standard mouse sepsis surgery, TP2‑5 either alone or combined with antibiotics cut bacterial counts, lowered markers of organ injury, reduced inflammatory messengers in the blood, and, strikingly, produced 100% survival in the polymicrobial sepsis model, where standard antibiotics helped but did not fully protect.
Balancing power and safety
Any new anti‑infective must do more harm to microbes than to patients. TP2‑5 showed this type of selectivity in cultured human and mouse cells, where harmful effects appeared only at concentrations many times higher than those needed to block bacteria. In mice, both intravenous and lung‑directed dosing caused no meaningful weight loss, organ damage, or blood cell abnormalities at levels relevant for treatment. The peptide tended to accumulate mainly in the liver and was gradually cleared over several days, supporting the idea that it can be given at adjuvant, helper doses without obvious toxicity.
What this means for future treatments
This work presents TP2‑5 as a “membrane‑sensitizing” partner for existing antibiotics: it loosens up the protective coat of Gram‑negative superbugs in a special energized state that makes them easier to kill, while also mopping up inflammatory triggers that can push patients into deadly sepsis. Although the study did not prove that the unusual hyperpolarized state is the direct cause of better drug activity, the strong link in timing and effect suggests it is an important piece of the puzzle. If similar peptides can be safely used in humans, they could extend the useful life of our current antibiotics and offer a two‑pronged approach—hitting the bacteria and calming the body’s response—that is especially valuable for hard‑to‑treat, multidrug‑resistant infections.
Citation: Yeh, JC., Hazam, P.K., Lin, YY. et al. Antimicrobial peptide-induced inner membrane hyperpolarization is associated with antibiotic sensitization and attenuated MIC escalation in multidrug-resistant Gram-negative pathogens. npj Antimicrob Resist 4, 33 (2026). https://doi.org/10.1038/s44259-026-00210-x
Keywords: antimicrobial peptides, antibiotic adjuvants, multidrug-resistant bacteria, sepsis, Gram-negative infections