Clear Sky Science · en
Mechanistic insights into plasmid transfer inhibition in Enterobacterales by nucleoside analogues
Why tiny DNA rings matter to us
Antibiotic resistance usually brings to mind overused drugs and stubborn infections, but another, less visible player helps these superbugs spread: small DNA rings called plasmids that jump between bacteria. This study asks a practical question with big implications: can medicines we already use for viral infections quietly slow, or even speed up, the spread of antibiotic resistance genes between gut bacteria? By probing how certain antiviral-like compounds affect plasmid transfer in two common hospital pathogens, the researchers uncover new ways to block resistance from moving through bacterial communities — and also warn that some drugs might accidentally make the problem worse. 
How resistance genes hitch a ride
Many of the most worrying antibiotic resistance genes in Escherichia coli and Klebsiella pneumoniae sit on plasmids, which can copy themselves and pass directly from one bacterium to another in a process called conjugation. This DNA hand-off helps resistance race through hospital wards and the human gut, even when bacteria are not under heavy antibiotic pressure. Because traditional antibiotics kill or slow bacteria but do not stop this gene swapping, scientists have begun to look for “anti-plasmid” compounds that leave bacterial growth largely intact while cutting the lines along which resistance travels.
Old antiviral drugs, new roles
The team tested 14 clinically approved nucleoside analogues — small molecules originally designed to disrupt viral or human DNA and RNA — to see whether they changed how often plasmids moved between bacteria. Using fluorescent tags and flow cytometry, they tracked transfer of one plasmid that carries genes for extended-spectrum β-lactamase (which breaks down many penicillin-like drugs) in E. coli, and another plasmid that carries a carbapenemase gene in K. pneumoniae. Several compounds, including azidothymidine (AZT), didanosine, stavudine, and trifluridine, clearly reduced plasmid transfer without slowing bacterial growth. Surprisingly, other nucleoside analogues, such as famciclovir, zalcitabine, aciclovir, and valaciclovir, actually boosted plasmid sharing in at least one of the species tested. That means some medicines given for viral infections could, in principle, nudge resistance genes to spread more easily in the gut microbiome.
What changes inside the bacterial cell
To understand how these compounds work, the researchers looked at several basic features of bacterial physiology. Most of the drugs did not damage cell membranes, disrupt electrical potential, or trigger bursts of reactive oxygen, and they left overall growth unchanged. Some of the plasmid-promoting compounds modestly lowered levels of ATP, the cell’s main energy currency, hinting that energy balance and the cost of running the plasmid-transfer machinery may be linked. For the strongest inhibitor, AZT, the team turned to RNA sequencing, which reads out which genes are turned up or down when bacteria encounter the drug. 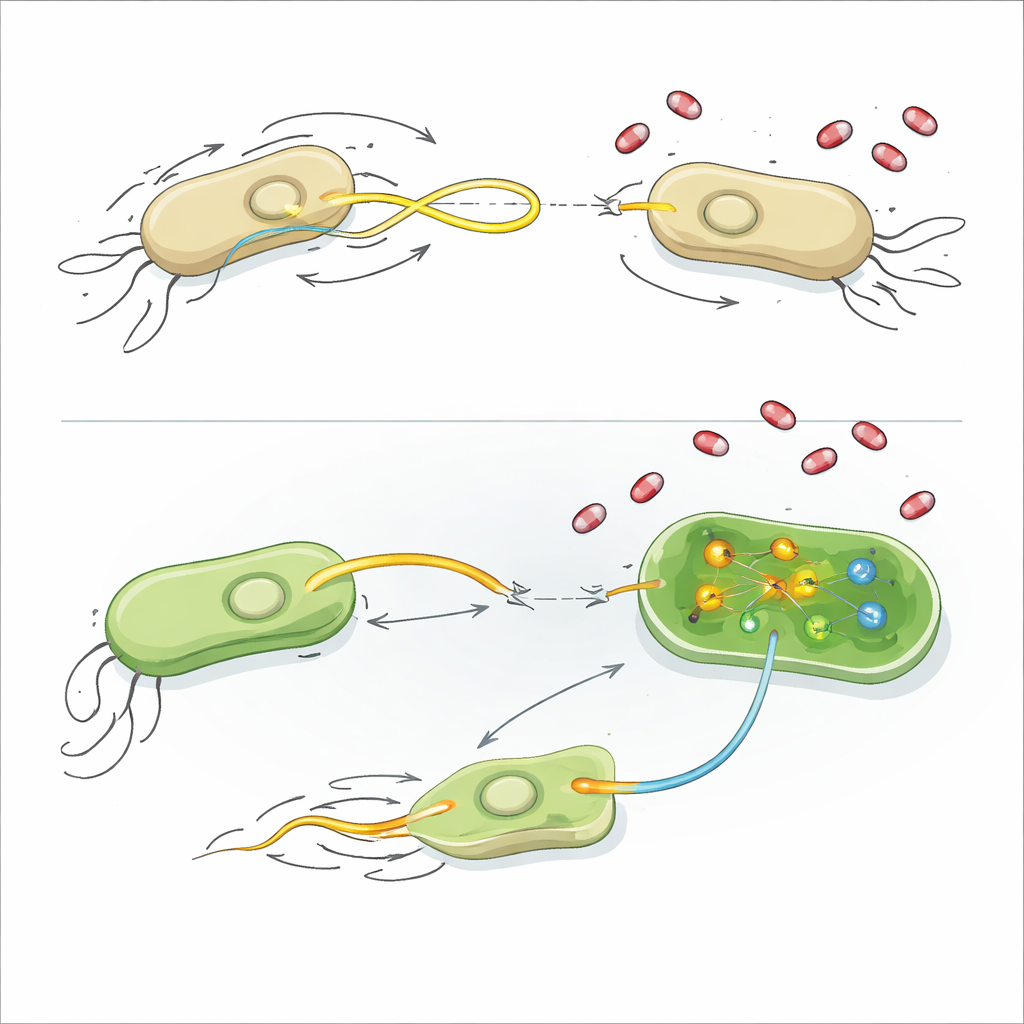
Slowing swimming and blocking repair
In E. coli, AZT sharply dialed down genes that build and control the flagellum, the rotating tail that lets cells swim. When researchers placed E. coli on soft agar, AZT almost completely stopped them from spreading outward, even though the bacteria were not being killed. Genetically disabling a key flagellar part had a similar effect on cutting plasmid transfer, and adding AZT on top of that caused no further drop — strong evidence that reduced motility alone can explain much of the inhibition. In K. pneumoniae, AZT took a different route: it switched on genes involved in DNA damage repair, including a central SOS-response protein called RecA, and switched off genes needed to make the amino acid methionine and its derivative S-adenosylmethionine (SAM), a universal methyl donor. Adding zinc acetate, which dampens RecA activity, or supplying extra SAM both restored plasmid transfer in the presence of AZT, indicating that heightened DNA stress and disrupted methylation help block incoming plasmids.
What this means for fighting resistance
This work shows that some existing nucleoside analogues can act as templates for a new kind of drug: agents that disarm the spread of resistance rather than killing bacteria outright. AZT, in particular, curbs plasmid conjugation in two major pathogens through different levers — stopping bacterial swimming in one case and reshaping DNA repair and basic metabolism in the other. At the same time, the discovery that other analogues increase plasmid transfer highlights the need to understand how non-antibiotic drugs influence the gut’s microbial gene traffic. Ultimately, fine-tuned plasmid transfer inhibitors could become part of our toolkit to slow the march of antimicrobial resistance, buying time for existing antibiotics and new treatments to work.
Citation: Alav, I., Ashraf, A., Pordelkhaki, P. et al. Mechanistic insights into plasmid transfer inhibition in Enterobacterales by nucleoside analogues. npj Antimicrob Resist 4, 23 (2026). https://doi.org/10.1038/s44259-026-00197-5
Keywords: antimicrobial resistance, plasmid transfer, nucleoside analogues, Escherichia coli, Klebsiella pneumoniae