Clear Sky Science · en
Breaking the ‘rule-of-five’ to access bridged bicyclic heteroaromatic bioisosteres
Why three dimensional pills matter
Many modern medicines are built from flat ring shaped molecules that fit into protein targets like keys into locks. Chemists have learned that swapping these flat pieces for small three dimensional frameworks can sometimes make drugs more soluble, longer lasting in the body and less likely to break down in unwanted ways. This article explores a new way to build such 3D building blocks that could give drug designers fresh options when tuning how a medicine behaves.
From flat rings to tiny frameworks
Drug molecules often contain nitrogen bearing rings, which are common in approved medicines but can suffer from issues such as rapid metabolism. In recent years, compact cage like structures called bridged bicycles have emerged as "stand ins" for simple benzene rings, offering a more three dimensional shape while keeping the spacing of key atoms similar. However, close relatives that mimic nitrogen containing rings have been much harder to make, especially versions in which a nitrogen atom sits inside a strained four membered ring. These rare frameworks, known as 6 azabicyclo[3.1.1]heptanes, are predicted to have appealing properties but have lacked general synthetic routes.
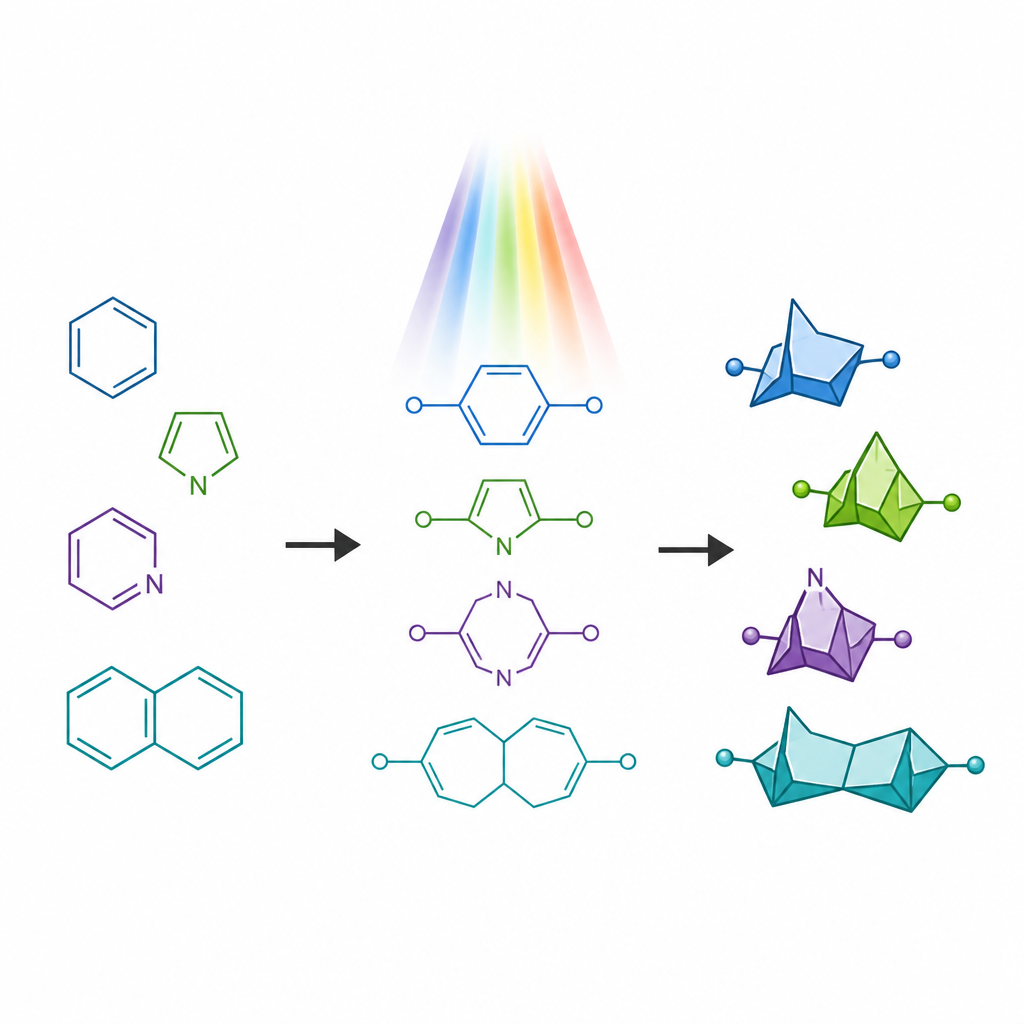
Breaking an old rule of ring building
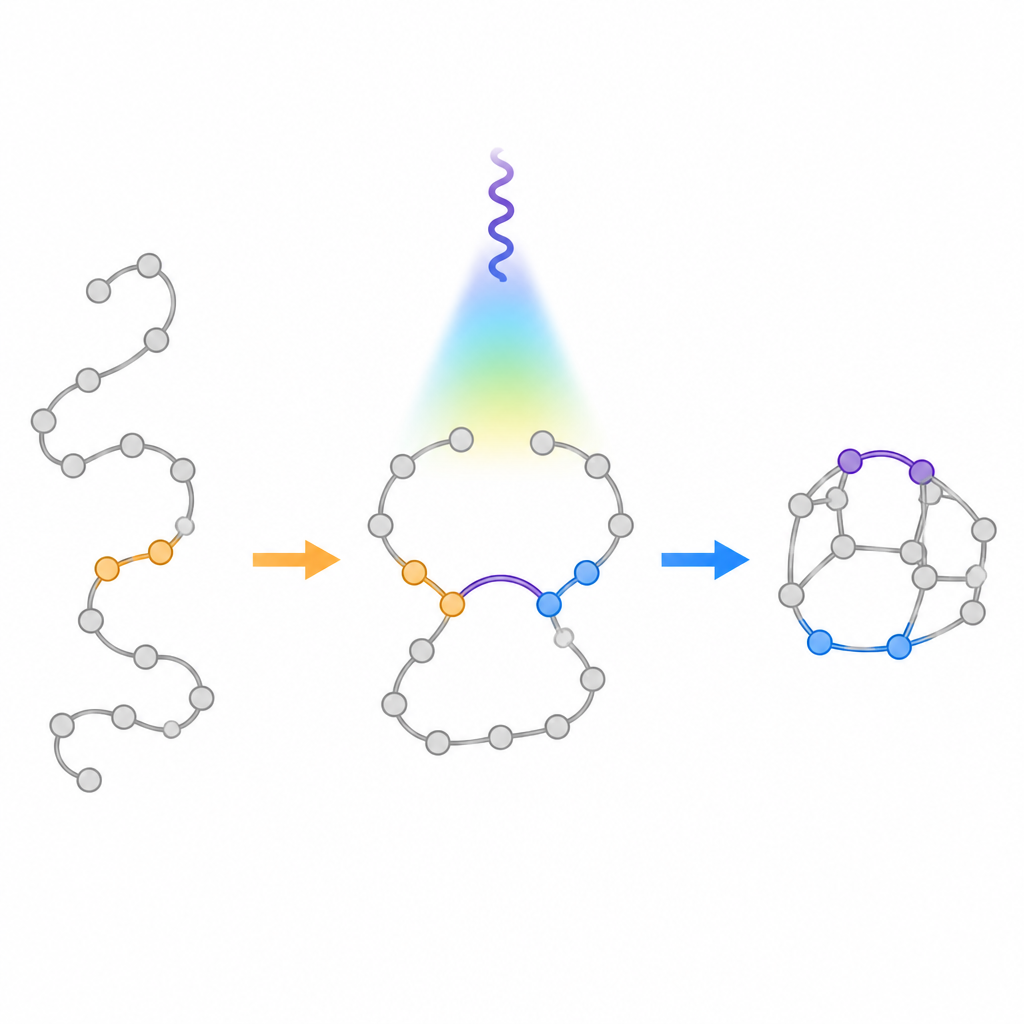
Traditional light driven methods for turning flexible chains into rings tend to obey an old guideline called the "rule of five", which favors the formation of five membered rings over six membered ones. When chemists shine light on certain chain like molecules with two double bonds, they usually fold up to give smaller fused rings rather than the larger bridged cages desired here. The authors set out to deliberately overturn this bias. They designed special starting materials called aza 1,6 dienes that contain both a carbon carbon double bond and a carbon nitrogen double bond linked in the same chain. By placing a group that stabilizes radicals at a specific position on the chain, they aimed to slow down the usual five membered ring pathway and instead steer the molecule toward the six membered bridged cage.
Using gentle light to guide the change
The team developed a visible light process that uses a photocatalyst to transfer energy to the aza 1,6 dienes without changing their charge. Once excited, these molecules briefly form a biradical intermediate in which two reactive sites can close in different ways. Computer calculations suggested that attaching a phenyl ring at the key position would stabilize one of these radicals and raise the barrier for the unwanted five membered closure. Experiments confirmed this design: under blue light in the presence of an iridium based sensitizer, the chains reliably folded into the targeted bridged bicycles, with only small amounts of the fused by products. The reaction worked across many different attached rings, including fragments taken from real drugs, and could be scaled to gram quantities.
Building a toolbox from the new cages
Once the new 6 nitrogen bridged frameworks were in hand, the authors showed that they can be further modified in many directions. One versatile product bearing a vinyl group was transformed into alcohols, acids, amines and other useful handles through standard steps like hydrogenation, ozonolysis and oxidation. In parallel, the nitrogen oxygen bond within the cage could be reduced to reveal a secondary amine, which was then dressed with ureas, sulfonamides, carbamates and amides. This rich follow up chemistry means the rigid core can support a wide variety of side chains, a key requirement for exploring structure activity relationships in drug discovery.
Testing the new shapes in a drug like setting
To probe how these cages might perform in a biological context, the researchers built an analogue of a known enzyme inhibitor, swapping a flexible piperazine ring for a new 3,6 diazabicyclo[3.1.1]heptane core. Measurements in human liver microsomes showed that the modified molecule had slightly higher molecular weight but lower lipophilicity, a change often linked to better solubility, while retaining similar metabolic stability to the original compound. These early results suggest that the new frameworks can stand in for common nitrogen rings without harming key drug like properties.
What this means for future medicines
In simple terms, the study shows how to coax light excited molecules into breaking an old preference for smaller rings and instead form compact three dimensional cages that closely mimic familiar nitrogen containing rings. Because these new shapes can be readily decorated and appear metabolically robust, they offer medicinal chemists fresh pieces to plug into drug designs when adjusting solubility, stability and shape. While more biological testing is needed, this anti rule of five strategy opens up new chemical space that may help future medicines work better and last longer in the body.
Citation: Zhang, ZX., Shu, K., Tilby, M.J. et al. Breaking the ‘rule-of-five’ to access bridged bicyclic heteroaromatic bioisosteres. Nat. Synth 5, 790–797 (2026). https://doi.org/10.1038/s44160-026-00990-0
Keywords: photochemistry, bioisostere, drug design, heterocycles, visible light catalysis