Clear Sky Science · en
Rare genetic diseases associated with G-quadruplex-induced replication stress
When DNA Knots Lead to Rare Diseases
Our genetic material is usually pictured as a smooth, twisting ladder. But in many places, DNA can fold into compact knots called G‑quadruplexes. These unusual shapes are especially common in important control regions of our genome and near chromosome ends. This review explains how such knots can slow or stall the copying of DNA, creating “replication stress” that damages chromosomes and contributes to a surprising range of rare, inherited diseases that often involve growth problems, immune defects, premature aging, and cancer risk.
Unusual DNA Shapes in Our Genome
G‑quadruplexes form in stretches of DNA rich in the letter G (guanine). Instead of pairing in the usual way, four guanines can stack into flat “plates” that pile up into a stable column. Modern mapping techniques show hundreds of thousands of these potential sites scattered across the human genome, especially in gene switches (promoters), untranslated regions of RNA, and the protective caps on chromosomes called telomeres. Cells also contain proteins that recognize or stabilize these structures. Together, this suggests that G‑quadruplexes are not rare curiosities but recurring features that help shape how genes are turned on, how chemical marks on DNA are placed, and how telomeres are maintained.
When DNA Copying Runs into a Roadblock
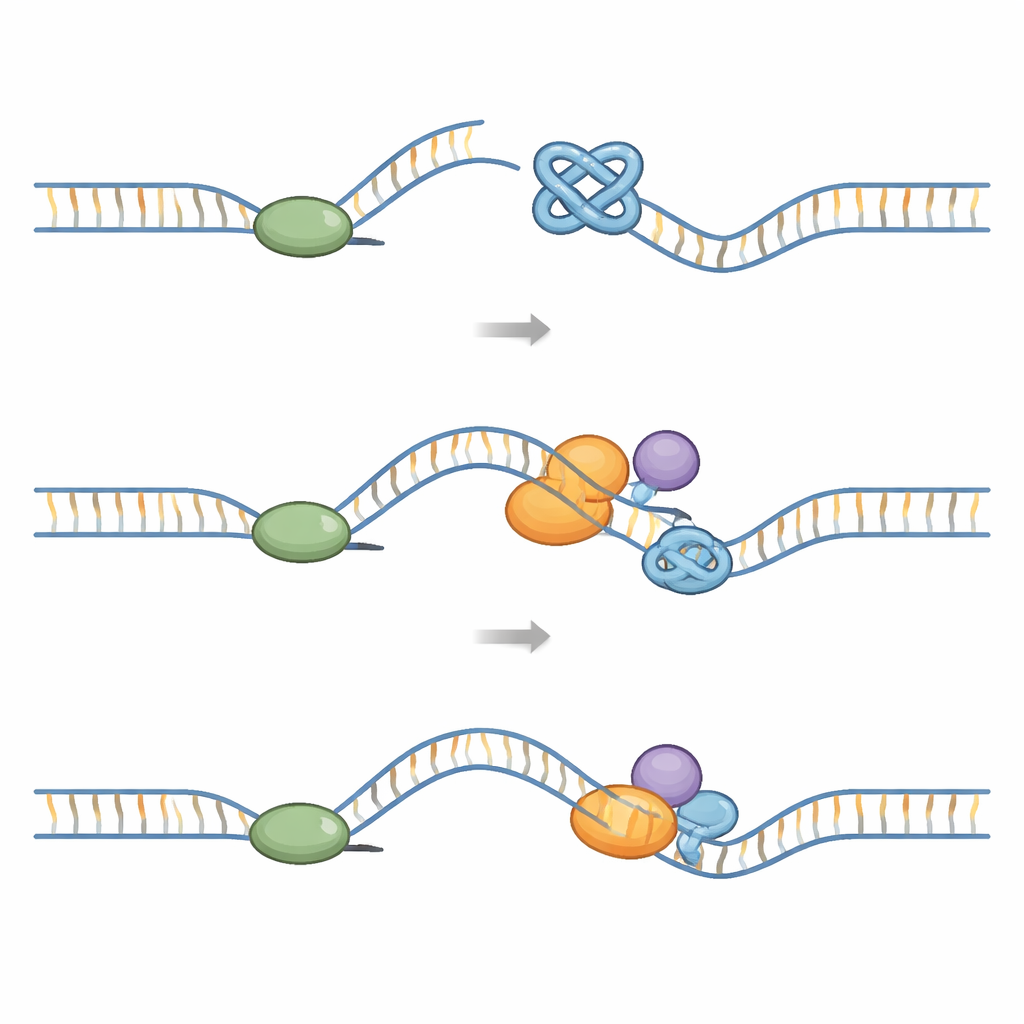
Every time a cell divides, a large “replisome” machine has to duplicate the entire genome. This machine includes a ring‑shaped helicase that opens the DNA and polymerases that build new strands. G‑quadruplexes pose a mechanical challenge: they are tougher to pull apart than ordinary DNA. Structural studies show that when the replisome meets a G‑quadruplex, the knot can lodge inside the helicase ring and block its movement. Other experiments reveal that G‑quadruplexes formed on the exposed template strand can stall the polymerase itself and cause the helicase and polymerase to become uncoupled. If these blocks are not cleared, the fork that is copying DNA can break, leading to DNA double‑strand breaks, chromosome rearrangements, and fragile sites, especially at telomeres.
Specialist Enzymes That Untie G‑Quadruplex Knots
Cells rely on a network of “helper” enzymes to prevent G‑quadruplexes from causing lasting harm. Several families of helicases—tiny motors that travel along DNA and unwind it—are particularly important. RecQ family helicases such as BLM, WRN, RECQL4, and RECQL1 can recognize and unfold G‑quadruplexes, including those at telomeres. A second group, the iron–sulfur (Fe–S) helicases like FANCJ, RTEL1, and DDX11, and enzymes such as DNA2 and PIF1, also dismantle G‑quadruplexes or clip them apart. Some have evolved special pockets or wedges that grab individual guanines or specific G‑quadruplex shapes, allowing them to peel these knots apart step by step while coordinating with DNA‑binding proteins and replication factors. Other remodelers, such as the DNA translocase HLTF and the G‑quadruplex/R‑loop helicase DHX36, reshape stalled forks or help the replisome bypass obstacles so copying can finish.
How Faulty DNA Helpers Cause Rare Disorders
Because these G‑quadruplex‑handling proteins also serve in broader repair and stress‑response roles, inherited mutations in their genes give rise to distinctive rare diseases. Defects in BLM cause Bloom syndrome, marked by growth deficiency, immune problems, and high cancer risk. Mutations in WRN lead to Werner syndrome, a form of premature aging with early cataracts, diabetes, and bone loss. Changes in RTEL1 underlie telomere‑related disorders such as dyskeratosis congenita and Hoyeraal–Hreidarsson syndrome, which feature bone‑marrow failure and very short telomeres. FANCJ and BRCA2 mutations are linked to Fanconi anemia and strong cancer susceptibility, while DDX11 mutations cause Warsaw Breakage Syndrome with microcephaly and cohesion defects between sister chromosomes. In many of these conditions, cells show accumulation of G‑quadruplexes, stalled replication forks, fragile telomeres, and chromosomal breaks—all hallmarks of G‑quadruplex‑induced replication stress.
From Rare Genetic Disorders to New Treatments
The authors argue that G‑quadruplexes sit at a crossroads between DNA structure, replication, and disease. When properly controlled, they may help regulate genes and telomeres; when not, they become roadblocks that damage the genome and contribute to rare syndromes, neurodegeneration, and possibly aspects of aging. Understanding exactly how different enzymes recognize and resolve specific G‑quadruplex shapes offers two opportunities: improving diagnosis of rare replication‑stress disorders and designing targeted therapies. On one side are drugs that stabilize G‑quadruplexes to selectively kill tumor cells with weak repair systems, as some early clinical trials suggest. On the other are emerging strategies—such as CRISPR‑guided proteins—that could one day tune G‑quadruplex formation at chosen sites to correct faulty gene control. In both cases, seeing DNA not just as a double helix but as a landscape of dynamic structures is key to understanding, and eventually treating, these diseases.
Citation: Herr, L.M., Mukhopadhyay, S., Anderson, O.M. et al. Rare genetic diseases associated with G-quadruplex-induced replication stress. Commun Biol 9, 522 (2026). https://doi.org/10.1038/s42003-026-09966-4
Keywords: G-quadruplex DNA, replication stress, rare genetic diseases, DNA helicases, telomere instability