Clear Sky Science · en
Multi-omics analysis reveals the key role of STIL in Li-Fraumeni syndrome and osteosarcoma
When Inherited Risk Meets Childhood Bone Cancer
Some families pass down more than eye color or height—they also inherit a much higher chance of developing cancer. One such condition, Li–Fraumeni syndrome, sharply raises the risk of osteosarcoma, a rare but aggressive bone cancer that often strikes children and teenagers. This study asks a pressing question: beyond the famous “guardian” gene TP53, what other molecules help turn this inherited risk into a fast‑growing, hard‑to-treat bone tumor, and could they become new targets for therapy?
A Hidden Connector Between Inherited Risk and Tumor Growth
The researchers focused on Li–Fraumeni syndrome, where a faulty copy of TP53 is present in every cell from birth. People with this syndrome are hundreds of times more likely to develop osteosarcoma than the general population. Using large collections of patient tissue and cell data, the team scanned thousands of genes to find those that are shared between Li–Fraumeni samples and osteosarcoma tumors. They discovered a small set of “bridge” genes whose activity was altered in both conditions and that clustered in processes controlling cell division and chromosome copying—core steps that, when disturbed, can drive cancer.
STIL Emerges as the Central Trouble‑Maker
By applying several machine‑learning methods, the scientists narrowed this bridge set down to four key genes and found one standout: a gene called STIL. In osteosarcoma samples, STIL levels were consistently higher in tumors that had already spread, and patients whose tumors made more STIL tended to live for a shorter time. Pathway analyses linked high STIL to brisk cell cycling and protein breakdown, while low STIL was associated with stronger immune and inflammation signals. This pattern suggested that STIL not only helps bone cancer cells divide but may also dampen the body’s natural defenses against them.
Stem‑Like Cells, Silent Immune System
To see where STIL acts inside a tumor, the team turned to single‑cell sequencing, which profiles individual cells one by one. They mapped more than 100,000 cells from osteosarcoma samples and identified several malignant cell types. STIL was especially abundant in a subset of highly “stem‑like” tumor cells that sat at the beginning of developmental paths—cells that appear to seed and sustain the cancer. In these cells, high STIL was tied to signals that encourage self‑renewal, activate bone‑eating cells that erode the skeleton, and reshape surrounding immune cells. Tumors rich in STIL showed fewer active killer T cells and key immune‑activating molecules, a pattern resembling an “immune desert” in which the immune system fails to recognize or attack the cancer effectively.
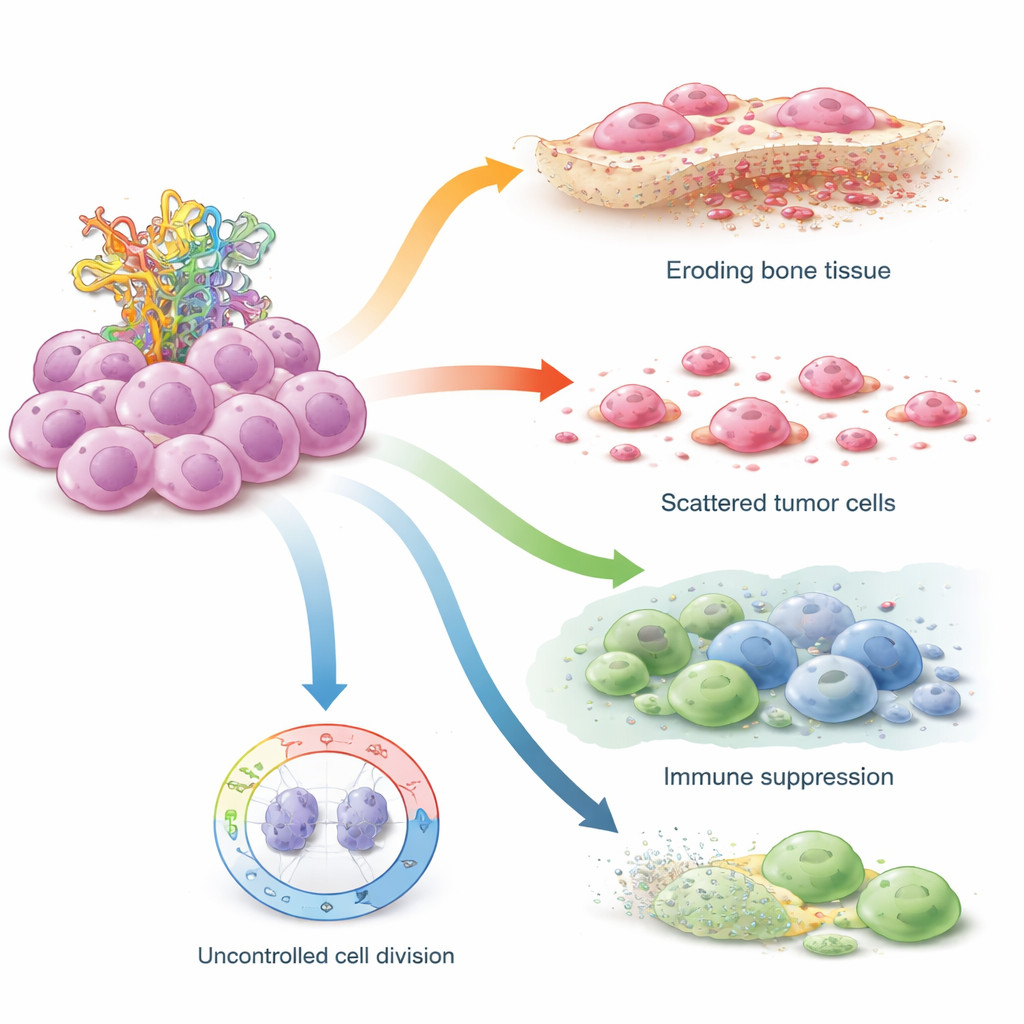
How STIL Undermines the Cell’s Safety Brakes
The researchers then tested STIL directly in osteosarcoma cell lines grown in the lab, comparing cells with normal TP53 to those carrying TP53 mutations that mimic Li–Fraumeni syndrome. When they used small RNA molecules to switch off STIL, levels of p53 protein rose in both cell types, and the cells formed fewer colonies and fewer free‑floating spheres—a hallmark of reduced stem‑like behavior. Cells with working TP53 were particularly sensitive, suggesting that STIL normally acts to weaken p53’s ability to halt the cell cycle. In TP53‑mutant cells, which already lack full p53 function, blocking STIL still had a strong impact on motility: aggressive mutant cells moved and “healed” wounds more slowly once STIL was removed, indicating that their invasive behavior depends heavily on this gene.
Implications for Future, More Precise Treatments
In everyday terms, this work paints STIL as an accomplice that helps inherited TP53 defects blossom into dangerous bone cancers. STIL both loosens the cell’s internal safety brakes and empowers a small pool of stem‑like tumor cells that can invade bone, spread to distant sites, and hide from immune attack. Because high STIL is also linked to altered sensitivity to certain drugs—such as WEE1 inhibitors that push already unstable cancer cells into lethal divisions—it may serve as a biomarker to match patients with tailored therapies. While more work is needed to test these ideas in patients, the study highlights STIL as a promising new handle for understanding and eventually disarming osteosarcoma in families living with Li–Fraumeni syndrome.
Citation: Qiao, Y., Hao, J., Yuan, F. et al. Multi-omics analysis reveals the key role of STIL in Li-Fraumeni syndrome and osteosarcoma. npj Precis. Onc. 10, 159 (2026). https://doi.org/10.1038/s41698-026-01432-y
Keywords: Li-Fraumeni syndrome, osteosarcoma, TP53, STIL, cancer stem cells