Clear Sky Science · en
A CXCR4 targeting peptide delivered by silica nanoparticles eliminates migrating cancer stem cells in pancreatic ductal adenocarcinoma
Why this research matters
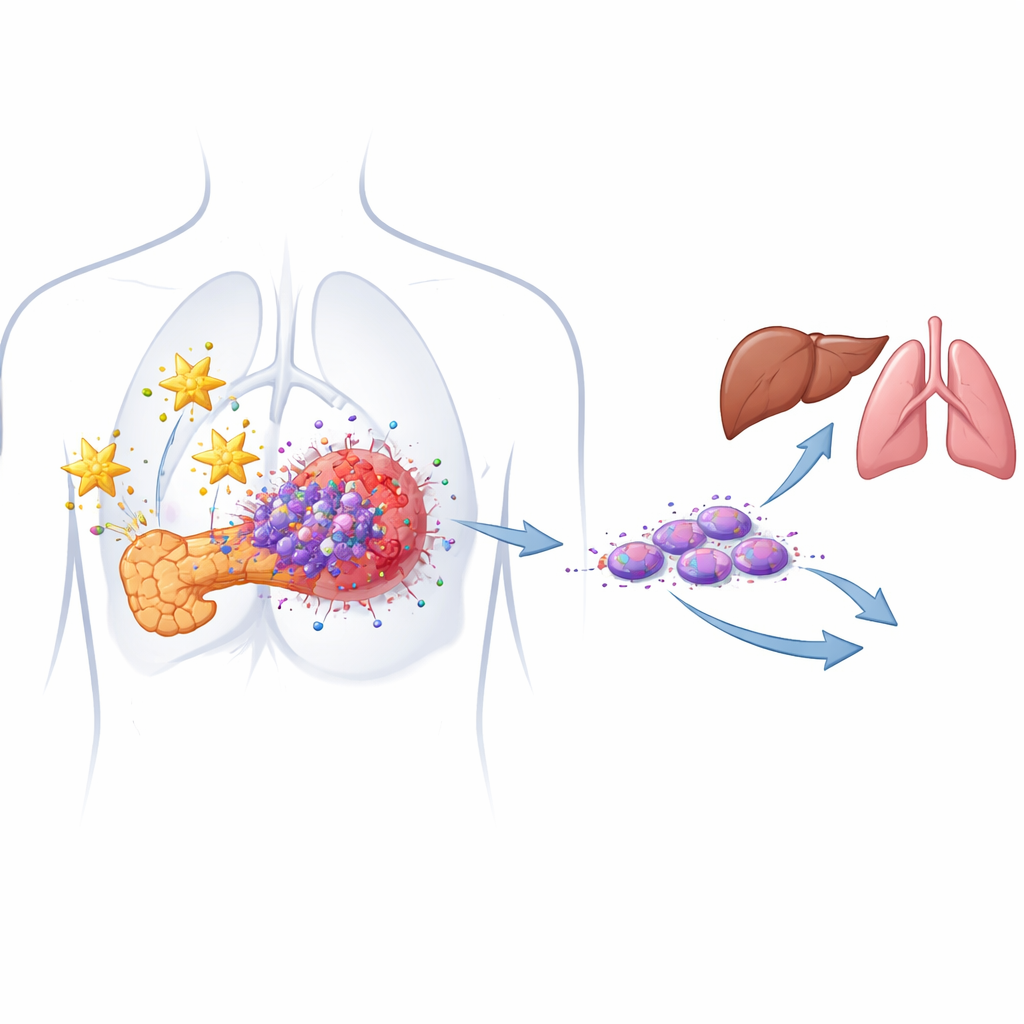
Pancreatic cancer is one of the deadliest cancers because it spreads early and resists almost every treatment doctors throw at it. A major culprit is a small, mobile group of tumor "seed" cells that travel through the body and restart the cancer elsewhere. This study explores how these wandering seeds are nurtured by their surroundings and introduces a new, highly targeted experimental therapy—delivered by tiny silica nanoparticles—that aims to track down and disarm them before they can cause further damage.
The hidden cancer seeds that drive relapse
Most pancreatic tumors are made of ordinary-looking cancer cells, but nestled within them is a minority population of so‑called cancer stem cells. These cells can self‑renew, survive chemotherapy, and regenerate a tumor after treatment. The authors previously identified an especially dangerous subset called migrating cancer stem cells, which carry two surface markers (CD133 and CXCR4) and are strongly linked to metastasis. In patient‑derived cell models, they now confirm that both markers are overproduced in pancreatic tumors compared with normal tissue and that higher levels of these cells correlate with more aggressive disease.
How the tumor’s neighbors fan the flames
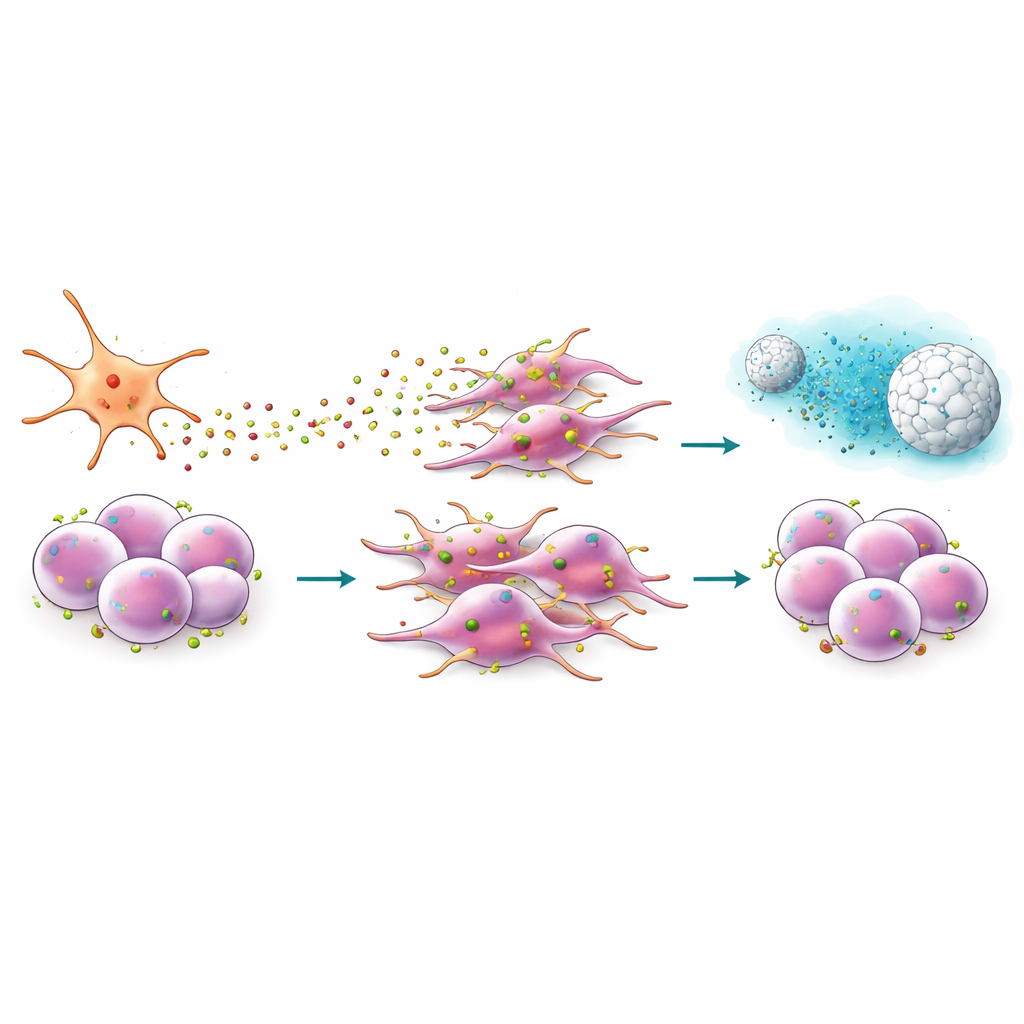
Pancreatic cancers grow in a dense, scar‑like tissue that is packed with support cells called pancreatic stellate cells. Far from being passive scaffolding, these stellate cells actively "coach" nearby cancer cells. The team recreated this interaction in the lab by co‑culturing patient‑derived tumor cells with stellate cells or exposing them to stellate‑cell–conditioned media. They found that stellate cells release a signaling molecule called CXCL12, which binds to the CXCR4 receptor on cancer stem cells. This crosstalk boosts stem‑like traits, encourages cells to shift from a compact, epithelial shape to a more mobile, mesenchymal form, and makes them more capable of forming spheres and migrating—features associated with metastasis and drug resistance.
A key molecular switch inside the cancer seeds
Digging deeper into the signaling network downstream of CXCR4, the researchers pinpointed a protein called BMI1 as a central control node. BMI1 is known to help normal stem cells maintain their self‑renewal. Here, when the scientists used genetic tools to reduce either CXCR4 or BMI1 in metastatic pancreatic cancer cells, the cells lost many of their stem‑like and migratory features. They formed fewer spheres, moved less toward CXCL12, and showed a shift back toward a more orderly, epithelial state. At the molecular level, blocking this pathway dampened pro‑survival and stress‑response circuits and reduced activation of pathways that normally fuel invasion and therapy resistance.
Designing a peptide and a nanoparticle to cut the signal
Because CXCR4 sits on the cell surface, it offers an attractive docking site for drugs. The team studied EPI‑X4, a naturally occurring human peptide that blocks CXCR4, and an engineered, more potent version called JM#21. In migration experiments, these peptides sharply reduced the ability of cancer cells to move toward CXCL12 and blunted changes in cell shape associated with invasion. They also lowered the activity of genes linked to stemness, epithelial‑to‑mesenchymal transition, and a developmental program (Hedgehog signaling) often hijacked by tumors. When combined with standard chemotherapies such as gemcitabine or paclitaxel, JM#21 made previously resistant cells far more vulnerable, shrinking the pool of surviving cancer stem cells after treatment.
Smuggling fragile drugs with silica nanoparticles
One obstacle to using peptides as drugs is that they are quickly broken down in blood and conventional culture media. To overcome this, the researchers loaded JM#21 into tiny, porous silica spheres—mesoporous silica nanoparticles—that act like protective sponges, slowly releasing the peptide while shielding it from degradation. In serum‑containing conditions that normally inactivate free JM#21, the nanoparticle‑bound form remained functional: it still blocked CXCL12‑driven migration and reduced the fraction of dangerous migrating cancer stem cells in co‑cultures of tumor and stellate cells. Gene‑level analyses showed that nanoparticle‑delivered JM#21 also toned down markers of drug resistance, antioxidant defenses, and BMI1 itself, while nudging cells back toward a less aggressive state.
What this could mean for patients
To a layperson, the message of this work is that pancreatic cancer’s most dangerous cells rely on a conversation with their surroundings and on a specific molecular switch to stay mobile, stem‑like, and drug resistant. By interrupting the CXCL12–CXCR4–BMI1 axis with a tailored peptide, and by packing that peptide into silica nanoparticles to keep it stable, the researchers show it is possible to weaken these metastatic seeds and make chemotherapy more effective in laboratory models. While further animal and clinical studies are needed, this approach offers a promising blueprint for therapies that do not just shrink tumors, but also target the root cells that drive spread and relapse.
Citation: Tiwary, K., Lahusen, A., Inaas, S. et al. A CXCR4 targeting peptide delivered by silica nanoparticles eliminates migrating cancer stem cells in pancreatic ductal adenocarcinoma. Sci Rep 16, 12588 (2026). https://doi.org/10.1038/s41598-026-48584-2
Keywords: pancreatic cancer, cancer stem cells, tumor microenvironment, CXCR4 inhibitors, nanoparticle drug delivery