Clear Sky Science · en
Role of the AmvAR efflux system on the pathogenesis of Acinetobacter baumannii
Why this hospital germ matters
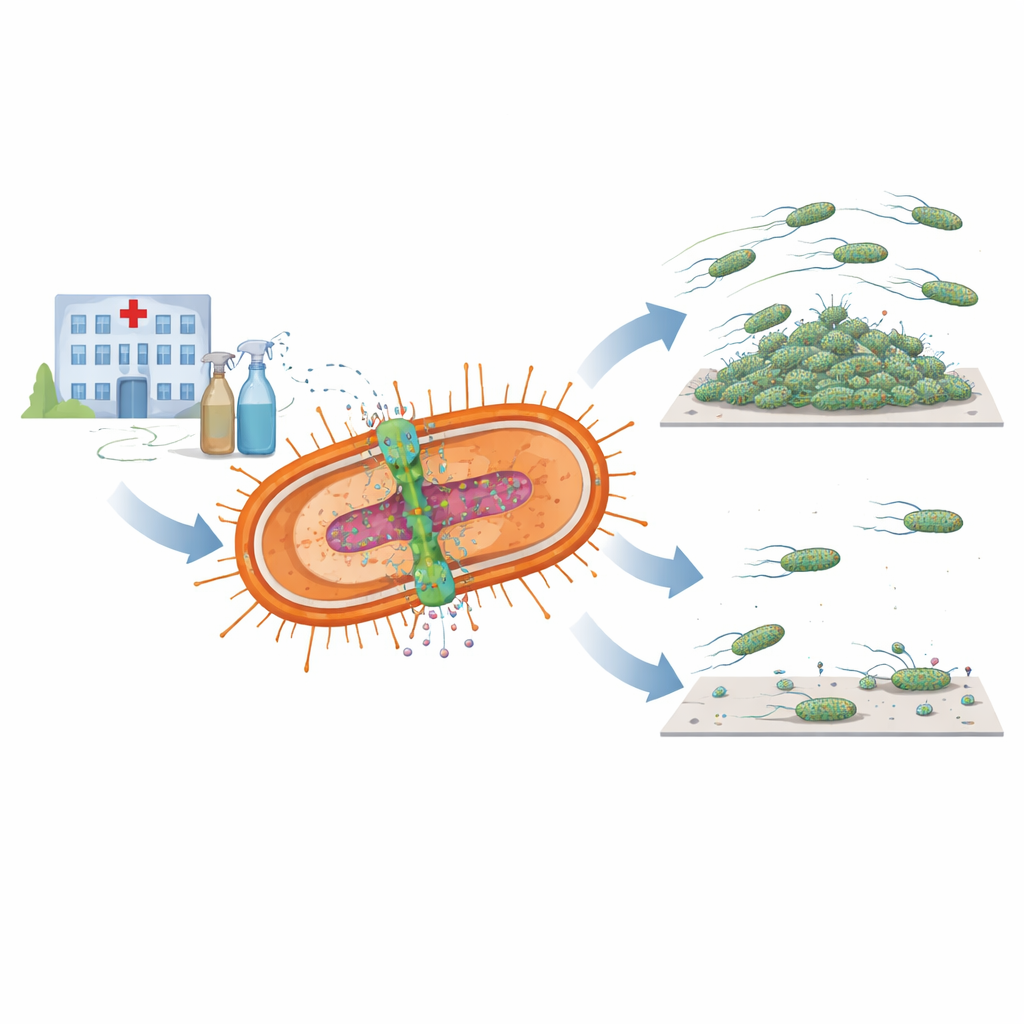
Acinetobacter baumannii is a hard-to-kill hospital germ that can cause pneumonia, blood infections, and serious wound and urinary tract infections, especially in very sick patients. It is notorious for shrugging off many antibiotics and even some disinfectants, making it a major concern on the World Health Organization’s list of critical threats. This study asks a key question: beyond helping the bacterium pump out toxic chemicals, does one of its defense systems also make it better at clinging to surfaces, forming slimy communities, and causing disease?
A double tool for survival
The researchers focused on a pair of closely linked bacterial components known together as the AmvAR system. One part, AmvA, sits in the cell membrane and works like a microscopic pump that pushes disinfectants and other harmful molecules out of the cell. The other part, AmvR, acts as a genetic on–off switch that normally keeps the pump in check. By scanning hundreds of bacterial genomes, the team found versions of both parts in almost all available A. baumannii strains, hinting that this system is central to how the germ survives harsh conditions in hospitals, including frequent contact with disinfectants like chlorhexidine.
How changing this system alters movement and slime
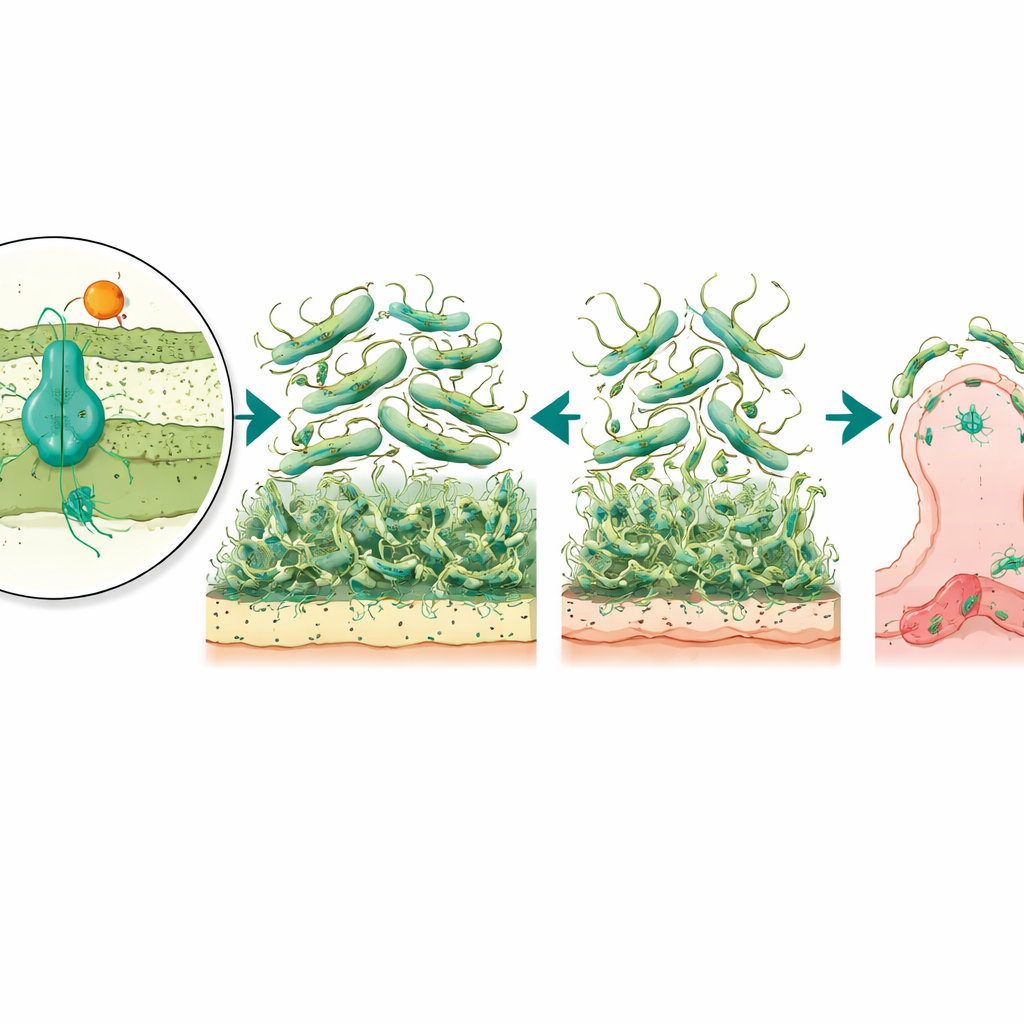
To understand what AmvAR does beyond drug resistance, the scientists created mutant bacteria lacking either the pump or its regulator and compared them to the normal strain. They saw that removing AmvR, the regulator, made the bacterium less able to spread across a soft surface, showing impaired motility. Both mutants, lacking either the pump or the regulator, formed much weaker biofilms: the sticky, layered communities on plastic and glass that help bacteria cling to medical devices and resist cleaning. When the researchers reintroduced the missing genes, they could restore or even boost biofilm formation, suggesting that the right balance of this system is crucial for building stable surface communities.
How the bacterium grips host cells
Because biofilms and surface structures help bacteria grab onto human tissues, the team next tested how well the mutants could attach to bladder cells grown in the lab, a model for urinary tract infections. Both mutants stuck far less effectively than the normal strain, with the pump-lacking version showing the greatest drop in attachment. This suggests that the AmvAR system not only helps the bacterium resist disinfectants but also fine-tunes its outer surface so it can latch onto our cells. Supporting this idea, the regulator mutant showed a dramatic overproduction of a pilus subunit, a building block of hair-like fibers that mediate surface attachment. Paradoxically, this overproduction was linked to poorer biofilms and movement, implying that too many or poorly coordinated fibers can actually disrupt normal surface behavior.
From wax worms to disease severity
To connect these laboratory findings to real infection, the scientists turned to a living model: larvae of the wax moth Galleria mellonella, an established stand-in for studying bacterial virulence. When infected with the normal A. baumannii strain, many larvae died, but those infected with the regulator-lacking mutant survived at much higher rates. In contrast, removing only the pump left overall disease severity unchanged. This pattern suggests that AmvR, the regulator, plays a broader coordinating role in controlling traits like movement, biofilm formation, and cell attachment that together determine how well the bacterium can invade and persist inside a host.
What this means for future treatments
In simple terms, this work shows that the AmvAR system in A. baumannii is more than a chemical pump: it helps shape the germ’s outer coat, its ability to move, to glue itself onto surfaces, to build protective slime layers, and ultimately to cause disease. Disrupting only the pump may not be enough to tame the infection, but interfering with the regulator or its connected pathways could weaken several key survival tricks at once. That makes AmvAR, and similar control systems, promising targets for new strategies aimed at both overcoming antibiotic resistance and dialing down the virulence of this stubborn hospital pathogen.
Citation: Gaona, M., Corral, J., Sánchez-Osuna, M. et al. Role of the AmvAR efflux system on the pathogenesis of Acinetobacter baumannii. Sci Rep 16, 10753 (2026). https://doi.org/10.1038/s41598-026-46007-w
Keywords: Acinetobacter baumannii, efflux pumps, biofilm, antimicrobial resistance, bacterial virulence