Clear Sky Science · en
Integrated electrical modeling of circulating tumor cells for enhanced dielectrophoretic trapping and electroporation
Why tiny electric jolts to blood cells matter
Cancer can shed a few rogue cells into the bloodstream long before a tumor is found on a scan. These circulating tumor cells are precious clues for early diagnosis and tailor‑made treatments, but they are rare and hard to handle without harming the rest of the blood. This paper explores how carefully shaped electric fields inside a small lab‑on‑a‑chip device can gently catch these wandering cancer cells, open tiny temporary gates in their membranes, and then let them recover — all while leaving healthy blood cells largely unharmed.
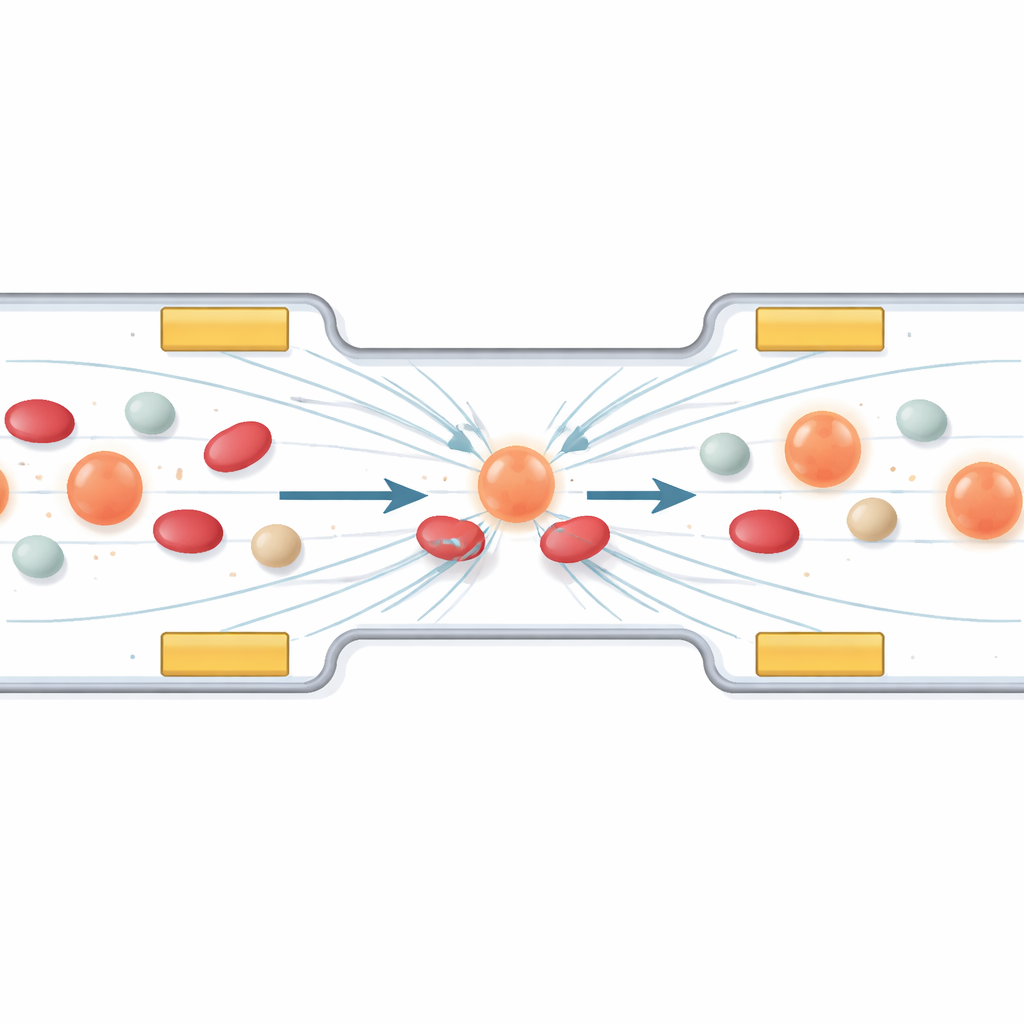
Using invisible forces to catch wandering cancer cells
The first part of the work looks at how to steer and collect specific cells using a phenomenon called dielectrophoresis. In simple terms, when an alternating electric field is applied across a microchannel, cells feel tiny pushes and pulls that depend on their size and electrical makeup. The authors design an arrangement of miniature electrodes so that circulating tumor cells drift into a stable “parking lane” between them, while smaller or less polarizable blood components pass by. Computer simulations show how changing the voltage and frequency shapes the field, guiding cells away from high‑field zones and lining them up in the right region for treatment. A protective layer over the electrodes keeps the cells from being damaged by direct contact.
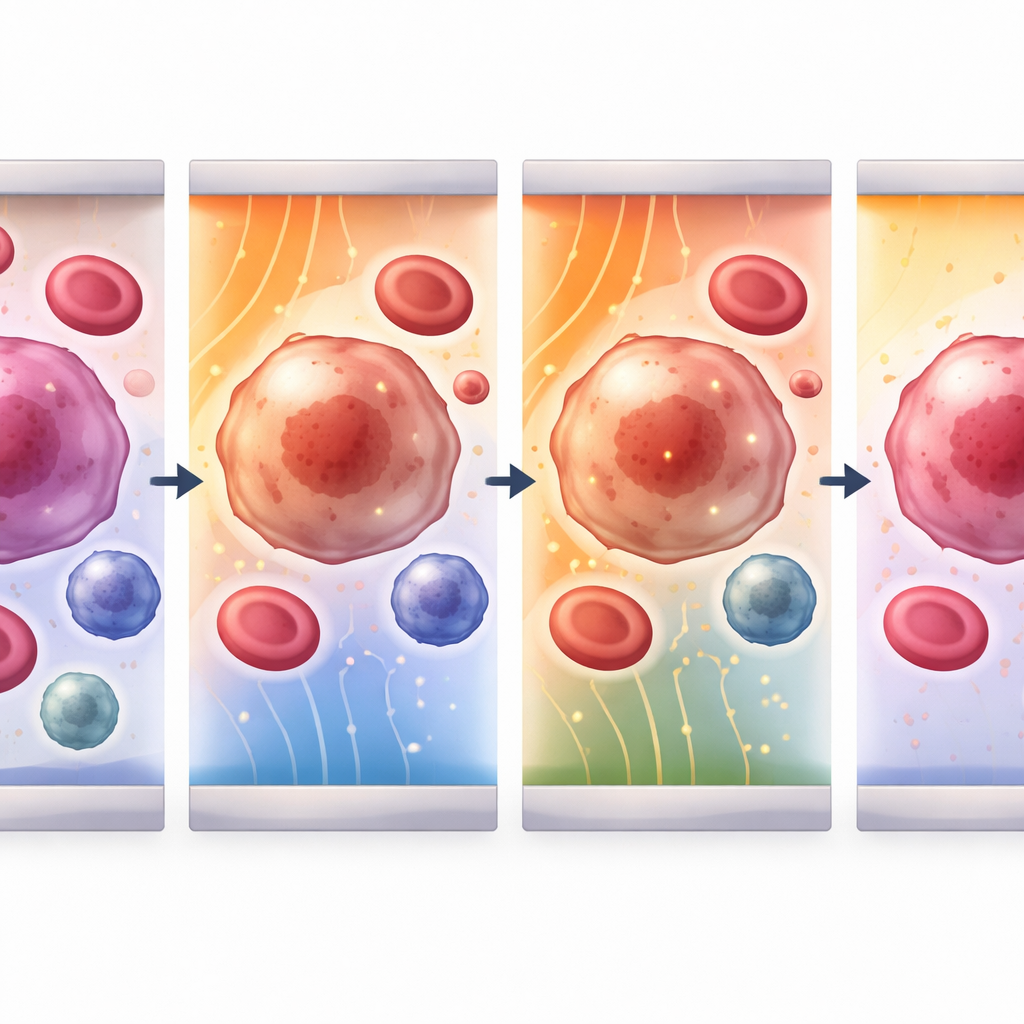
Opening and closing microscopic doors in cell skins
Once cells are held in place, the device delivers ultrashort electrical pulses that briefly punch nanometer‑scale pores into the outer membrane. The study builds a detailed mathematical model of this process for three key players in blood: circulating tumor cells, white blood cells, and platelets. It tracks how the voltage across each cell membrane rises, when pores first appear, how big and how numerous they become, and how quickly the membrane reseals. By tuning both field strength and pulse length, the team identifies sweet spots where the membrane becomes leaky enough to admit drugs or genetic material but then heals without killing the cell. For tumor and white blood cells, effective fields fall in the range of 1–4 kilovolts per centimeter, whereas the much smaller platelets need about ten times stronger fields to reach the same effect.
Different blood cells, different sweet spots
The modeling reveals how cell size and electrical properties change the way each cell responds. Larger circulating tumor cells and white blood cells charge more slowly and reach peak membrane voltage around two microseconds into the pulse; their pores grow and then close over tens of microseconds to seconds. Tiny platelets, by contrast, respond almost instantly, with faster pore formation and resealing. Across all cell types, the most intense action happens at the “hyperpolarized” side of the cell facing the incoming field, where the forces concentrate. This means that by choosing the right field strength for a given cell size, it is possible to favor opening pores in tumor cells while keeping damage to healthy neighbors low, laying the groundwork for highly selective treatments.
Testing the idea with real cells on a chip
To check whether the simulations match reality, the authors built a microfluidic chip that combines trapping electrodes with a central sensing zone. They flowed a model white blood cell line (THP‑1 cells) through the channel and applied increasing voltages across a fixed gap. Instead of watching the pores directly, they measured how easily an alternating current passed through the suspension. As the electric field grew, the overall resistance of the cell mixture dropped and its capacitive behavior faded, signatures that the membranes had become more conductive and less like intact insulators. At the highest tested fields, the electrical response was dominated by conduction through open pores, closely mirroring the trends predicted by the computer model.
What this could mean for future cancer care
Put simply, this study shows that one compact chip can both sort out rare cancer cells from blood and gently open their membranes in a highly controlled way. By tailoring pulse strength and timing to each cell type, the approach can either keep cells alive for detailed analysis and drug testing or push them toward irreversible damage for targeted destruction, all while minimizing heating and harm to healthy cells. For a lay audience, the key message is that with the right “electrical choreography,” we can turn tiny currents into powerful tools for early cancer detection, precision drug delivery, and possibly safer, more focused cancer therapies.
Citation: Sherif, S., Ghallab, Y.H. & Ismail, Y. Integrated electrical modeling of circulating tumor cells for enhanced dielectrophoretic trapping and electroporation. Sci Rep 16, 12072 (2026). https://doi.org/10.1038/s41598-026-45747-z
Keywords: circulating tumor cells, microfluidic electroporation, dielectrophoresis, cancer diagnostics, lab-on-a-chip