Clear Sky Science · en
Cilostazol mitigates amiodarone-induced pulmonary toxicity and fibrosis by regulating the cAMP/TGF-β1 pathway-mediated epithelial-to-mesenchymal transition in rats
Why this lung study matters
Pulmonary fibrosis is a slow, scarring injury to the lungs that makes every breath harder, and it can be triggered by some life-saving heart drugs. This study in rats explores whether an existing medication, cilostazol, can shield the lungs from damage caused by the widely used antiarrhythmic drug amiodarone. By tracing both the visible lung injury and the hidden chemical signals inside lung cells, the researchers ask a hopeful question: can we keep vital heart treatments while better protecting the lungs?
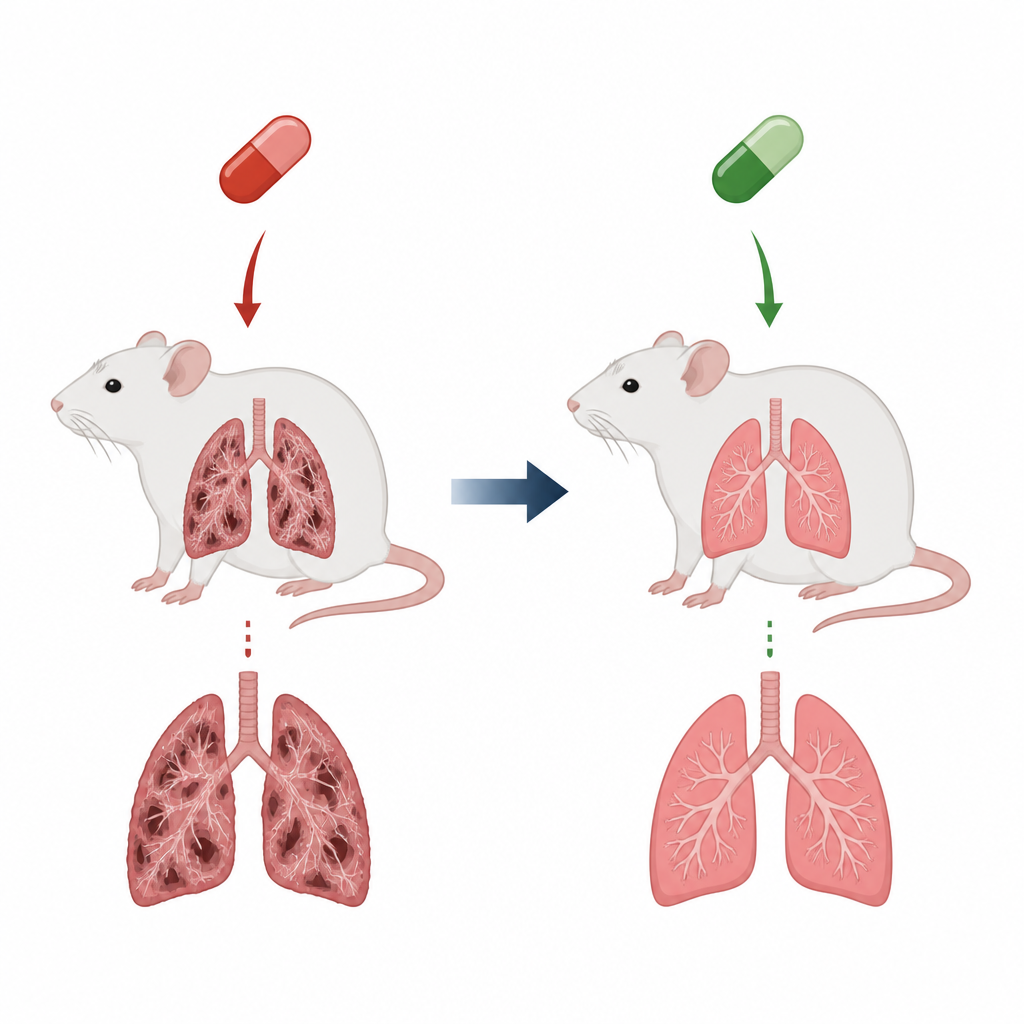
A heart drug with a hidden cost
Amiodarone is prescribed to correct dangerous heart rhythm problems, but a serious drawback is its potential to damage lung tissue and lead to pulmonary fibrosis. In fibrosis, the delicate air sacs that exchange oxygen become stiff and scarred, making breathing labored and life expectancy shorter. In this rat model, daily amiodarone doses for several weeks reproduced many features of this condition: heavier lungs, inflamed airways filled with immune cells, and thick bands of collagen that distorted normal lung structure. These changes mirror what clinicians fear in patients who develop drug-related scarring in their lungs.
Testing a protective partner drug
Cilostazol is a pill already used to improve blood flow in clogged leg arteries and to reduce certain types of stroke risk. It works in part by raising levels of a small signaling molecule called cAMP, which can calm inflammation and limit scarring in tissues. The researchers divided rats into three groups: healthy controls, animals receiving amiodarone alone, and animals given cilostazol before each amiodarone dose. They then collected fluid from the lungs, examined tissue slices under the microscope, and measured chemical markers of oxidation, inflammation, and scarring to see whether cilostazol could blunt the lung damage.
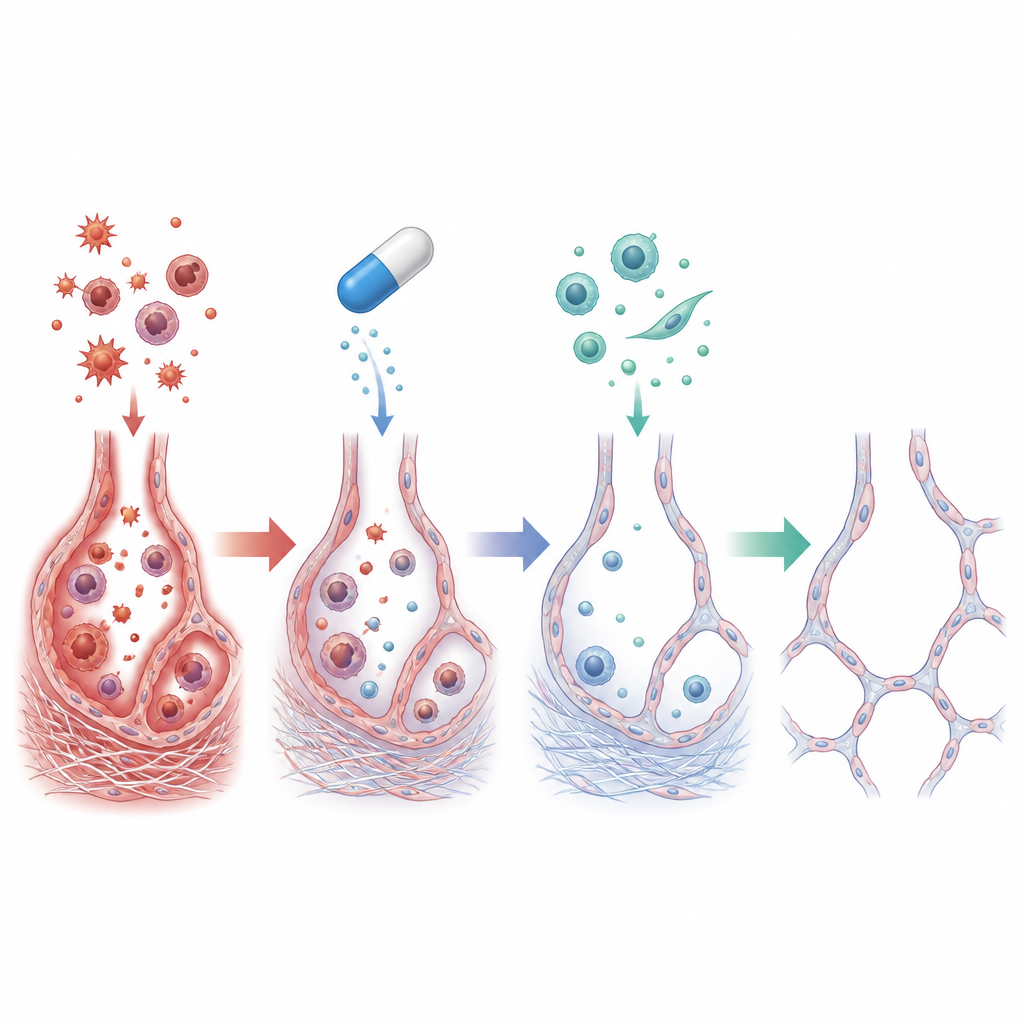
What happened inside the injured lungs
In rats that received only amiodarone, the lungs showed intense inflammation: immune cell counts in airway fluid surged, and levels of inflammatory messengers such as TNF-alpha and IL-1 beta rose several-fold. Chemical fingerprints of oxidative stress increased, with more lipid damage and depleted antioxidant defenses. Under the microscope, the team saw collapsed and distorted air sacs surrounded by heavy collagen deposits. Deeper inside the cells, there were signs that epithelial cells lining the air sacs were starting to behave more like scar-forming cells, a shift known as epithelial-to-mesenchymal transition that drives long-lasting fibrosis.
How cilostazol changed the picture
When cilostazol was added, many of these harmful changes were sharply reduced. Lungs from treated rats weighed less and looked far closer to normal, with much milder collagen build-up and better-preserved air sacs. Inflammatory cells and cytokines dropped toward control levels, and oxidative damage markers declined while natural antioxidants rebounded. At the molecular level, cilostazol boosted cAMP and increased activity of protective players such as SIRT1 and EPAC1, while dialing down signals linked to scarring, including TGF-beta1 and vimentin. Together, these shifts suggest that cilostazol not only quieted inflammation and oxidative stress but also interrupted the cellular reprogramming that turns healthy lining cells into scar-producing cells.
From rat lungs to future treatment ideas
The study concludes that cilostazol can markedly soften amiodarone-induced lung injury and scarring in rats by restoring a healthier balance of cell signals and blocking key steps that lead to permanent fibrosis. While these findings do not yet show how cilostazol will perform in people, they point to a practical strategy: repurposing an approved drug to protect the lungs of patients who rely on powerful heart medicines. Further work in different models and, eventually, clinical trials will be needed, but the results offer a clear message for non-specialists: some of the harm caused by essential drugs may be preventable by pairing them with carefully chosen partners.
Citation: El-Gammal, M.A., Yousef, E.H., Abd Elhameed, A.G. et al. Cilostazol mitigates amiodarone-induced pulmonary toxicity and fibrosis by regulating the cAMP/TGF-β1 pathway-mediated epithelial-to-mesenchymal transition in rats. Sci Rep 16, 15055 (2026). https://doi.org/10.1038/s41598-026-45341-3
Keywords: pulmonary fibrosis, amiodarone lung toxicity, cilostazol, drug-induced lung disease, cAMP signaling