Clear Sky Science · en
Identification of novel blood-borne soluble binding partners of factor H-related proteins
Why tiny blood proteins matter
Our immune system relies on a powerful chemical cascade, called complement, to fight germs and clear cellular debris. When this system is not properly controlled, it can start to damage our own tissues and contribute to diseases such as age‑related macular degeneration (a leading cause of blindness) and certain kidney disorders. This study looks at a little‑known group of blood proteins, the factor H‑related (FHR) proteins, and asks a simple but crucial question: which other molecules in the blood do they latch onto, and how might those partnerships tip the balance between healthy protection and harmful inflammation? 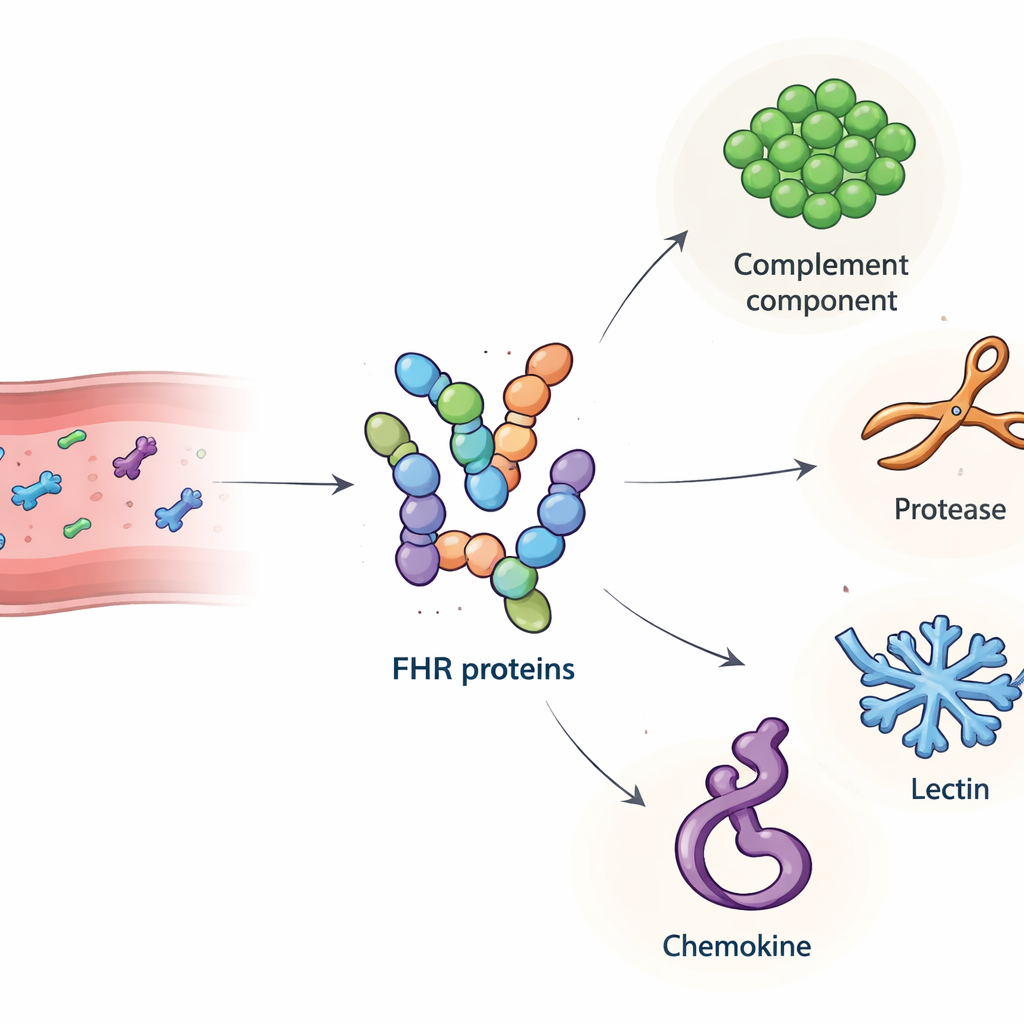
A family of immune fine‑tuners
Complement factor H is a well‑studied “brake” on the complement cascade, preventing over‑activation on our own cells. FHR proteins are close relatives of factor H, but they lack the key region that directly turns complement off. Instead, FHRs can compete with factor H for binding sites, and in some settings they seem to act more like accelerators than brakes. Genetic studies link changes in FHR levels or structure to diseases of the eye, kidney and joints, yet the exact ways these proteins work in the bloodstream have remained murky. Because FHRs are mainly made in the liver and then travel throughout the circulation before settling into tissues, the authors reasoned that discovering their soluble blood‑borne partners could reveal new layers of immune control.
Fishing for partners in the bloodstream
The team first produced highly purified versions of all major human FHR proteins, plus the shorter factor H‑like protein 1 (FHL‑1), in cultured cells. They confirmed that these lab‑made proteins behaved like their natural counterparts, including binding to known targets and to heparin, a sugar that mimics structures in tissue matrices. Next, they used each FHR as bait: immobilizing it on tiny beads, incubating the beads with human serum, and then using mass spectrometry to identify which blood proteins stuck. This screening turned up 34 candidate partners. From these, the researchers focused on four secreted molecules with clear immune roles and clean signals in the assay: complement component C4, the enzyme cathepsin G (CTSG), mannose‑binding lectin 2 (MBL2), and a platelet‑derived chemokine called platelet basic protein (PPBP). Follow‑up plate‑based binding tests confirmed that these interactions are direct and selective, with different FHR family members showing distinct binding patterns to each partner. 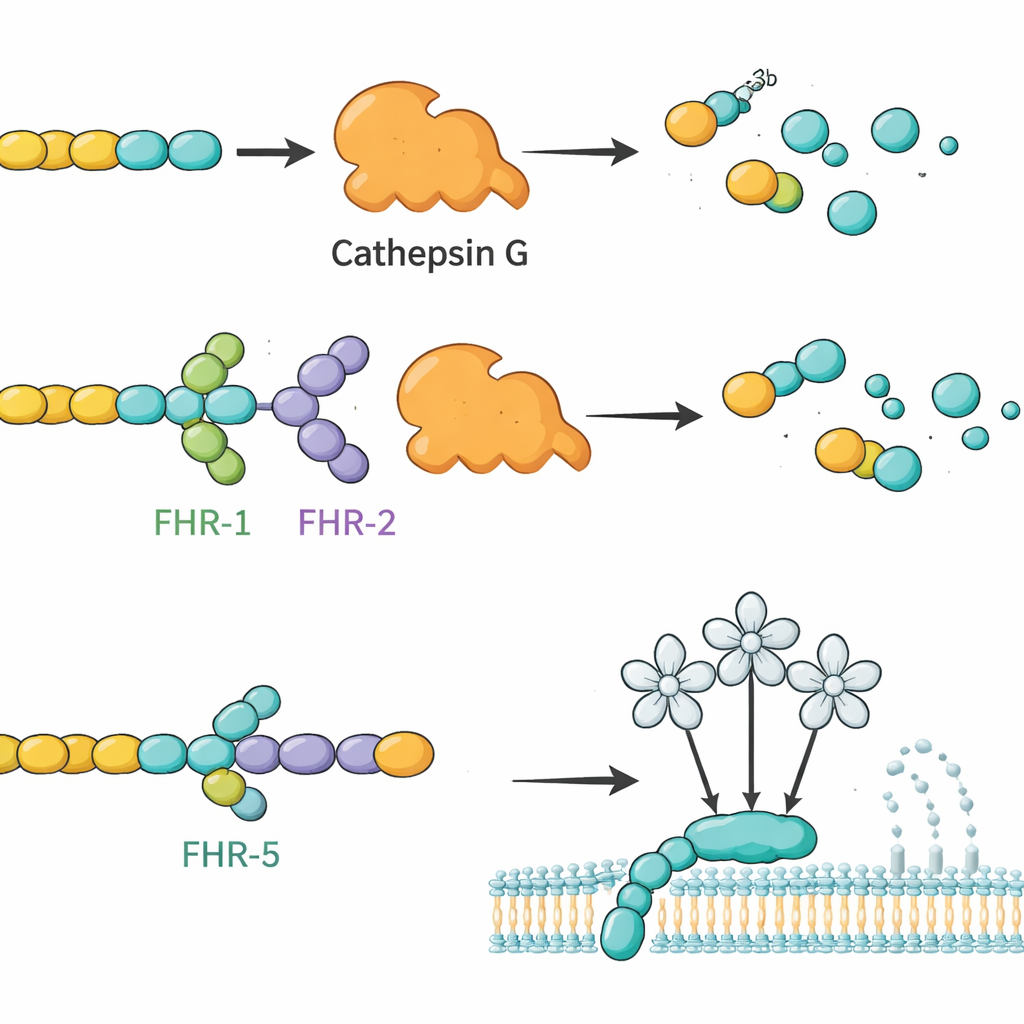
How new links reshape complement activity
Building on these binding maps, the authors asked whether FHRs could change what these partners actually do. They focused first on C3b, a central complement fragment, and CTSG, a neutrophil enzyme known to chop up C3b without help from other factors. In test‑tube reactions, CTSG readily degraded C3b into inactive pieces. When FHR‑1 or FHR‑2 was added, this breakdown slowed noticeably, suggesting these FHRs act as competitive regulators that shield C3b from CTSG. In contrast, factor H and FHL‑1 made CTSG‑driven cleavage faster, underscoring how closely related proteins can have opposite functional effects. The group also probed how FHRs influence the lectin arm of complement, which is triggered when MBL2 recognizes certain sugar patterns. Here, FHR‑5 (and to a lesser extent FHL‑1 and factor H) dampened lectin pathway activity in a dose‑dependent manner, consistent with FHR‑5 binding to MBL2 and modulating its ability to start the cascade.
Connecting immune attack, clotting, and tissue damage
The newly confirmed interactions with C4 and PPBP hint at broader crosstalk between complement, blood clotting, and inflammation. C4 is a key early component of the classical complement pathway, while PPBP is released by activated platelets and helps recruit white blood cells. The finding that several FHRs bind PPBP suggests that these complement‑linked proteins may not only tune chemical cascades on cell surfaces, but also intersect with how immune cells are drawn into inflamed tissues, including the retina and kidneys. Although the exact consequences of these bindings in living organisms remain to be worked out, they fit with a growing picture in which FHR proteins help position and shape immune reactions at specific tissue sites, rather than acting as simple on/off switches.
What this means for disease and treatment
Taken together, this work expands the “interactome” of the FHR family and reveals that these proteins engage a network of soluble partners involved in complement activation, tissue remodeling, and cell recruitment. For a lay audience, the key message is that small changes in how FHRs bind to other blood proteins can influence whether complement quietly maintains tissue health or spirals into self‑damage. Because genetic variants that alter FHRs are linked to conditions like age‑related macular degeneration, IgA nephropathy, and other complement‑mediated disorders, these newly mapped interactions offer fresh clues for why some people are more vulnerable to such diseases. In the long run, targeting specific FHR partnerships—such as the FHR‑1/FHR‑2 interaction with CTSG or the FHR‑5 interaction with MBL2—could inspire more precise therapies that restore balance to the complement system without shutting down our natural defenses.
Citation: Tang, J., Woerz, F., Beyer, T. et al. Identification of novel blood-borne soluble binding partners of factor H-related proteins. Sci Rep 16, 9651 (2026). https://doi.org/10.1038/s41598-026-44779-9
Keywords: complement system, factor H-related proteins, age-related macular degeneration, innate immunity, protein–protein interactions